Child Drug Overdoses Rise as Other Accidental Deaths Fall: CDC

Federal health experts indicate that accidental poisonings and drug overdoses among children are on the rise, even as other causes of accidental injury and death among minors are declining.
The drug overdose and poisoning deaths echo a trend that is also seen among adults, with a rising number of prescription drug overdose deaths, which have surpassed fatalities caused by illegal drugs like cocaine and methamphetamines in recent years.
In the latest issue of the U.S. Centers for Disease Control and Prevention (CDC)’s Morbidity and Mortality Weekly Report (MMWR), officials note that the drug overdose and accidental poisoning trend among children stands out among nearly across-the-board declines in other areas.
The poisoning death rate among 15 to 19 year olds skyrocketed 91% from 2000 to 2009, increasing from 1.7 deaths per 100,000 to 3.3 deaths. The number of those deaths that can be attributed to prescription drug overdoses rose from 30% in 2000 to 57% in 2009, suggesting that at the turn of the century less, than one-in-three teen poisoning deaths were caused by prescription drugs, but they now account for more than half.
In late January, the CDC released a report warning that prescription drug overdoses nationwide have reached epidemic proportions among adults in general. In that report, the CDC indicated that there were about 27,000 unintentional prescription drug overdose deaths in the United States in 2007, labeling the phenomenon as a “U.S. epidemic.”
Nine million people currently report long-term use of OxyContin and similar painkillers in the U.S. and another 5 million use the drugs for nonmedical purposes; typically abuse.
The CDC suggests that it will take a society-wide effort to stem the growing problem that will have to include collaboration of federal state and local agencies, parents, health care professionals, youth influencers and policy makers.
Last year, the FDA initiated a painkiller abuse reduction plan, asking drug manufacturers to develop a Risk Evaluation and Mitigation Strategy (REMS) for all long-action opioids that would help keep them out of the hands of abusers. The FDA Opioid Strategy will require educating doctors on proper pain management, patient selection and will educate patients on proper use, dangers and disposal of the drugs.
A dozen extended-release, long-acting brand-name opioids and four types of generic opioids were identified by the FDA for the development of a REMS program. FDA officials say they hope the drug manufacturers will work together to develop a single REMS system.
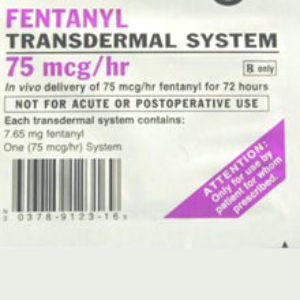
The brand-name drugs affected by the strategy include Duragesic, Palladone, Dolophine, Avinza, Kadian, MS Contin, Oramorph, Embeda, OxyContin, Opana ER, Exalgo and Butrans. The generic drugs affected by the strategy include fentanyl, methadone, morphine, and oxycodone.
Get more articles like this sent directly to your inbox.
"*" indicates required fields




0 Comments