ISMP Issues Analysis, Recommendations On Avoiding COVID-19 Vaccine Administration Errors
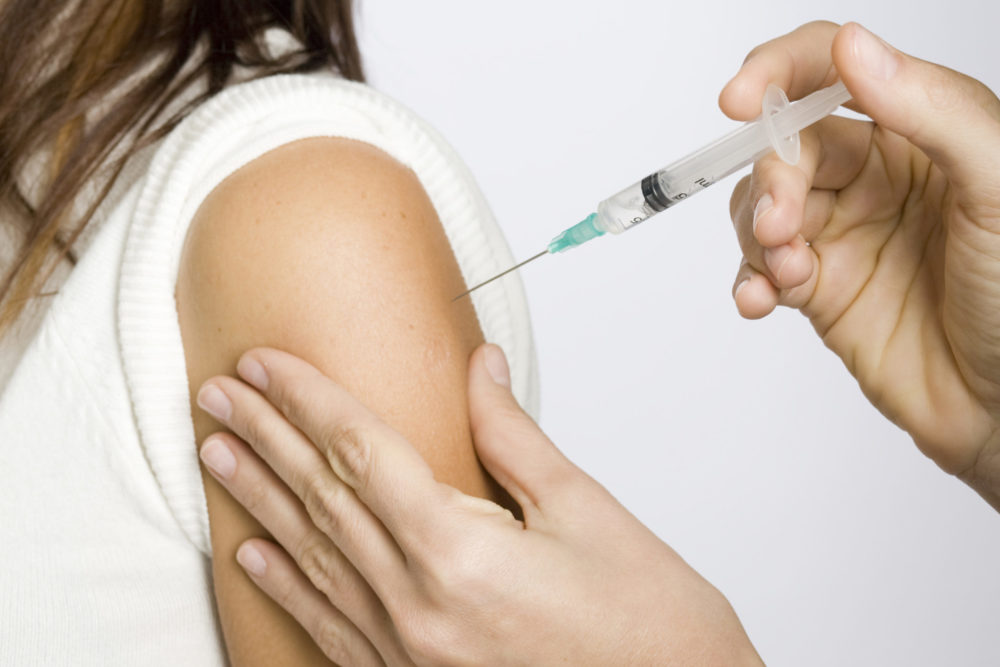
A medication safety group says while errors administering the various COVID-19 vaccines are rare, the medication mistakes being made by doctors and other healthcare professionals vary greatly, but indicate some safety precautions could eliminate many of them.
The Institute for Safe Medication Practices (ISMP) released a report on April 22, analyzing safety errors linked to the Pfizer-BioNTech, Moderna and Johnson & Johnson COVID-19 vaccines. It is their second review of the vaccine distribution. The first was in January, when vaccines began, and the second comes four months into the process, which has seen Americans receive 231 million doses, with about 96 million Americans fully vaccinated.
According to the ISMP review, the largest number of general errors made with all types of vaccine include patients receiving a lower dose than authorized, and the vaccines being given to patients younger than the recommended ages.

Did You Know?
Millions of Philips CPAP Machines Recalled
Philips DreamStation, CPAP and BiPAP machines sold in recent years may pose a risk of cancer, lung damage and other injuries.
Learn MoreMany of the cases where patients received a lower dose than authorized were due to patients pulling away from the needle before the health care professional injecting them were finished. However, the report notes that at one mass vaccination clinic, about 4,300 patients received a 0.2 mL dose of the Pfizer vaccine, instead of the 0.3 mL dose they were supposed to receive. This occurred because the syringes from the national stockpile left about a third of the vaccine stuck in the bottom of the plastic syringe.
The age-related problems occur generally when the vaccine providers fail to ask age-related questions.
There are also a number of incidents which are specific to the two-dose vaccines by Pfizer and Moderna. In some cases, patients received the wrong vaccine for the second dose, and in other cases, they were given the doses during the wrong time intervals. Patients receiving the Pfizer vaccine have to wait at least 17 days between the two doses, and those receiving the Moderna vaccine have to wait at least 24 days. In some cases vaccines are given too soon, too late, or they received a third dose by accident.
In many cases, these mistakes occur during the interview of patients before they receive their vaccines, ISMP found.
The ISMP issued the following recommendations to help avoid the most common COVID-19 vaccine administration errors:
- Educate and orient all vaccination staff, including volunteers, on their role in vaccination check-ins, patient screening, preparation, administration and common errors that occur.
- Verify the competency of all vaccinators
- Verify competency of staff who screen and check-in patients.
- Schedule patients for a second dose, if needed, before they leave the vaccination site after receiving the first dose.
- Establish a scheduling system that does not allow patients younger than 16 to obtain an appointment.
- Ask patients age-related screening questions, including date of birth, and comply strictly with vaccine age restrictions.
- Check state and local databases for documentation of COVID-19 vaccinations for those requesting a first or second dose.
- Require all patients who arrive for a second dose to present their vaccine card and have them carry the card throughout the process.
The ISMP also recommends pharmacies verify the number of vaccines needed each day and dispense prefilled, labeled syringes, separate the preparation of each brand of vaccine, and withdraw doses from one vaccine vial at a time before accessing the next vial, and label vaccine syringes immediately after preparation.
The report also recommends that, during administration, each vaccine station be stocked with a sharps container for syringe and needle disposal, each syringe be checked for the proper dose, air bubbles, and a tight fit between the needle hub and syringe, and check patients’ vaccine cards to verify correct time interval and manufacturer. In addition, those administering the vaccines should immediately engage the needle safety device and dispose of the syringe in a sharps container, report any syringe or needle malfunctions to the pharmacy, and be sure to complete the patient’s vaccine card.
The ISPM also urges those administering the vaccine to have an independent double check of the Pfizer vaccine dilution process and stock vaccination sites with epinephrine autoinjectors to visually differentiate them from vaccine syringes.
“Millions of doses of the COVID-19 vaccines will be administered over the next few months,” the ISMP report states. “As we work toward expanding vaccinations to all, we must also learn from the vaccine errors that have already happened and implement strategies to minimize the risk of making these same vaccine errors.”
Get more articles like this sent directly to your inbox.
"*" indicates required fields





0 Comments