Eligible for a Tepezza lawsuit?
Hearing Problems from Tepezza Impacted 16.5% of Participants in New Study
A significant portion of users reported hearing loss, tinnitus, autophony and other hearing problems from Tepezza use.
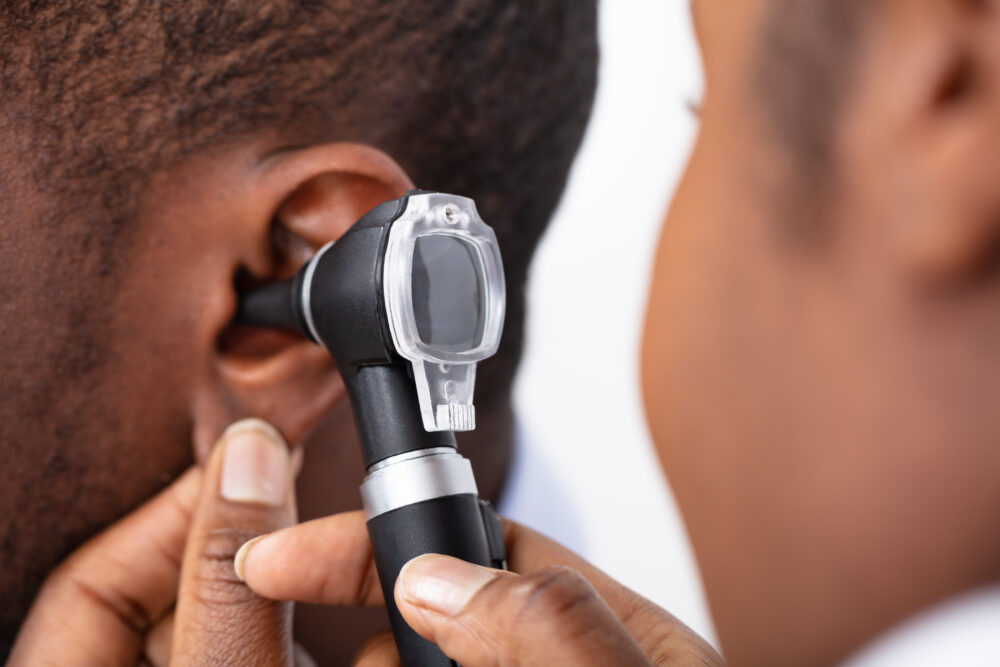
As a growing number of Tepezza hearing loss lawsuits continue to be filed against Horizon Therapeutics, a new study released by the drug maker reports that more than 16% of participants experienced hearing problems after receiving the thyroid eye disease drug.
Researchers from the University of Michigan and Horizon Therapeutics recently published findings from a small study in the medical journal Endocrine Practice, indicating that most patients reported satisfaction with the drug’s treatment of thyroid eye disease symptoms, but acknowledged that many also complained of hearing problems from Tepezza, including hearing loss, tinnitus, autophony and eustachian tube dysfunction.
Tepezza (teprotumumab-trbw) is a new-generation treatment that was just introduced in January 2020, as the first medication for bulging eyes and double vision that results from thyroid eye disease. These conditions are caused by hyperthyroidism and linked to Graves’ disease, resulting in inflammation of eye muscles, eyelids, tear glands and fatty tissues behind the eye.
Many patients require multiple Tepezza infusions to treat the condition, and combined with aggressive marketing and a high cost for treatment, Horizon was able to quickly push the drug to blockbuster status, generating sales that exceed $1 billion per year. However, concerns have quickly emerged that the drug maker failed to adequately research the potential hearing side effects from Tepezza, and withheld important safety information for consumers and the medical community.
Horizon Therapeutics now faces product liability lawsuits brought by former users, indicating that they have been left with irreversible hearing problems problems from Tepezza, claiming that they could have avoided permanent damage to their ears if the drug maker had warned about the importance of monitoring hearing before and during treatments.
Tepezza Lawsuits
Side effects of Tepezza may cause permanent hearing problems. Lawsuits are being pursued over the drug maker's failure to warn about the risk. Find out if qualify for a settlement.
Learn More About this Lawsuit SEE IF YOU QUALIFY FOR COMPENSATIONIn this new study, researchers looked at data from three clinical trials involving patients who reported hearing-related adverse events after conducting a 24-week study involving the use of Tepezza.
Out of 121 patients, 20, or 16.5% reported suffering adverse events linked to hearing on Tepezza. Of those, 12 patients, 9.9%, reported events that affected the quality of their hearing, including tinnitus, hearing loss or impairment, hyper- or hypoacusis, autophony and eustachian tube dysfunction.
Despite the problems, the drug maker and researchers indicate that patients who suffered hearing symptoms from Tepezza reported that their overall quality of life, as it related to their eye problems, improved.
While the findings are being promoted as a positive result by the drug maker, the extent of hearing problems reported in the small study raise further questions about why Horizon didn’t research the risk of hearing loss on Tepezza or warn consumers and the medical community about the potential for long-term damage that some users may experience.
Tepezza Hearing Loss Litigation
Lawsuits claim that Tepezza causes hearing loss by blocking a protein called IGF-1R, which can harm hair cells responsible for hearing and balance. Hair cells in the inner ear play a crucial role in detecting sound waves and transmitting them to the brain, and damaged hair cells can lead to hearing loss and other auditory issues.
Although Horizon Therapeutics knew or should have known that this mechanism of action may lead to hearing loss problems among users of the thyroid eye disease drug, the lawsuits claim that the drug maker failed to conduct proper follow up or update the Tepezza drug label warning to disclose the potential side effects.
Tepezza was submitted to FDA for approval using less than one hundred patients enrolled in clinical trials, according to the complaints, which indicate that Horizon Therapeutics failed to conduct sufficient tests to properly understand the mechanism of action for their drug.
Although Tepezza infusions were originally intended as a niche treatment, with only a limited market, during the second year the drug was on the market in the United States sales doubled to $1.66 billion, and critics have expressed concern that Tepezza was aggressively marketed without adequately disclosing all of the potential risks.
July 2023 Tepezza Lawsuits Update
Hundreds of former users are now coming forward to report that they experienced various forms of hearing loss from Tepezza, including persistent ringing in the ears known as tinnitus, and complete hearing loss. In many cases, the problems persist long after the thyroid eye disease treatments, leaving users with irreversible hearing damage.
Given common questions of fact and law raised in complaints filed in federal courts nationwide, a Tepezza MDL (multidistrict litigation) was established last month, centralizing the lawsuits before U.S. District Judge Thomas Durkin in the Northern District of Illinois for coordinated discovery and pretrial proceedings.
As part of the litigation, it is expected that a “bellwether” program will be established, where a small group of representative cases will be selected to go through case-specific discovery in preparation for early trial dates, which will help the parties gauge how juries are likely to respond to certain evidence and testimony that will be repeated throughout various cases in the litigation.
Following coordinated discovery and trials in the MDL, if the parties fail to reach a Tepezza settlement agreement or other resolution for the litigation, each individual claim may later be remanded back to the U.S. District Court where it was originally filed for trial.
Want a weekly update on top lawsuits, recalls & warnings?
"*" indicates required fields





0 Comments