Phillips Adaptive Servo-Ventilation Therapy Linked to Cardiovascular Death Risk
Phillips subsidiary Respironics, Inc. is warning that a device used for complex sleep apnea, the Adaptive Servo-Ventilation (ASV) Therapy system, may actually increase the risk of death among patients with chronic heart failure.
The company issued a ASV therapy warning on June 11, in response to a study conducted by ResMed, which found a statistically significant increase in cardiovascular mortality among sleep apnea patients with chronic heart failure who used ASV therapy. The findings have made Phillips start evaluating the safety of its own line of ASV therapy devices.
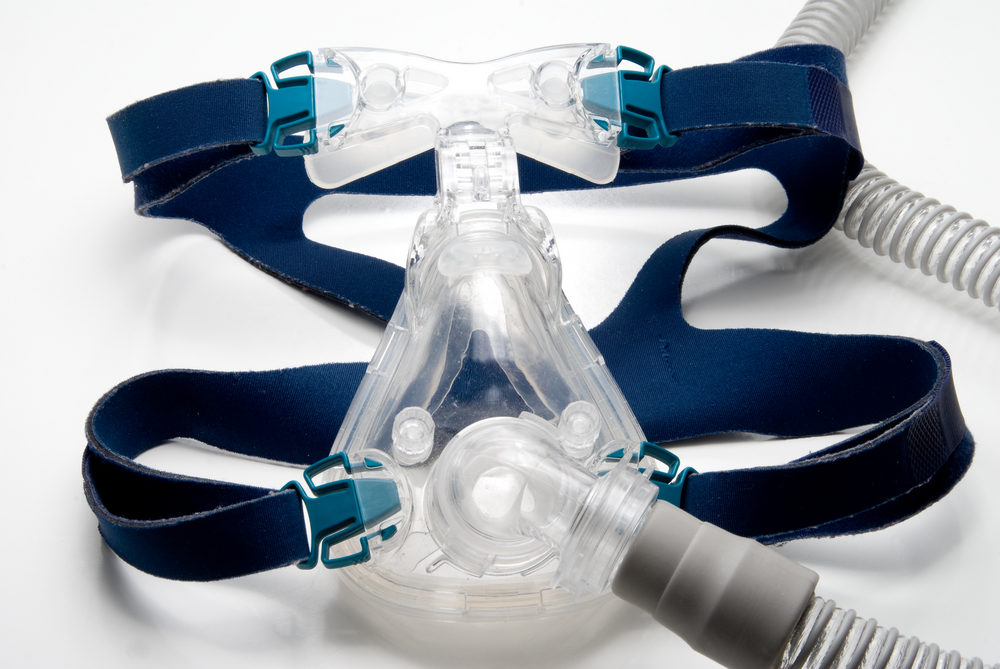
The ASV therapy systems are similar in many ways to continuous positive airway pressure (CPAP) therapy devices. It consists of a box linked to a face mask that regulates airflow and pressure in order to counteract the effects of sleep apnea and regulate breathing in sleepers. Unlike the CPAP, however, the ASV systems monitor the patient’s breathing and increases pressure when the user is having trouble breathing, and drops to a lower pressure that’s enough to maintain open airways when the patient is breathing fairly normally.

Did You Know?
Millions of Philips CPAP Machines Recalled
Philips DreamStation, CPAP and BiPAP machines sold in recent years may pose a risk of cancer, lung damage and other injuries.
Learn MoreIn May, ResMed released data on phase IV of a study called SERVE-HF, warning that the study did not meet its primary endpoints. The study was designed to see if ASV therapy systems could reduce mortality and morbidity in patients with both sleep apnea and chronic heart failure.
The company’s research found a statistically significant 2.5% increase in the annual risk of cardiovascular mortality in a randomized trial among ASV users when compared to a control group. The study found that 10% of the ASV group experienced cardiovascular death, compared to 7.5% of the control group. This represents an overall 33.5% increased relative risk of death from heart problems among the ASV users.
ResMed issued an Urgent Field Safety Notice (PDF) in response to its findings.
“Patient safety is our first and foremost priority. We have alerted and are working with appropriate global regulatory authorities about the safety signal observed in this study,” Dr. Glenn Richards, ResMed’s Chief Medical Officer, said in the May 13 press release. “The safety signal in SERVE-HF was observed only with the use of ASV therapy in people who have predominant central sleep apnea and symptomatic chronic heart failure with reduced ejection fraction. We are further analyzing the data to understand why this unexpected result was observed in this trial.”
Now, Phillips, which manufacturers the ASV therapy devices known as the BiPAP autoSV and BiPAP autoSV Advanced, says it is evaluating its own devices and ResMed’s data as a result.
“As part of this ongoing investigation, we are working with ResMed in order to better understand their study data,” Phillips wrote in its press release. “We are also evaluating post market surveillance data, public adverse event data and other published data to identify and assess other safety concerns that may be present.”
Phillips recommends that while its investigations continue, doctors follow current recommendations against using ASV therapy on patients with chronic heart failure and moderate to severe prominent central sleep apnea.
The company recommends that adverse reactions or quality problems experienced in the use of ASV therapy devices be reported to the FDA’s MedWatch Adverse Event Reporting program.
Get more articles like this sent directly to your inbox.
"*" indicates required fields
1 Comments




DavidDecember 20, 2019 at 11:39 am
I started using BiPap about 4 years ago and immediately started retaining fluid. Went to several specialists including Mayo in Rochester. My case is well documented there. I continue having difficulty with fluid retention for which I take prescribed medicine. Doctors assure me that it is not caused by BiPap but I am certain that it is taking a toll. Please help.