Eliquis Bleeding Injury Resulted in Hospitalization, Lawsuit Claims
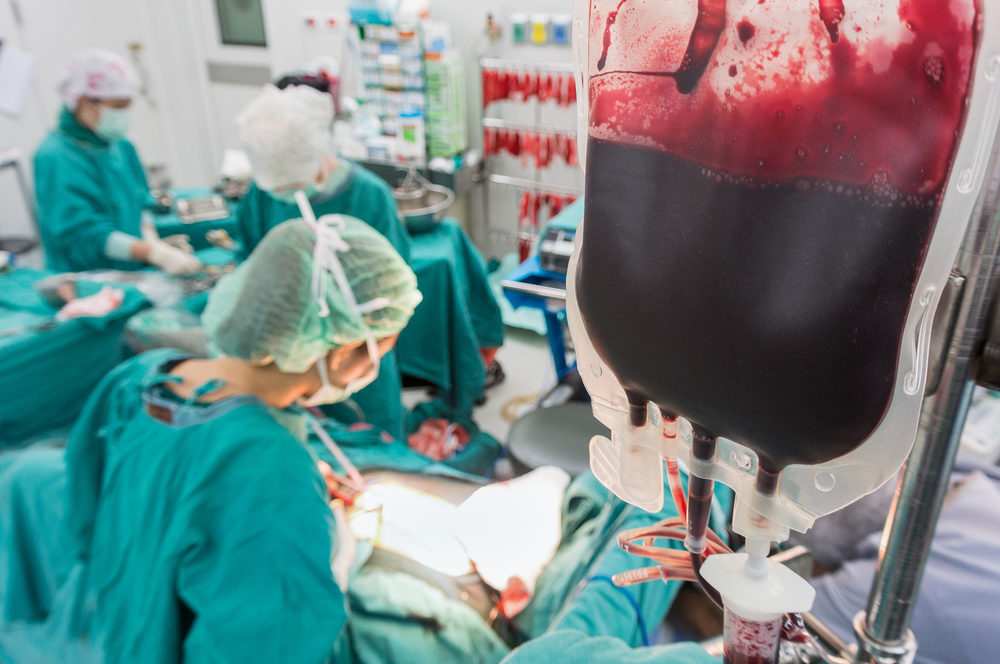
According to allegations raised in a lawsuit filed against the makers of Eliquis, side effects of the controversial new-generation anticoagulant caused a California woman to suffer a severe bleeding injury, which led to extensive hospitalization.
The complaint (PDF) was filed this week in Delaware Superior Court by Carol Woody, indicating that Bristol-Myers Squibb and Pfizer failed to adequately warn users and the medical community about the risk of severe bleeding injury from Eliquis.
Eliquis is part of a new crop of oral anticoagulants introduced in recent years, which also includes the other blockbuster treatments Xarelto and Pradaxa. The drugs have been aggressively marketed as a replacement for Coumadin (warfarin), which has been the go-to anticoagulation treatment for decades. However, unlike warfarin, where the blood thinning effects of the medication can be reversed in an emergency to stop bleeding problems, there was no approved reversal agent when Eliquis, Pradaxa or Xarelto were introduced.

Did You Know?
Millions of Philips CPAP Machines Recalled
Philips DreamStation, CPAP and BiPAP machines sold in recent years may pose a risk of cancer, lung damage and other injuries.
Learn MoreWoody indicates that she was prescribed Eliquis to prevent strokes from atrial fibrillation. However, in November 2014 she suffered a gastrointestinal hemorrhage, which was so severe that she required “extensive” blood transfusions due to the uncontrollable bleeding injury, and required an extended hospital stay.
The lawsuit indicates that the manufacturers gave false information about the safety of Eliquis and that they knew or should have known that the drug could cause bleeding events that could become uncontrollable due to the lack of an antidote, also known as a reversal agent, for its blood thinning effects.
“In written information about the safety and risks of Eliquis, Defendants negligently and fraudulently represented to the medical and healthcare community, including Plaintiff’s prescribing doctor, the Food and Drug Administration (hereinafter referred to as the ‘FDA’) , to Plaintiff and the public in general, that Eliquis had been tested and was found to be safe and effective for its indicated uses,” the lawsuit states. “Defendants concealed their knowledge of Eliquis’ defects, from Plaintiff, the FDA, the public in general and the medical community, including Plaintiff’s prescribing doctor.”
Since the new-generation oral anticoagulant was introduced in 2014, large number of serious or fatal Eliquis bleeding injuries have been reported nationwide, since doctors were often left unable to stop or control gastrointestinal bleeds, brain bleeds and other complications.
The case joins a growing number of similar Eliquis lawsuits being filed in courts nationwide, many of which involve wrongful death claims. Each of the cases raise similar allegations that the drug makers knew or should have known that individuals face serious risks, yet withheld information about the lack of an effective reversal agent.
Amid an increasing number of cases filed in courts nationwide, Bristol-Myers Squibb and Pfizer filed a request in October with the U.S. Judicial Panel on Multidistrict Litigation (JPML), seeking to transfer all Eliquis failure to warn cases to one judge for coordinated discovery and pretrial proceedings, suggesting that the number of complaints is likely to continue to increase over the coming weeks and months.
The JPML heard oral arguments on January 26, and decided a month later that a multidistrict litigation (MDL) should be established for the Eliquis cases, transferring all lawsuits pending throughout the federal court system to U.S. District Judge Denise L. Cote in the Southern District of New York for coordinated pretrial proceedings.
Similar MDL proceedings have previously been established for all Pradaxa lawsuits and Xarelto lawsuits, which raised nearly identical allegations about the same risks associated with these other new-generation anticoagulants, which hit the market before Eliquis was introduced.
Following several years of litigation over the failure to warn about the bleeding risk with Pradaxa, which was the first member of this class of drugs to enter the market, Boehringer Ingelheim agreed to pay $650 million in Pradaxa settlements to resolve about 4,000 cases in 2014.
The Xarelto litigation is currently centralized in the federal court system, where about 16,000 complaints are pending before U.S. District Judge Eldon Fallon in the Eastern District of Louisiana, with the first of a series of early “bellwether” trials currently underway.
Over the coming months and years, as Eliquis bleeding injury lawyers continue to review and file cases for individuals nationwide, it is ultimately expected that several thousand related cases will be filed against Bristol-Myers Squibb and Pfizer.
Get more articles like this sent directly to your inbox.
"*" indicates required fields
3 Comments






ambresMay 21, 2017 at 11:14 pm
I have been taking eliquis for 4 months now .been having stomach problems back in four to the bathroom sleeping all time .anariztium in my stomach that goes to my heart .all ways week
RaymondMay 11, 2017 at 7:30 am
Why is the number of victims allowed to go so high ? Why are these drugs not pulled off the market at least untill a satisfactory judgement ? Why is FDA allowing these companys to keep marketing these drugs and making more people into victims ? Why are doctors still prescribing these horrific drugs ?
DianaMay 3, 2017 at 11:33 pm
I started this drug in january, 2017, and passing blood in my urine four weeks ago. Doctors will not take me off of it. I was treated for a kidney infection with antibiotics and still have blood in my urine. I am really scared.