Eligible for a Gardasil lawsuit?
Gardasil Lawsuit Filed Over Autoimmune Disorder Side Effects
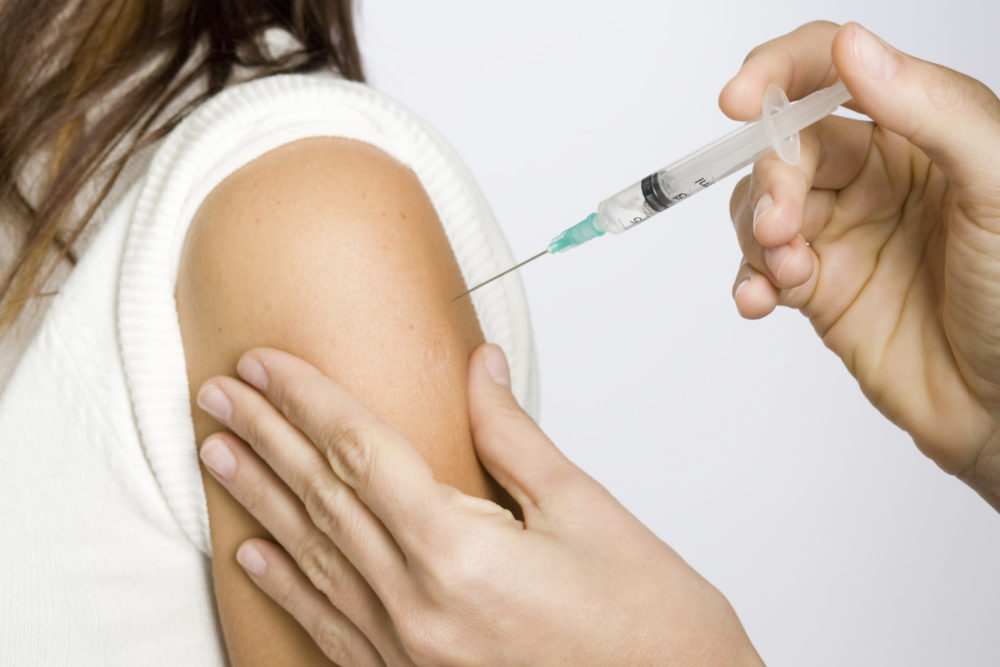
A Rhode Island woman has filed a lawsuit over Gardasil, alleging Merck & Co. falsely promoted the efficacy and safety of the human papillomavirus vaccine, indicating the medication caused her to suffer “serious and debilitating autoimmune injuries.”
The complaint (PDF) was filed by Jula Balasco in Rhode Island federal court on August 19, presenting claims for negligence, failure to warn, manufacturing defect, breach of warranty, common law fraud and violation of the Rhode Island Deceptive Trade Practices Act.
Gardasil is a widely used vaccine for the prevention of certain types of sexually transmitted strains of HPV, which can cause cervical cancer. It was approved by the FDA in 2006, and is given to young girls before adolescence and potential sexual activity.
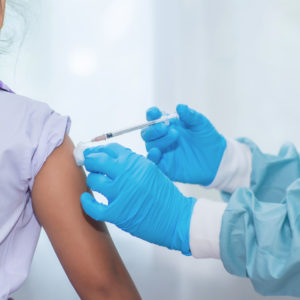
Gardasil HPV Vaccine Lawsuit
Side effects of the Gardasil HPV vaccine have been linked to reports of serious and debilitating autoimmune injuries. Lawyers review cases nationwide.
Learn More About this Lawsuit See If You Qualify For CompensationAccording to the lawsuit, Balasco received her first Gardasil injection in August 2014, when she was 13 years old. She then received a second shot in October 2014. However, she indicates the side effects of Gardasil caused her to immediately begin experiencing flu-like symptoms, resulting in aches, headaches, nausea, fever, and then ear pressure, tinnitus, headaches, dizziness, fatigue, migraines and vertigo.
Over time, her symptoms worsened, resulting in loss of balance, gastrointestinal problems, hives and numerous other ailments. In November 2016 she was diagnosed with a number of autoimmune conditions, including fibromyalgia, orthostatic intolerance (OI), and postural orthostatic tachycardia syndrome (POTS).
The Gardasil lawsuit notes that similar ailments have been linked to side effects of the HPV vaccine in medical literature, but warnings were withheld from consumers and the medical community.
Gardasil POTS Side Effects
Since it’s introduction, concerns of the Gardasil vaccine causing POTS and other autoimmune disorders have emerged after one of the lead researchers responsible for developing the HPV vaccine, Dr. Diane Harper, indicated that the drug’s protection may only last a few years, suggesting that the risks may outweigh the benefits for young girls.
Dr. Harper reportedly said at a conference in 2009, that while Gardasil was tested on 15 year old girls, it is commonly being given to girls as young as nine years old. She has called for more detailed warnings to parents about the Gardasil risks and to provide additional information about the unknown long-term benefits for girls who are not likely to be sexually active for several years.
Studies have shown Gardasil contains a number of ingredients which are known to sometimes cause problems with the autoimmune system, including Amorphous Aluminum Hydroxyphosphate Sulfate (AAHS) and HPV LI-DNA fragments.
Critics claim Merck failed to adequately disclose the presence of these ingredients, and also claim Merck used them in the placebos given to “control” test subjects during clinical trials, leading to an inaccurate assessment of Gardasil side effects.
As a result, teens and young adults throughout the United States are now pursuing Gardasil POTS lawsuits against Merck, indicating that they were left with autoimmune disorders and severe pain and injuries after receiving the HPV vaccine injections, including potentially life-threatening autoimmune disorders and cancers.
See If You Qualify for Gardasil Vaccine Compensation
13 Comments





MichelleAugust 20, 2022 at 12:24 am
I have a thyroid condition. My thyroid glands don't produce the hormones needed by my body to function normally so now I take medication daily that I'm on for the rest of my life . I'm also at an extremely low possibility of getting pregnant as a result. I love kids ..
DeeOctober 9, 2021 at 5:20 pm
Sick since 2009 after this in 2008 was scared into getting it and health declined since. Sjogrens also and Hashimotos mast cell etc etc horrible arthritis. Must find lawyer. Sick people. Poison vaccines please don’t give this to your babies. AND I WAS 25 way too late should sue the doctors. Also.
ReginaAugust 16, 2021 at 1:40 pm
I received this vaccine about 14 years ago (25 years old) and developed Celiacs Disease a year later and now have just been diagnosed with Stage IV Endometriosis. I’ve had cysts and pain for years prior to my diagnosis of Endometriosis.
DawnMarch 30, 2021 at 2:27 am
My daughter had such a severe reaction to the third shot of the Gardasil series that we had to go to the Emergency Room. She had it in 2009 when she was 15. She is still dealing with chronic debilitating problems.
JosephineMarch 23, 2021 at 3:54 pm
I received this when I was 19 because after multiple times my dr ask me I finally said yes, I am now 27 and I’ve been having arm pains for the last 9 years
SarahMarch 1, 2021 at 3:52 am
My daughter received the first vaccine against my better judgment. Her arm became swollen and red with pain which expanded the size of her entire upper arm. The next day she was in pain and later that next day became unable to move her neck or upper body freely for 3 days. Her PCP office reported this to the CDC and we have not opted to give her any other dose obviously.
LindaFebruary 16, 2021 at 3:55 am
I got my teenage daughter vaccinated at 16 or 17 abd she started having health problems 1 year later...Hashimotos/ fibromyalgia/ gastro problems/polycystic ovarian I need a good lawyer in Arkansas....
jolynFebruary 11, 2021 at 11:23 pm
I was given the Gardisil in 2008. I had cervical cancer while pregnant in 2006 and the Dr, pushed for me to have it. I'm in my 30s now and have been suffering from many issues for the last 3-4 years of consistent inability to do daily activities. I suffer from Hashimotos thyroid disorder, Shogren's Disease, RA that is so severe getting out of bed is a huge challenge, and also have had Chronic kidn[Show More]I was given the Gardisil in 2008. I had cervical cancer while pregnant in 2006 and the Dr, pushed for me to have it. I'm in my 30s now and have been suffering from many issues for the last 3-4 years of consistent inability to do daily activities. I suffer from Hashimotos thyroid disorder, Shogren's Disease, RA that is so severe getting out of bed is a huge challenge, and also have had Chronic kidney Disease for the last 10 years.
EmilyFebruary 9, 2021 at 7:25 pm
I received my shots when I was 10 years old. I’m now 18 years old my symptoms started a year later when I was 11 having super heavy periods. I went to the doctor and found out I have hashimoto’s thyroid disease along with a lot of other things including unexplained gastrointestinal problems. It is a constant fight & struggle every day. In 7th grade I missed 41+ days and in 8th grade I missed 56+ d[Show More]I received my shots when I was 10 years old. I’m now 18 years old my symptoms started a year later when I was 11 having super heavy periods. I went to the doctor and found out I have hashimoto’s thyroid disease along with a lot of other things including unexplained gastrointestinal problems. It is a constant fight & struggle every day. In 7th grade I missed 41+ days and in 8th grade I missed 56+ days due to my symptoms.
AutumnFebruary 2, 2021 at 3:50 pm
I received my shot shot 4 weeks ago and I’m still having pain in my left arm . On pain pills or working received a injection that’s not working and I’m in therapy now nothing working at all .
KarenJanuary 24, 2021 at 2:51 am
I received gardasil in my early thirties. At 46 I was diagnosed with UCTD, Fibromyalgia, UColitis. After teaching school for 28 years, I had to go out on disability.
MariaDecember 30, 2020 at 4:48 pm
I cannot believe this! My daughter had the vaccine and by 2013 developed severe Crohn’s disease. This resulted in anal fistula and several fistula surgeries and most recently a bowel resection. This has ruined her late high school. Years and college years. Unbelievable. I need to know where I can find a lawyer!!!
HarmoneeDecember 4, 2020 at 5:47 pm
Wow