Surgical Stapler Risks Targeted By New FDA Rules and Guidelines To Make Devices Safer
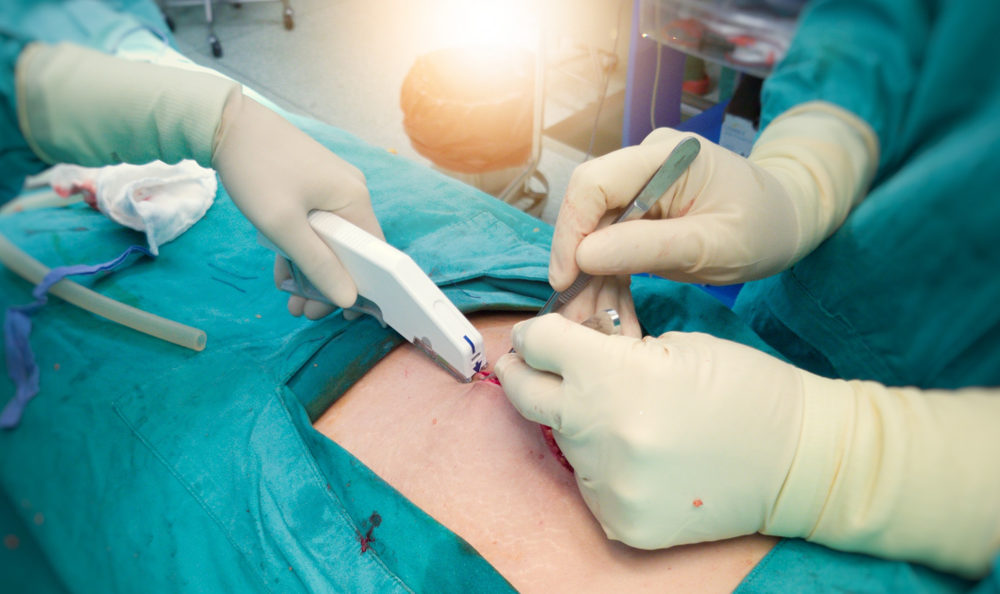
Following reports of serious and often life-threatening surgical stapler risks which have plagued certain products sold in recent years, new federal regulations have been introduced which will require a more stringent review process before devices are approved for use during medical procedures.
On October 7, the U.S. Food and Drug Administration (FDA) announced it has finalized new rules and guidelines designed to improve surgical stapler safety, including a final rule reclassifying them as Class II surgical devices, which will now require any new surgical staplers to undergo premarket review before getting FDA approval.
The change of surgical staplers from class I medical device (general controls) to class II medical devices (special controls) has been anticipated for several years, and does not affect the classification of the staples themselves.

Did You Know?
Millions of Philips CPAP Machines Recalled
Philips DreamStation, CPAP and BiPAP machines sold in recent years may pose a risk of cancer, lung damage and other injuries.
Learn MoreIn March 2019, the FDA warned about the rising rate of problems with surgical staplers, indicating that at least 41,000 adverse event reports were received between January 2011 and March 2018, including 366 patient deaths. As a result of the problems, the FDA proposed new rules and guidelines designed to make surgical staplers safer in April 2019, and decided to classify the products as moderate risk medical devices, as opposed to low-risk devices.
The new classification requires more stringent pre-market approval for new surgical staplers and forces manufacturers to provide more thorough safety warnings and instructions.
“The increasing reliance on surgical staplers by surgeons to perform more procedures that are minimally invasive, together with the agency’s analysis of adverse events associated with surgical staplers and implantable staples, prompted the FDA to increase regulatory oversight of these devices while continuing to educate health care providers and patients about their benefits and risks,” Dr. William Maisel, chief medical officer and director of the Office of Product Evaluation and Quality in the agency’s Center for Devices and Radiological Health, said in an FDA press release. “Following our rigorous internal review and input from the public, today’s actions will increase the safe use of surgical staplers and staples as an important alternative to manual suturing.”
In addition to the final rule reclassifying surgical staplers, the FDA also issued a final guidance on labeling recommendations to better convey specific risks, instructions and limits to doctors using surgical staplers.
The FDA also sent out a letter to healthcare providers on October 7, giving healthcare professionals additional recommendations on the safe use of surgical staplers for internal use.
Some of the recommendations include calling for healthcare professionals to carefully read the stapler manufacturers’ instructions, to consider selecting a different staple size if they have difficulty squeezing the stapler handle, to avoid using staplers on tissue too thick or thin for the selected staple size, to be aware of the different color schemes used by different manufacturers to represent staple size, and to have methods of blood vessel control in case of stapler failure, among many others.
The most commonly reported surgical stapler problems involve an opening of the staple line, malformation of staples, staplers misfiring, staplers being too difficult to fire, staplers failing to fire and staplers that were misapplied. The FDA warns that stapler malfunctions or misuse can cause prolonged surgical procedures, the need for additional surgeries, and other complications; such as bleeding, sepsis, internal organ damage, and death.
Medical device manufacturers face a growing number of surgical stapler lawsuits filed by individuals nationwide who suffered serious injuries or who lost loved ones due to stapler malfunctions.
Get more articles like this sent directly to your inbox.
"*" indicates required fields





0 Comments