Recall for Da Vinci Robot Issued Due Over Problems With Arms ‘Stalling’
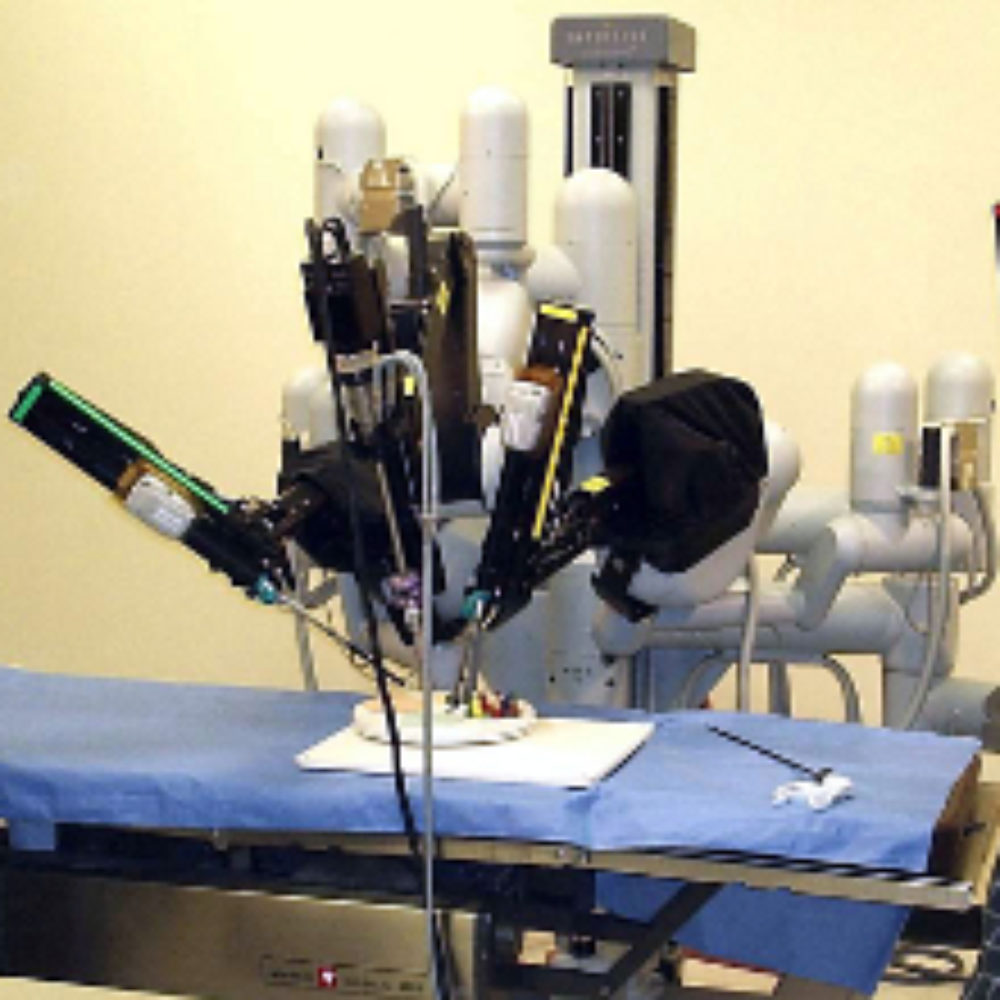
Intuitive Surgical indicates that the mechanical arms on some of it’s da Vinci robots could stall during robotic surgery, resulting in a recall that impacts about 1,400 components.
The da Vinci robot recall was announced on Tuesday by the FDA, after the manufacturer issued a “voluntary product correction” for da Vinci Patient Side Manipulators (PSMs).
Intuitive Surgical warned hospitals and other medical providers that the da Vinci robot instrument arms need to be replaced. The FDA has categorized the action as a Class II medical device recall, which suggests that the agency believes the stalled arms would typically result in temporary or medically reversible health problems, but could pose a risk of serious injury in rare cases.

Did You Know?
Millions of Philips CPAP Machines Recalled
Philips DreamStation, CPAP and BiPAP machines sold in recent years may pose a risk of cancer, lung damage and other injuries.
Learn MoreAccording to at least three complaints, friction within the PSM instrument arms may interrupt the da Vinci robot arm movement, resulting in them stalling or getting stuck. In at least one case, the problem resulted in an imprecise cut during robotic surgery, but none of the incidents led to da Vinci surgical complications, Intuitive Surgical claims.
“This can be felt by the surgeon as resistance in the movement of the master,” the FDA reports. “In this situation, the instrument can stall momentarily and then suddenly catch-up to the master position if the surgeon pushes through the resistance.”
The recall affects 1,386 Patient Side Manipulators on da Vinci S, Si and Si-e Surgical Systems.
Intuitive Surgical says it is addressing the problem by inspecting affected robots and repairing or replacing the arms if necessary. The company said in its November statement that nearly 70% of the inspections had been completed and the large majority of the arms did not need adjustment. However the company does not indicate how many robot arms were affected by the problem.
Da Vinci Robot Concerns
The da Vinci Surgical System is a remote control robot featuring four arms that are controlled by a surgeon through the use of hand and foot controls while sitting at a console that provides a virtual reality representation of the patient’s internal organs. The surgical robot has been increasingly used at hospitals throughout the U.S. in recent years for a number of different urologic, laparoscopic, gynecologic and non-cardiovascular thoracoscopic surgical procedures and thoracoscopically assisted cardiotomy procedures.
The action is just the latest in a series of da Vinci robot problems for Intuitive Surgical in recent months.
The surgical robots have come under increasing scrutiny amid concerns about the adequacy of training provided for surgeons and reports of patients suffering burns, tears and other internal injuries following robotic surgery. This is at least the third known recall involving da Vinci robots issued this year.
In May, Intuitive Surgical issued a warning to health care providers about problems with the da Vinci robot cauterizing scissors, which are known as “Hot Shears”, indicating that the components may develop micro-cracks that allow electrosurgical energy to “leak” to nearby tissue and cause thermal injury or burns to internal organs.
In July, it was reported that a Class 2 da Vinci robot recall was issued after it was discovered that about 30 of the surgical systems were not properly tested before being distributed. Although all of the robots were subsequently tested in the field and no problems were discovered, concerns emerged about how the factory testing was not in compliance with UL standards.
The FDA issued a warning letter to Intuitive Surgical, complaining that the company was not adequately reporting da Vinci robot problems. That concern has been echoed by some investment analysts, including Citron Research, which has warned investors that Intuitive Surgical stock price is in danger of tumbling once the full story of da Vinci robot problems come to light.
In October, Citron Research issued an updated report that criticized Intuitive Surgical over how it has handled da Vinci surgery concerns, warning investors away from the company’s stock. Citron identified more than 2,332 adverse events reported to the FDA’s MAUDE (Manufacturer and User Facility Device Experience) database over the first eight months of 2013. This compares with 4,603 reports received for the 12 year period between 2000 and 2012.
Reports include cases of bowel perforations, serious injuries and deaths, with many of the cases appearing to be years old and only surfacing now because of the ongoing product liability lawsuits filed against Intuitive Surgical. Citron indicated that all of the complaints appear to have addendums from Intuitive Surgical that suggest the manufacturer is attempting to dodge any and all responsibility.
Intuitive Surgical also faces a growing number of da Vinci robot lawsuits brought by patients who suffered complications that were allegedly caused by the robot.
Get more articles like this sent directly to your inbox.
"*" indicates required fields
1 Comments




AUDREYApril 13, 2022 at 3:49 pm
I had surgery and this machine was used. I was cut before the surgery started. Causing more issues. Life is not the same.