CDC Warns Of Errors In Giving Shingrix Shingles Vaccines
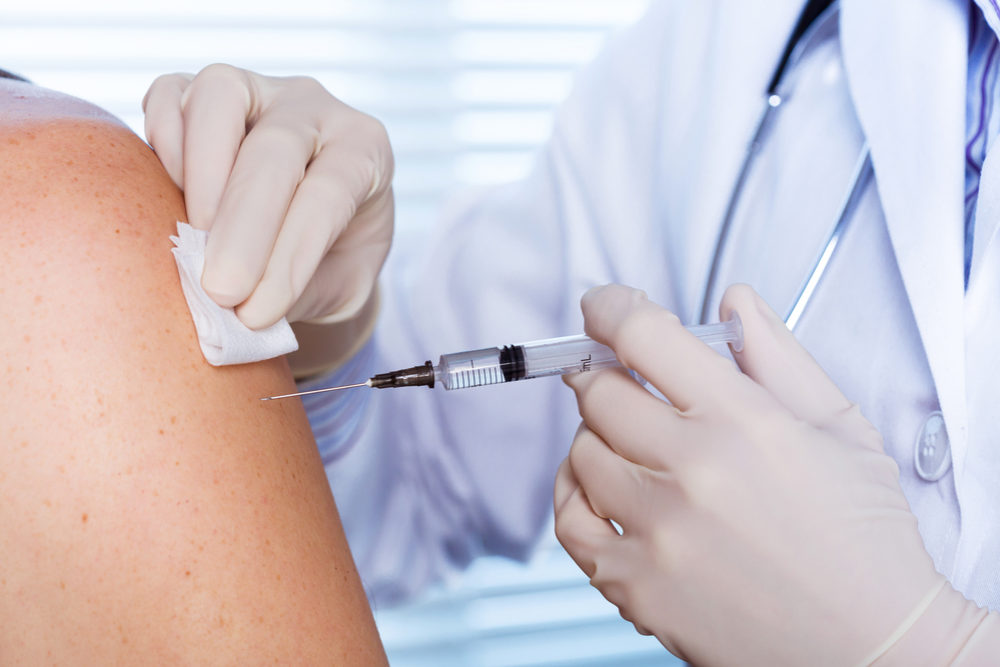
Federal health experts are warning about the risk of problems with Shingrix, GlaxoSmithKline’s shingles vaccine, indicating that errors have been reported in recent months due to confusion between the new drug and its older competitor, Zostavax.
The U.S. Centers for Disease Control and Prevention (CDC) published a report this week in its Morbidity and Mortality Weekly Report (MMWR), highlighting the number of adverse event reports submitted in recent months involving Shingrix administration errors.
Shingrix (recombinant zoster vaccine; RZV) is a new GlaxoSmithKline shingles vaccine that was just placed on the market in October 2017. It is only approved for adults age 50 and older, and is one two shingle vaccines that are currently approved in the United States. The other is Merck’s Zostavax (zoster vaccine live; ZVL).
Learn More About
Side effects of the shingles vaccine Zostavax may result in the development of a painful and persistent strain of shingles
Learn More About this Lawsuit SEE IF YOU QUALIFY FOR COMPENSATIONThe report indicates that 155 reports were submitted to the Vaccine Adverse Event Reporting System during the first four months Shingrix was available, including at least 13 problems with Shingrix administration. According to the findings, nine of those errors were because the drug was administered in a single subcutaneous dose, instead of as a 2-dose intramuscular injection, which is how the vaccine should be given. The single subcutaneous dose is how Zostavax is administered, suggesting confusion among doctors.
All but one of those patients suffered injection site reactions, including pain, erythema, and pruritus.
In two cases, patients were given the vaccine information statement for Zostavax instead of for Shingrix, and not instructed to return for their second dose, which also suggests whoever administered the vaccine thought they were giving the patients Zostavax, or thought that Shingrix worked the same way.
In the other four cases, the patients given the vaccine were younger than the minimum age of 50, for which the vaccine is approved.
The CDC researchers suggest that early monitoring indicates that the vaccine providers are confused over administration procedures, and storage requirements of Shingrix. Nine of the 13 vaccine administration errors occurred in a pharmacy, while two happened in a health care provider’s office, and two occurred at unknown sites.
“To prevent RZV administration errors, vaccine providers should be aware of prescribing information, storage requirements, preparation guidelines, and (Advisory Committee on Immunization Practices) recommendations for herpes zoster vaccines,” the CDC report recommends.
Shingles Vaccine Problems
The report comes amid concerns over the potential side effects of Zostavax, another shingles vaccine that was introduced in May 2006. Merck now faces a growing number of Zostavax lawsuits, which claim that the drug not only fails to protect against shingles as advertised, but actually causes a more persistent strain of the infection.
According to allegations raised in dozens of product liability lawsuits filed over the shingles vaccine, Merck used an under attenuated live strain of the varicella zoster virus (VZV) in Zostavax, which was not weakened enough to prevent reactivation of the virus. Instead of the body developing the proper immune response, the live virus combined with the old virus in some users, resulting in a more virulent strain of shingles.
Zostavax sales have fallen sharply in recent years, while Shingrix sales have far surpassed original projections, with full year sales now projected to exceed $600 million for the new drug.
Shingles is caused by the same virus, varicella zoster, which causes chickenpox. Later in life, the virus can be reactivated and cause shingles, which is a painful rash that usually appears as a stripe of blisters on the body. Pain can continue even after the rash is gone, which is known as postherpetic neuralgia.
Shingles itself can cause scarring, bacterial infections, encephalitis, hearing loss, vision problems and other complications.
Get more articles like this sent directly to your inbox.
"*" indicates required fields
129 Comments




HeatherJanuary 8, 2024 at 3:20 am
Seems we are being used as guinea pigs. I'll take my chances and not have this vaccine.
TuawanaNovember 26, 2023 at 7:17 pm
I received the Shingles vaccine a month ago, and I continue to suffer with severe pain in my shoulder and arm. This pain does not stop, and I have tried everything to try to help ease the pain.
GlendaAugust 27, 2023 at 3:07 pm
I got the shringix shot November 6,2022 and developed the frozen shoulder I had to take therapy but had to quite because it was making it worse probably will have to have surgery because I have some tears in my shoulder
ConstanceJuly 12, 2023 at 6:27 pm
I received a Shingrix vaccination (my second - minimal reaction to the first) in early January. I had redness and a slight fever for two days, then felt fine. Several days later I developed a red rash with itchy bumps on my head, welts on my trunk, and blisters on my legs and several on my arms. I had tachycardia and post-menopausal bleeding, which added to the mystery. I also had gastritis. I w[Show More]I received a Shingrix vaccination (my second - minimal reaction to the first) in early January. I had redness and a slight fever for two days, then felt fine. Several days later I developed a red rash with itchy bumps on my head, welts on my trunk, and blisters on my legs and several on my arms. I had tachycardia and post-menopausal bleeding, which added to the mystery. I also had gastritis. I was prescribed Benadryl at night and Claritin during the day by a dermatologist, and the rash disappeared. However, the tachycardia persisted for more than two weeks, and the digestive issues as well. It seems my entire body was affected. Apparently I had a very strong histamine reaction to the Shingrix. I have no way of knowing for sure, but I have never had a strong reaction to vaccinations in the past. I have been diagnosed with a Histamine Intolerance Disorder and have been on a low histamine diet for three months. My digestive difficulties have improved. Could the rash, etc. be attributed to the Shingrix?
KellyMay 6, 2023 at 2:45 pm
54-year-old male first Shingrix shot felt lousy for a few days. Got the 2nd shot 2 days ago low-grade fever, flu like symptoms. Still feeling little under the weather although I stayed up till 2am to watch King Charles Coronation. Might be just lack of sleep other than that no side effects like the other comments here. Didn't know about all these side effects people have had hopefully I don't[Show More]54-year-old male first Shingrix shot felt lousy for a few days. Got the 2nd shot 2 days ago low-grade fever, flu like symptoms. Still feeling little under the weather although I stayed up till 2am to watch King Charles Coronation. Might be just lack of sleep other than that no side effects like the other comments here. Didn't know about all these side effects people have had hopefully I don't have them.
MelanieMarch 26, 2023 at 11:27 pm
I received both shingrex vaccines one month apart as directed. I keep getting outbreaks a few years later. I usually get one big blister but have the burning pain so bad like I could have a huge break out. Blister on back but excruciating pain in upper arm underarm and breast. I'm at wits end with this.
ReginaAugust 6, 2022 at 11:41 am
I took the Shingrix vaccine in November, 2021. Exactly one week later I started having problems. On Wednesday everything was fine. Woke up on Thursday morning and I could not walk. For a fairly healthy person, I walked on a cane for 3 weeks. Did not drive for 2 months. I had no feeling in my legs and feet. I can walk now but I have been diagnosed with neuropathy. I feel that the shot sta[Show More]I took the Shingrix vaccine in November, 2021. Exactly one week later I started having problems. On Wednesday everything was fine. Woke up on Thursday morning and I could not walk. For a fairly healthy person, I walked on a cane for 3 weeks. Did not drive for 2 months. I had no feeling in my legs and feet. I can walk now but I have been diagnosed with neuropathy. I feel that the shot started all my issues. I will not take the second dose. I’m just praying that I can get some relief soon.
VickieAugust 1, 2022 at 2:49 pm
I received my first shingrex vaccine at a Kroger pharmacy. I has Flu like symptoms and a sore arm for a few day. Within a month I developed severe muscle joint body pain, stiffness, couldn’t walk, or do any daily work or personal care. I also ran a fever. I was in such severe pain I wouldn’t eat and lost 30 pounds in two weeks. At the end of this horrible first week I went to see my GP. He said[Show More]I received my first shingrex vaccine at a Kroger pharmacy. I has Flu like symptoms and a sore arm for a few day. Within a month I developed severe muscle joint body pain, stiffness, couldn’t walk, or do any daily work or personal care. I also ran a fever. I was in such severe pain I wouldn’t eat and lost 30 pounds in two weeks. At the end of this horrible first week I went to see my GP. He said I had developed a severe PMR. My seg rate was 110, but my c reactive protein was under 5. So he wasn’t worried about GCA He immediately place me on 40 mgm of prednisone and told me to never take the second Shingrix vaccine. My question : was my GP told by a pharma rep they knew the shingrix vaccine was causing first time PMR flare in some patient. It’s been over three years of decrease prednisone therapy and I’m still having deteriorating severe muscle, joint , back,leg, hand, and pelvic nerve pain. I’m pissed this pharmaceutical corporation has hiding these side effects from the FDA and public. I am in my 60’s and as in good health. I will never be the same. I have developed a life long disease complication with no cure, just palliative medical care that’s not working into my future. I want to know how many people have developed or reported PMR or Epstein Barr syndromes after their initial shringrex vaccination.
GaryJuly 9, 2022 at 10:04 pm
got three shingles vax, two different kinds total, due to cvs bad record keeping and them being understaffed and in a hurry. now have guillain-barre. hans are nearly useless. i’d sue if I could but big pharma are protected by our corrupt politicians. beware of cvs and their careless business practices. i have proof of getting three shots.
DavidJune 16, 2022 at 3:48 pm
Got the second shingrix vaccine March 2022. Ended up in the hospital in April for 18 days. Encephalitis caused by shingles virus.
HerbertMay 6, 2022 at 2:49 pm
I am 77 and wife is 64. We have both taken the Shingrix vaccines, but we continue to get Shingrix attacks 2-3 times a year. What can we do?
TheronApril 25, 2022 at 11:46 pm
I received the shingrix shot and within a week I was broke out on my side all around to the middle of my back. I have been to three different doctors for treatment of the shingles this has been a terrible experience I wouldn't want nobody to go through this pray for recovery please
PatriciaApril 8, 2022 at 3:24 pm
My bf mike had a severe reaction...complete nightmare.he got the shot on Dec 27th and my big strong guy was getting an x ray 3 days later because of severe back pain.by Jan 4th he couldn't even get out of bed and x rays n MRI were no help .he ended up with a nightmare ride to Er...they couldn't even move him without morphine and he screamed in so much pain...think back contractions with any moveme[Show More]My bf mike had a severe reaction...complete nightmare.he got the shot on Dec 27th and my big strong guy was getting an x ray 3 days later because of severe back pain.by Jan 4th he couldn't even get out of bed and x rays n MRI were no help .he ended up with a nightmare ride to Er...they couldn't even move him without morphine and he screamed in so much pain...think back contractions with any movement you make and stabbing knife like pain.they had to give him adavan and dolotin and put him on oxygen to take more MRI n cat scans.he ended up in the hospital 4 days .then rehab.still no relief.broght him home almost 1month later in the same pain and unable to barely stand to go from bed to wheel. Chair.now now 31/2 mo later he still can't walk but back spasms have stopped.he has lived a. Nightmares.now he is being told he may have myeloma blood cancer.
JoanneMarch 9, 2022 at 8:52 pm
I got a first Shingrix shot in November, no side effects. Three months later, even though I had a few small irritating sores on my back, I got the second. Now two weeks later, This "inflammation" is increasing in area and I'm wondering if I should have had the second shot when I did. Somehow I thought that the first Shingrix would provide some protection. Doctor I consulted thought it might be som[Show More]I got a first Shingrix shot in November, no side effects. Three months later, even though I had a few small irritating sores on my back, I got the second. Now two weeks later, This "inflammation" is increasing in area and I'm wondering if I should have had the second shot when I did. Somehow I thought that the first Shingrix would provide some protection. Doctor I consulted thought it might be something fungal but treatment recommended isn't working. Now for a second opinion ...
SteveMarch 7, 2022 at 7:26 pm
I'm a registered nurse and I received the shingles vaccine shingrix and ended up in the hospital within a week with meningo encephalitis I do not see anybody being warned that the shingles shingrix vaccine clearly has some issues as all of my other tests were negative and the only thing that my doctors feel it could have been was the vaccine.
RobinMarch 3, 2022 at 3:25 pm
I am 59y/o. I had my First Shingles Shot ( Shingrex) Jan 3, 2022. I had NO Problems!! 5 Months Later, due to various health problems I had to wait, (it is recommended the 2nd Shot be given 2 to 6 months later). OMG, I had Both Shots at My Pharmacy but Different Pharmacist, that Shot Felt Like a Spike going into my arm. 3 Hours Later I had SEVERE PAIN in my Shoulder and Arm down to The Elbow. As[Show More]I am 59y/o. I had my First Shingles Shot ( Shingrex) Jan 3, 2022. I had NO Problems!! 5 Months Later, due to various health problems I had to wait, (it is recommended the 2nd Shot be given 2 to 6 months later). OMG, I had Both Shots at My Pharmacy but Different Pharmacist, that Shot Felt Like a Spike going into my arm. 3 Hours Later I had SEVERE PAIN in my Shoulder and Arm down to The Elbow. As The Hours Progressed I could barely raise my arm or pick up 1/2 Gal of Milk. I contacted The Pharmacist who stated, “ that was a Possible Side Effect and could last Several days Up To 2 Weeks.” It has NOW Been 3 Months and my Shoulder and Arm are NO BETTER!!!! Due to an Upcoming Total Hip Replacement Arthroplasty I am NOT able to Receive A Cortisone Shot or Take Oral Prednisone. I have Been Alternating Heat & Ice and Using Deep Heat Rubbing Down The Arm & Shoulder then Wrapping it with an Ace Bandage. Some days I think it’s getting Better then days like today it comes back with a Vengeance!! I have been unable to locate any Additional Home Remedies for it. So, will continue to Suffer with this Severe Chronic Shoulder and Arm Pain. If I had it to do All OVER AGAIN I Would NOT HAVE TAKEN THE SHINGLES VACCINE AND TAKE MY CHANCES!!!
FARONFebruary 3, 2022 at 6:02 pm
59 years old and 5 months ago i went to get the shingles shot & my life has never been the same ! My arm never stops hurting & i developed several trigger points on my back its been horable to be honest i wont be getting any more shots !
geoJanuary 17, 2022 at 1:26 pm
Husb (75) & wife (68) 1st shingrix vax Nov.10: husb: over-extreme sore arm, lasting days. 2nd vax Jan 13 following yr: he found out what the wife experienced with chills, nausea, dizziness (most on day # 3 and still unabated: taking broth only days 2 1/2-3, after being able to eat days 1-2 1/2. He is on calquence for leukemia but pcp paperwork showed he needed shingrix vax to prevent shingles (h[Show More]Husb (75) & wife (68) 1st shingrix vax Nov.10: husb: over-extreme sore arm, lasting days. 2nd vax Jan 13 following yr: he found out what the wife experienced with chills, nausea, dizziness (most on day # 3 and still unabated: taking broth only days 2 1/2-3, after being able to eat days 1-2 1/2. He is on calquence for leukemia but pcp paperwork showed he needed shingrix vax to prevent shingles (he had the zoster vax, many years prior). Wife: If you are reading these comments and you've had #1 vax and are considering #2, please do not ignore these comments. Response to #1 was: "How can this be on the market considering side-effects?" Called pcp and noted all-systems-reaction with the most painful being the "dots/ankles" which turned more into wheals (stayed exactly same-sites as time passed): pain caused from the "itch-feeling" tho n-e-v-e-r- scratched once; ice-cubes were the best pain-solution, lotion w/aloe aided some. Systemic-r,esponse not limited to any body part as felt vax immed.-entering my syst. w/ "gut-punch" reaction; dizzy, balance horribly effected; noted pain in calves but did not attribute to vax until read (never had such pain prior (& exercise is zumba-type, shovel a 3-car-driveway =active);left arm/wrist in which 1st vax administ'd became weak and wrist almost unusable. That was the 1st vax. Why did I go for the 2nd vax? I knew the nurses & online sites said it would, likely, be worse than the 1st. "Yes" I did go for #2 vax: told administrator-pharmacy site of "gut-punch" and rash and she didn't have any warnings and did not say "not to get #2; neither did the pcp-nurse say not to get #2." #2 Vax was not so much "gut-punch" but felt it underneath back of ribs on same side & told administ'r, "feel it underneath vax-site (lymph-syst) ; the spots/rash from #1 had cleared up, but recently returned: thus became "same as the first-round with intense itch-feeling & pain (not scratching once); unbalanced in body-movements noted as w/vax #1. Overall: vision changes noted w/ both #1 & #2: believe this is (continues) with a "like-reaction" to shingles, as at age 15 I remember the Dr. warning to "guard-eyes" as then had an intense shingles itch & swelling. Should I have taken #2 vax? Well, if I don't (ever) get shingles it may (may) be well worth it. The only other vax in recent years was the 3-vax-covid-series...to which I had an intense reaction to the #1 covid-vax; also had a pneumonia vax in past; otherwise, have avoided vaxes. Where does that leave us? Husb.: day #4 now...sleeping; taking broth only, feels "like shit." Wife: I was feeling better yest...until last nite when the "itch-feeling leading to nausea" hit: answer: toast w/nutmeg, cinnamon & ginger and aspirin. Noted that most of "feeling off-balance" is waning (didn't want to crash into anything which could, otherwise, be quickly skirted-around); was able to eat what I wished. I am hoping the lower-leg rash will abate-itching, but from the comments on this website, I don't know if I can hope all signs of this vax will be gone come summer grass mowing---I may consider some type of light-therapy. I still find myself reflecting on the ques: "Why would this be on the market with the known reactions." Again, if you are reading this, it appears as many persons with whom I have spoken who have had "no-reaction" to shingrix-vax...so there are as many who have had a very undesired, if not changing life for a long-period of time or permanent life-altering reaction..............So......If you are considering that #2 shingrix vax, consider just how strong the #1 vax was for you, how long the reacion(s) lasted and whether you are willing to face #2 with what is commonly cited by both online-comments and clinic-nurses as "worse than vax #1 reactions."
MichaelJanuary 11, 2022 at 12:14 am
I stand by my statement above and have my medical records to prove. As far as health; 2021 was the worse year of my life.
LoisJanuary 2, 2022 at 2:04 am
I had shingrix shot at the pharmacy spring of 2020. A few days later my back broke out in little crusty scabs, but no other reaction so I got the second shot 6 months later.... the scabs continued and since then have spread down my butt and back of thighs. Some of them are infected and full of pus. I'm a mess, and it is getting worse as time goes on. Dr has taken punch biopsies and we're waiting[Show More]I had shingrix shot at the pharmacy spring of 2020. A few days later my back broke out in little crusty scabs, but no other reaction so I got the second shot 6 months later.... the scabs continued and since then have spread down my butt and back of thighs. Some of them are infected and full of pus. I'm a mess, and it is getting worse as time goes on. Dr has taken punch biopsies and we're waiting for identification. I'm 74 and have never had any skin problems before this time, or any reaction from vaccines like covid. .This vaccine has not been properly tested, except on all of us. I'm disgusted and worried.
LindaDecember 1, 2021 at 3:31 pm
I am 51 yrs old and had the shingles vaccine, Shingrix on August 31 2021. I thought I was doing a good thing for my body and it has turned into a nightmare Within 4 daysI ended up in the ER. I couldnt put weight on my leg without having horrible pain in my right leg. Strong muscle spasms that feel like electric shock along with constant sharp pain. I'm using a walker. I tried to tell my D[Show More]I am 51 yrs old and had the shingles vaccine, Shingrix on August 31 2021. I thought I was doing a good thing for my body and it has turned into a nightmare Within 4 daysI ended up in the ER. I couldnt put weight on my leg without having horrible pain in my right leg. Strong muscle spasms that feel like electric shock along with constant sharp pain. I'm using a walker. I tried to tell my Dr's I believed it had to be the shot. Finally today a nurse practioner agreed. The condition is postherpetic neuralgia. It can take yrs to go away! I'm doing water therapy 3 times a week. It has improved my ability to move my leg better but pain isn't going away. I wish I would have never taken Shingrix 😔
CathyNovember 30, 2021 at 10:31 am
I had my shot on the 23rd of November. Perfectly healthy and feeling good. No cold, flu or sinus issues. On the 25th, I stood up from sitting down and felt something in my left ear, and then it began roaring. I couldn't hear anything but the roaring sound. Still can't. Went to the doc yesterday and then sent me to audiology to do a hearing test. Nothing in my left ear but they said my eardr[Show More]I had my shot on the 23rd of November. Perfectly healthy and feeling good. No cold, flu or sinus issues. On the 25th, I stood up from sitting down and felt something in my left ear, and then it began roaring. I couldn't hear anything but the roaring sound. Still can't. Went to the doc yesterday and then sent me to audiology to do a hearing test. Nothing in my left ear but they said my eardrum looked fine. Sent me to ENT who said this is Sudden Sensiserial hearing loss and my hearing may not come back. He said he has no idea what caused it but it is rare and the only possible treatment is with Steroids and that they don't really know how well that works. I can't take steroids because even a low dose shoots my bp up. I had taken a low dose of them from going to the urgent care the night before and even with double bp meds my bp was 190/92
SandyNovember 29, 2021 at 10:14 pm
I’m 68 and I have migraines my whole life I got the shingles shot about a week ago. I woke up at 3am with a migraine from hell I couldn’t get out of bed until 2 pm that afternoon I really don’t want to go through that again with the second shot.
SidneyNovember 16, 2021 at 3:06 pm
In response to K on page 1. Believe the unbelievable stories because they are all true. My symptoms immediately after getting Shingrix (my FIRST and only vaccine injury): squeezing chest pain, all over muscle pain, the arm I received the shot in has now lost the ability to grip, push, or bear weight, deep bone pain, massive hair loss, inflamed eyes, pain in gland under my neck, and all around sys[Show More]In response to K on page 1. Believe the unbelievable stories because they are all true. My symptoms immediately after getting Shingrix (my FIRST and only vaccine injury): squeezing chest pain, all over muscle pain, the arm I received the shot in has now lost the ability to grip, push, or bear weight, deep bone pain, massive hair loss, inflamed eyes, pain in gland under my neck, and all around system wide pain and chaos. I’m two months into this already so when does it end? Shingrix is dead virus? What are the symptoms of internal shingles? The very thing charged with protecting me from this type of event actually “caused” the event? This sounds like criminal negligence if you ask me because these injuries have been reported innumerable times.
MichaelNovember 4, 2021 at 1:27 pm
67 yr old male, got the 1st shot on April 6, 2021, on April 8th I had severe chest pain and did nothing but waited til the pain went away. Then on April 14 I developed a fever 105 degree and took ibuprofen and aspirin, the next day the fever was still present with dry heaving so I went to ER. DX w/ epstein-barr--mono and UTI w/ ecoli present. I was not around anyone prior to those events who w[Show More]67 yr old male, got the 1st shot on April 6, 2021, on April 8th I had severe chest pain and did nothing but waited til the pain went away. Then on April 14 I developed a fever 105 degree and took ibuprofen and aspirin, the next day the fever was still present with dry heaving so I went to ER. DX w/ epstein-barr--mono and UTI w/ ecoli present. I was not around anyone prior to those events who were sick. I went to my cardiologist and had a stress test that later on ended up with a heart cath and stent. On May 23rd I developed severe Left leg pain which awoke me from sleep and I could not even get out of bed. The next day I went to the ER because I was worried about a blood clot which turned out to be negative but I am still having difficulty with walking.
LarrySeptember 16, 2021 at 9:34 pm
I received my first Shingrix shot in Dec 2019. A few days later I developed severe arm pain and rash. My pharmacist recommended I not get the 2nd shot. Within a week, both of my legs developed a disgusting rash with scaly patches. Went to the Dermatologist and they biopsies my thigh, gave me 2 steroid shots, and put me on 3 months of doxycycline. Also put me on triamcinolone 1% and told me t[Show More]I received my first Shingrix shot in Dec 2019. A few days later I developed severe arm pain and rash. My pharmacist recommended I not get the 2nd shot. Within a week, both of my legs developed a disgusting rash with scaly patches. Went to the Dermatologist and they biopsies my thigh, gave me 2 steroid shots, and put me on 3 months of doxycycline. Also put me on triamcinolone 1% and told me to moisturize 10 to 12 times a day. Bottom line is that I suffered with this for 2 years! Also, I have some pretty disturbing photos of this whole s▪︎▪︎t show.
ScottAugust 18, 2021 at 12:52 am
I received the shingrix vaccine last October with in about a few days I started feeling like I was getting shingles like feeling in the arm the vaccine was administered I assumed it would go away but it only got worse I went back to the doctor and was told I might be coming down with Ms and should get a mri which I thought was not the case because the feeling was in the arm I got vaccinated by th[Show More]I received the shingrix vaccine last October with in about a few days I started feeling like I was getting shingles like feeling in the arm the vaccine was administered I assumed it would go away but it only got worse I went back to the doctor and was told I might be coming down with Ms and should get a mri which I thought was not the case because the feeling was in the arm I got vaccinated by the time I got the MRI the symptoms had gone through my intire body the MRI was normal I have lost most of my strength in my arms and legs my eyes and throat and chest have been severely affected to the point I went to the emergency room thinking I was going to die I have had to go on short-term disability on June 9th which will run out on September 8th and will have to go on long-term disability in the course of all of this I have had nerve tests lumbar puncher and have been prescribed several medications anti vitals steroids and many other treatments to no avail I believe that the shingrix vaccine is responsible for all this and feel that they should be accountable. Scott
ScottAugust 10, 2021 at 2:02 am
Has been many weeks and still severe deltoid pain at injection site on arm. Cannot sleep through the night, have trouble working, and can’t even watch TV without severe pain at times during the day and night.
KJuly 17, 2021 at 6:22 am
Shingrix is not a live vaccine, and therefore cannot give anyone shingles. It takes some time for protection to develop, and no vaccine is 100% - but if someone gets shingles after getting the vaccine, it didn’t cause it. The vaccine is overwhelmingly safe and effective. This comment section has an awful lot of unbelievable stories.
ToddJuly 12, 2021 at 6:57 pm
I had these shots over a year ago and since then I have had a rash all over my body. My face gets swollen. Life has been difficult. My doctor says this will most likely be something I have to live the rest of my life with.
CarolJune 22, 2021 at 11:02 pm
My first comment is 7 responses up. I continue with severe joint pain in my left knee and bilateral shoulders. I can barely get in and out of bed. Turning over and finding a comfortable position continues all night.I end up flat on my back. Putting on clothing over my head causes pain in shoulders. Had both X rayed yesterday and will find out what they find on Friday. I am taking Celebrex 200 mg [Show More]My first comment is 7 responses up. I continue with severe joint pain in my left knee and bilateral shoulders. I can barely get in and out of bed. Turning over and finding a comfortable position continues all night.I end up flat on my back. Putting on clothing over my head causes pain in shoulders. Had both X rayed yesterday and will find out what they find on Friday. I am taking Celebrex 200 mg at 9 am and again at 9 pm. I pray this hell will end soon. I am 80 and prior to this incident was a vibrant Senior. The human body does not just turn on it’s self. There was a catalyst and that was Shingrix!!
tanishaJune 18, 2021 at 1:16 am
Living with herpes virus was the worst situation I've ever been in due to work and tending to my family needs but finally tested negative after using the herbal medicine I ordered from him. his Email drodiagbesolutionhome gmail com
JamieMay 16, 2021 at 9:25 am
I just got the first dose of Sbingrix and I am 41. I was told to get the shot by my neurologist because my myelitis was caused by the shingles virus. 2 weeks later I now have shingles down my left arm, on my stomach, and I can feel them starting on my right arm. I don't want to get the second shot and worse....if I have one more neurological attack, it will be MS. What do I do?
ConnieApril 26, 2021 at 10:15 pm
Took my first shot in April 2019. Hade flu symptoms for days continued to not feel well. Took my second shot at the end of august. I did not feel well at all. Had heart palpitations. Went to the Doctor had a EKG that was questionable. I was waiting to go in for follow up with heart doctor. I was having difficulty breathing. My husband took me to the hospital. My lungs were filled with blood clots.[Show More]Took my first shot in April 2019. Hade flu symptoms for days continued to not feel well. Took my second shot at the end of august. I did not feel well at all. Had heart palpitations. Went to the Doctor had a EKG that was questionable. I was waiting to go in for follow up with heart doctor. I was having difficulty breathing. My husband took me to the hospital. My lungs were filled with blood clots. I was in the hospital for 5 days and will be on Eliquis for the remainder of my life. I would never recommend this shot to anyone. I was in really good health up to that point.
AndrewApril 22, 2021 at 2:30 am
63 years old, no major medical problems.First dose 2/1/21 was sick for about two days. Seemed like no big deal. Second dose Sunday 4/18/21 whole different story. Was incontinent for 24 hours, then had severe back pain but was too sick to try to get to the doctor, so I messaged her and she said don't worry it will go away in a week. Then I started having such bad pain from breathing that I couldn't[Show More]63 years old, no major medical problems.First dose 2/1/21 was sick for about two days. Seemed like no big deal. Second dose Sunday 4/18/21 whole different story. Was incontinent for 24 hours, then had severe back pain but was too sick to try to get to the doctor, so I messaged her and she said don't worry it will go away in a week. Then I started having such bad pain from breathing that I couldn't sleep so today (Wednesday) I went to the ER. Over 5 hours, and doc tells me a now have pleurisy. Coincidence? I think not! Just hope more problems don't develop. When you check the side effects on-line there is a whole different tone between government propped pages and independents.
MarshaApril 8, 2021 at 9:05 pm
Since 2018, I have contacted no less than 50 law firms due to the permanent side effects from the smithglaxokline shringrix vaccine. Not one law firm was interested in my case that left me with nerve damage in my hand. Also contacted the gov't agencies listed on the info sheet given to me at time of vaccination - All said I would be entitled to compensation - never happened! Disgraceful that this[Show More]Since 2018, I have contacted no less than 50 law firms due to the permanent side effects from the smithglaxokline shringrix vaccine. Not one law firm was interested in my case that left me with nerve damage in my hand. Also contacted the gov't agencies listed on the info sheet given to me at time of vaccination - All said I would be entitled to compensation - never happened! Disgraceful that this can happen to people in this country and no one takes responsibility!
JeanneApril 3, 2021 at 9:28 pm
I have had severe ongoing skin rashes since getting this horrible vaccine. I had a tiny patch of poison ivy (which I am very allergic to) when I got the shot. Within 24 hours, Ii was covered with it from head to toe. it sort of morphed into an ongoing autoimmune skin disease. My docs and I guess the vaccine jut put my immune system into overdrive to react to the poison ivy and it has never gone[Show More]I have had severe ongoing skin rashes since getting this horrible vaccine. I had a tiny patch of poison ivy (which I am very allergic to) when I got the shot. Within 24 hours, Ii was covered with it from head to toe. it sort of morphed into an ongoing autoimmune skin disease. My docs and I guess the vaccine jut put my immune system into overdrive to react to the poison ivy and it has never gone away. It felt like maggots crawling under my skin 24 hours a day. for months I was living on benedryl (which upsets your stomach at a high dose) and coffee for several months. I lost over 10% of my body weight due to that and the sheer misery of the rash. This was everywhere--on thle soles of my feet, on my eyelids. I coud not wear any clothes because anything that rubbed caused the rash and itching to start. I finally took a 3 week dose of steroids and it got a lot better. But I still have horrible itching all day long. This has set off a chain of related horrendous medical problems, including significant bone loss. I will never be the same and never stop scratching or coating my body with lotions. I now have to go on bone meds. I never had skin problems before--not once. Worse yet--the doctors are hawking this like ice cream. I told my doc time and time again that I never had chicken pox, but she insisted it would be no harm to get the vaccine. Well not really. My sister and father are doctors and they confirm I never had chicken pox. no one in our family did. I was never afraid of vaccines so I thought it would do no harm in case somehow I had a symptomless case as an adult--which is virtually impossible. Promotional material for this vaccine tell doctors to assume that anyone within a certain ag group had chickenpox. If they deny it, the literature says to assume they just forgot. Right--you forget you have chicken pox. I am now very anti-vaccine. this has ruined my life. I can barely move without getting the rash and crawling maggot sensation when any article of clothing rubs on my skin.
CarolMarch 16, 2021 at 5:31 pm
I received my first Shingrix vaccine October 2020. My arm was sore but tolerable. My second was on January 26, 2021. That evening I began feeling freezing cold, headache, super flu like symptoms. Right side chest discomfort. My joint aches were horrendous. This lasted 3 days. The headaches continued. I had clay colored stools for several days. I think my liver was whacked. This finally subsided, [Show More]I received my first Shingrix vaccine October 2020. My arm was sore but tolerable. My second was on January 26, 2021. That evening I began feeling freezing cold, headache, super flu like symptoms. Right side chest discomfort. My joint aches were horrendous. This lasted 3 days. The headaches continued. I had clay colored stools for several days. I think my liver was whacked. This finally subsided, but the headaches they continued. My neck hurts and Back aches. I feel terrible. Went to the Dr. and he sent me to Physical therapy, traction to my neck with massage, 2 weeks and no improvement. Had X rays of neck and showed abnormal cervical 6-7. Said that was the cause of headaches. Went for M R I and he said I would’ve need surgery but could probably benefit from a epidural. Each morning my joints hurt so bad I can hardly get out of bed. I wish I had never has the Shingrix vaccine. I am almost crippled from it.
SteffieMarch 14, 2021 at 4:18 pm
At 61 years old in excellent health, mowing half acre with a push mower for years and years. Have NEVER had a reaction from any inoculation. Working years in the medical/dental/legal fields and not witnessing the issues with the old shingles shot (which I took).I saw no reason not to take the new one with my younger husband who had not taken the old shot and had full blown shingles years ago. [Show More]At 61 years old in excellent health, mowing half acre with a push mower for years and years. Have NEVER had a reaction from any inoculation. Working years in the medical/dental/legal fields and not witnessing the issues with the old shingles shot (which I took).I saw no reason not to take the new one with my younger husband who had not taken the old shot and had full blown shingles years ago. Off we went to the pharmacy to receive the first shots. That evening husband developed a large rash, 10 inches by 6 inches on the inoculated arm. Flu like symptoms, headache, joint aches followed. Lasted 3 days for him. Sicker than I had ever seen him in 30 years of marriage. A few days after I receive the shot, feeling very sick, I headed to my lab folks and had over $350 worth of labs taken. I developed Epstein Barr Virus, had NEVER had Mono, but here I was very sick. Had to start antibiotics immediately due to the kidney, bladder and spleen infections. Also developed mouth and vaginal sores that were very painful. I am now living with this virus and NO I will never take the second shot. I can only hope and pray that I will be able to take the Moderna inoculation in just over 2 weeks! I will be researching EXACTLY what will be in the Moderna jab for sure!
JanMarch 13, 2021 at 12:24 pm
I had my first Shingrix shot Sept 2020. Only a sore hardness at injection site. I’m ready for the second dose today (March 2021) and hope I have the same reaction. I know one thing, it hurt more than the Covid vaccine shot.
RobertMarch 2, 2021 at 2:13 pm
June 1, 2018, I had my first Shingrix vaccine shot. Had a bit of a sore muscle for three days were I was given the shot. Normal for this vaccine, similar to the Tetanus shot, and to be expected. Two months later on Aug, 3, 2018, I had my second Shingrix shot. Again a bit of a sore muscle, but only for about two days. My wife got her Shingrix shots with me. We’re both fine. Her grandmother had Shin[Show More]June 1, 2018, I had my first Shingrix vaccine shot. Had a bit of a sore muscle for three days were I was given the shot. Normal for this vaccine, similar to the Tetanus shot, and to be expected. Two months later on Aug, 3, 2018, I had my second Shingrix shot. Again a bit of a sore muscle, but only for about two days. My wife got her Shingrix shots with me. We’re both fine. Her grandmother had Shingles really bad and suffered horrible pain for a long long time after afterwards. That convinced my wife to get the Shingrix shots. My brother just told me he’s got Shingles in his head, and close to one eye, and lots of pain. He told me he was never vaccinated against Shingles. Happy my wife and I got our Shingrix shots. I highly recommend the Shingrix Shingles vaccine shots.
EveFebruary 28, 2021 at 3:44 am
My experience was almost identical to Lisa’s above from November 2020. I am 57, in s 35+ year monogamous relationship, and had my first shot a week before Thanksgiving in November 2020. I similarly had fever, chills, body aches and malaise for 2 days, and extreme irritation on labia making it difficult to sit for over a week. By the time I was able to get an appointment with gynecologist, the symp[Show More]My experience was almost identical to Lisa’s above from November 2020. I am 57, in s 35+ year monogamous relationship, and had my first shot a week before Thanksgiving in November 2020. I similarly had fever, chills, body aches and malaise for 2 days, and extreme irritation on labia making it difficult to sit for over a week. By the time I was able to get an appointment with gynecologist, the symptoms had abated, so he assumed it was probably inconsequential. I just had the second shot and had fever plus aches/malaise for 10 hours and I am again experiencing irritation on labia. I have never had herpes or even a cold sore. When I had chicken pox I only had 4 or 5 spots. Perhaps my immune system has a strong reaction to this virus... I am glad I won’t have to have any more of thes shots.
MaryFebruary 20, 2021 at 4:12 pm
Nor am I an anti-vaxxer. I had my first dose on 22 Nov 2019 with the local pharmacist. It took me several weeks to connect the fact of my recent vaccine and the pain in my left upper arm and limited reach could be connected with the shingles vaccine. At the six-week mark, I decided to make an appointment with my PCP. Prior to the appointment, the pain went away and my reach returned. I went for my[Show More]Nor am I an anti-vaxxer. I had my first dose on 22 Nov 2019 with the local pharmacist. It took me several weeks to connect the fact of my recent vaccine and the pain in my left upper arm and limited reach could be connected with the shingles vaccine. At the six-week mark, I decided to make an appointment with my PCP. Prior to the appointment, the pain went away and my reach returned. I went for my second dose in early February 2020, this time prepared to track the pain and use of my arm with the vaccine. Two, going on three weeks later, I still have pain in my left arm, with limited mobility. Over-the-counter pain medications do nothing for it. I can still use it, but find the pain increases the more active I am. I spent the past two days shoveling snow and - it really hurts and my reach is limited. Both the pharmacist and my PCP have dismissed my comments about the pain and mobility. I was hoping they would share my feedback with Shingrix. I recognize this is mild to many of the people who have posted but I'm frustrated that no official cares to collect this data of adverse reactions and hope that the pain will recede soon.
RebeccaFebruary 12, 2021 at 7:12 pm
Had the shingrex shot, the 1st shot gave mea bad headache for 3 days, the second shot made my arm muscle hard with redness and a rash, that night I had very bad shivering with fever, the next day, today I have a very bad headache again. Even my teeth hurt, with a small irritation on roof of mouth. This is a rough vaccine to take. I hope it works after all this. And I hope these side effects go a[Show More]Had the shingrex shot, the 1st shot gave mea bad headache for 3 days, the second shot made my arm muscle hard with redness and a rash, that night I had very bad shivering with fever, the next day, today I have a very bad headache again. Even my teeth hurt, with a small irritation on roof of mouth. This is a rough vaccine to take. I hope it works after all this. And I hope these side effects go away.
PaulFebruary 3, 2021 at 1:42 pm
I am a 61 year old man who bikes regularly. I consider myself to be quite active with no underlying health issues. Received my first Shingrex vaccine shot and experienced fever and flu like symptoms lasting 36 hours. Subsequent to flu like symptoms I have developed what can best be described as tennis elbow. I am right handed, injection and tennis elbow are associated with my left arm. Three month[Show More]I am a 61 year old man who bikes regularly. I consider myself to be quite active with no underlying health issues. Received my first Shingrex vaccine shot and experienced fever and flu like symptoms lasting 36 hours. Subsequent to flu like symptoms I have developed what can best be described as tennis elbow. I am right handed, injection and tennis elbow are associated with my left arm. Three months hence and I’m left with limited use of my left arm. Would think long and hard about injecting this vaccine into your body.
ValerieJanuary 25, 2021 at 5:36 am
I had 2nd Shingrix shot on December 30, 2020. On January 13, 2021 a large Shingles eruption occurred (largest blister grouping I’be had in over ten years). Before the first Shingles outbreak healed, another, even larger outbreak erupted. I have had 2 Shingles outbreaks in less than 1 month. Back on the Acyclovir (not good because I have Chronic Kidney Disease) I am now having acyclovir side ef[Show More]I had 2nd Shingrix shot on December 30, 2020. On January 13, 2021 a large Shingles eruption occurred (largest blister grouping I’be had in over ten years). Before the first Shingles outbreak healed, another, even larger outbreak erupted. I have had 2 Shingles outbreaks in less than 1 month. Back on the Acyclovir (not good because I have Chronic Kidney Disease) I am now having acyclovir side effects I never had before. I didn’t have issues with Zostravax.
SarahJanuary 11, 2021 at 6:03 pm
I received both shots many months ago. I still cannot use my right arm. Made the mistake of getting it in my dominant arm. Extreme pain radiating down my arm and into my shoulder and back
KathyJanuary 11, 2021 at 4:59 am
I received my first Shingrix injection on 12/3/20. Immediately I had terrible pain in my arm, but figured it was just the possible side effect that would dissipate. It has been over 5 weeks now and I have limited use of my left arm and am in chronic, terrible pain. I move my arm certain directions and the pain is unbearable. I went to Urgent Care after one week and they diagnosed severe infl[Show More]I received my first Shingrix injection on 12/3/20. Immediately I had terrible pain in my arm, but figured it was just the possible side effect that would dissipate. It has been over 5 weeks now and I have limited use of my left arm and am in chronic, terrible pain. I move my arm certain directions and the pain is unbearable. I went to Urgent Care after one week and they diagnosed severe inflammation and pain and gave me meds for it as well as had me wear a splint. After 3 weeks I spoke to my pain doctor who gave me more anti-inflamatories. I am already on Morphine because of severe scoliosis of my spine, and it doesn't even touch the pain in my arm. I am going to physical therapy now to try to get more movement back in my arm. There is no way I will get the 2nd vaccine which is due 2/1/21...not after this nightmare and everything I've read here. I can only hope and pray the pain will diminish, but I am doubtful it will.
MaryKJanuary 9, 2021 at 3:47 pm
On 11/14/2020 @ my local Rite Aid where I have been using for all meds, vaccines, etc. for 8 1/2 years. I have never had a bad reaction to any vaccine in my whole life until that day w/ my 2nd shingles vaccine in 5 years. My normal Pharmacist was not in & the other pharmacist said he could give me this NEW strain. I've had much medical training & my Mom was a fine LPN for 50 years. This man hit a [Show More]On 11/14/2020 @ my local Rite Aid where I have been using for all meds, vaccines, etc. for 8 1/2 years. I have never had a bad reaction to any vaccine in my whole life until that day w/ my 2nd shingles vaccine in 5 years. My normal Pharmacist was not in & the other pharmacist said he could give me this NEW strain. I've had much medical training & my Mom was a fine LPN for 50 years. This man hit a nerve. He obviously wasn't trained correctly & I've been in severe pain since 11/14 & gotten. worse. My dr said go to ER in Red Bluff. I did. Reported back & dr saw the hospital report. He then prescribed a nerve medication & after taking 2 pills I had hallucinations & called to say that I can't take medication. It's been one crazy mishap & 4 weeks now I've been waiting for therapy on right arm. With holidays & misinformation for the referral I still suffering. I'm done! I need advice & called 4 days ago to say my MELOXICAM wasn't working, is there anything I can take? No word from drs ordering perhaps Celebrex? My health has totally become debilitating & I'm already disabled. Please can you help me? I called one of these sites back in Nov but I never got much satisfaction. Thank you
normDecember 23, 2020 at 8:43 pm
I had a single Shingrix shot on Oct 21,19 during a routine physical. I was not told there would be a second shot. At the time, I was a very physically fit 60 year old who swam two miles three times a week, no drinking, no drugs. Within a week of the shot I lost all use of my right arm. I regained much of its use but a month later lost all of my left arm function and everything below the waist. I w[Show More]I had a single Shingrix shot on Oct 21,19 during a routine physical. I was not told there would be a second shot. At the time, I was a very physically fit 60 year old who swam two miles three times a week, no drinking, no drugs. Within a week of the shot I lost all use of my right arm. I regained much of its use but a month later lost all of my left arm function and everything below the waist. I was subsequently diagnosed with transverse myelitis and have been unable to stand or walk ever since.
NancyDecember 16, 2020 at 5:24 am
I got my first vaccine at cvs. The pharmacist gave it IM and I had a little soreness but nothing else. Yesterday I went back for my second vaccine. The pharmacist started to give it to me subcutaneous and I told him that I had the first one IM. He said no, you get it subcutaneously. The second he pushed it in my arm started burning. Now it is a large red swollen warm area where it was put it. It[Show More]I got my first vaccine at cvs. The pharmacist gave it IM and I had a little soreness but nothing else. Yesterday I went back for my second vaccine. The pharmacist started to give it to me subcutaneous and I told him that I had the first one IM. He said no, you get it subcutaneously. The second he pushed it in my arm started burning. Now it is a large red swollen warm area where it was put it. It also has a knot in the red area. I’m really upset as I’ve also been getting very dizzy. I don’t know what to do.
JenniferDecember 6, 2020 at 4:56 am
I was ill with fever and chills my first injection but on my second injection of shingrix I was severely ill with a massive headache, severe body aches, fever for about 36 hours, injection site pain, and fatigue. It was horrific. It was like flu on steroids. I am a nurse and I could not have imagined I could feel that ill from a vaccination.
LisaNovember 27, 2020 at 4:24 am
I went for my yearly physical on 10/22/20. At the end of the visit my doctor recommended the Shingrix shot, flu shot and tetanus booster. I agreed to the Shingrix and Tetanus. I never take the flu shot. Had I known the hell I was in for, I would never had had the Shingrix. Within hours my arm was aching....then the chills, fever and body aches came. I work up to lesions covering the inside of my m[Show More]I went for my yearly physical on 10/22/20. At the end of the visit my doctor recommended the Shingrix shot, flu shot and tetanus booster. I agreed to the Shingrix and Tetanus. I never take the flu shot. Had I known the hell I was in for, I would never had had the Shingrix. Within hours my arm was aching....then the chills, fever and body aches came. I work up to lesions covering the inside of my mouth, within hours started to feel discomfort when urinating. Turns out I developed lesions on my labia. I'm a healthy 57 year old woman in a steady faithful relationship. I could barely sit without pain for a week. Went back to the medical center where I was given an exam and was tested for herpes since that's how it looked apparently. The test came back negative (which I knew it would). I was given anti viral meds. Also took some nerve numbing medication for shingles. Thankfully all the lesions just disappeared as fast as they came. However, I lost a week of work, questioned my amazing relationship due to feeling like a leper at the doctor and never felt so sick. It was Lot# YZ325. I believe this should be addressed. I can't be the only one.
CarolNovember 18, 2020 at 5:09 am
The pharmacist at the Kingsway Publix Store, Sue Le, administered the shot. She kept ramming and ramming and ramming my arm. The pain I have experienced from her performance, or I should say lack of, has put my left arm into severe, horrifying pain. There was no reason for her to do to me what she did. I have had to take prescription pain pills for my arm pain and anti inflammatory pills.
ValerieNovember 16, 2020 at 5:03 pm
I received the Shindrex vaccine in one dose on Oct.30th. A week later I experienced severe pain on my left posterior back radiating down to my hip. Nov.14 the raised blisters rash appeared and I was diagnosed with shingles in a medicentre. I am now on antiviral medication and have to take T3's and Ketoralac to manage the pain.
LarissaNovember 3, 2020 at 7:59 am
I just got my Shingrix booster at age 52. After the 1st shot, two months ago, I felt under the weather for a day or so. But this time along with the general malaise, I’ve got other symptoms. It’s 3 days after the 2nd vaccine booster and I’m having sharp pains off and on in my left ear. (Same side I got shot in). I’ve never in my adult life had ear pain. Or any physical problems really. Hoping it’[Show More]I just got my Shingrix booster at age 52. After the 1st shot, two months ago, I felt under the weather for a day or so. But this time along with the general malaise, I’ve got other symptoms. It’s 3 days after the 2nd vaccine booster and I’m having sharp pains off and on in my left ear. (Same side I got shot in). I’ve never in my adult life had ear pain. Or any physical problems really. Hoping it’ll go away on its own. Also the roof of my mouth on left side is sore and ulcerated. And generally feeling tired. The ear pain is concerning me the most as I’ve never had that before, nor do I have a cold or allergies.
EileenOctober 23, 2020 at 7:12 am
Five days after the Shingrix vaccine my left shoulder started hurting and it is still very painful. It has been 11 days now. I can’t raise my arm without crying. This sucks! I also felt like I was getting sick day 4 and had red itchy skin where they gave me the shot. I hope it improves or I don’t know what I’ll do.
ValorieOctober 21, 2020 at 3:14 am
I received the shingles vaccine at the Lansing VA during a doctor visit for a different reason. The nurse talked me into getting the vaccine due to the possible pain of shingles. I have had chicken pox in the past. That evening my tongue swelled some. The next two days i was in horrible pain all over my body. On the following day, a Monday, I called the VA. The pain was still in my legs. The p[Show More]I received the shingles vaccine at the Lansing VA during a doctor visit for a different reason. The nurse talked me into getting the vaccine due to the possible pain of shingles. I have had chicken pox in the past. That evening my tongue swelled some. The next two days i was in horrible pain all over my body. On the following day, a Monday, I called the VA. The pain was still in my legs. The pain has remained in my legs ever since. I was advised by my pcp not to take the 2nd part. I'm not an anti-vaccer. I am a veteran who had a much better quality of life prior to this vaccine.
LyleOctober 18, 2020 at 10:11 pm
I’m a 75 year old man, I had the original shingles shot almost twenty years ago and never had shingles. On October 2, 2020 I was talked into getting the new improved shingles shot Shingrix. I’m very healthy don’t smoke or drink with perfect blood pressure. Two days after receiving this shot I felt terrible, The shot was given to me on my right shoulder and was very painful for several days. Two da[Show More]I’m a 75 year old man, I had the original shingles shot almost twenty years ago and never had shingles. On October 2, 2020 I was talked into getting the new improved shingles shot Shingrix. I’m very healthy don’t smoke or drink with perfect blood pressure. Two days after receiving this shot I felt terrible, The shot was given to me on my right shoulder and was very painful for several days. Two days after receiving this shot the muscle in the right side of my back was hurting, I went to my chiropractor that Monday four days after receiving the shingles shot and not knowing why my back was hurting so bad, two days later Wednesday it was hurting worse and went back to my chiropractor and didn’t resolve my issue! That night Wednesday/ Thursday 3 AM my back and spine was broken out with shingles. I was able to make Dr. appointment that morning and before I came in he checked to see if there was any data on people getting shingles after receiving this shot produced by GSK he said there was any data. He confirmed I had shingles and put me on Valtrx and Gapentetin. Here I am 10 days later after getting that shot on my right shoulder and I’m having to deal with shingles and unbearable nerve pain down my right thigh and up my right chest 24/7 this is not a coincidence like they want you to believe. And after reading all these stories similar to mine GSK has a legal problem and I’m going to file a lawsuit pain and suffering and fraud. I can’t believe how I was duped into getting this poison injected into my body with the potential of lingering nerve pain that could last for months or even years.
SusanOctober 11, 2020 at 2:15 pm
I had the Shingrix vaccine on 10/7/20 at Sam's Club.I was not given a handout or told of any possible adverse effects. About 6 hours after receiving the vaccine I started feeling terrible. I had chills, shivering, fever, weakness, severe headache, dizziness and difficulty walking or moving. I was completely bed bound and slept around the clock. On 10/9/20 I was still feeling weak and like I had a [Show More]I had the Shingrix vaccine on 10/7/20 at Sam's Club.I was not given a handout or told of any possible adverse effects. About 6 hours after receiving the vaccine I started feeling terrible. I had chills, shivering, fever, weakness, severe headache, dizziness and difficulty walking or moving. I was completely bed bound and slept around the clock. On 10/9/20 I was still feeling weak and like I had a terrible case of flu. In the early afternoon I had a sudden onset of tachycardia and went to the ER. I was diagnosed with paroxysmal Afib. It resolved on its own, but I was admitted to the hospital. I was discharged late in the evening the next day and am having to take Metropylol to prevent a recurrence of Afib.Today is 10/11/20 and I still feel terrible with headache & profound weakness & dizziness leaving me bedbound. I have never had any cardiac issues and have been in excellent health at age 64. I will not get the second dose & I am concerned these adverse effects may cause permanent damage to my health. I would never have agreed to the Shingrix vaccine if I'd been told of possible adverse side effects.
DavidSeptember 5, 2020 at 9:41 pm
I received a Shingrix vaccine on the day of my physical, which was stellar for a man of 61 years old. I do not smoke, drink, have never done drugs and do not take any medications for any underlying health conditions. I took the 1st shot on 8/17/20, and on 8/27/20 began having pain in my legs. The next day I began having several falls and by that afternoon was unable to walk. I was admitted to [Show More]I received a Shingrix vaccine on the day of my physical, which was stellar for a man of 61 years old. I do not smoke, drink, have never done drugs and do not take any medications for any underlying health conditions. I took the 1st shot on 8/17/20, and on 8/27/20 began having pain in my legs. The next day I began having several falls and by that afternoon was unable to walk. I was admitted to the hospital and diagnosed with Gullien-Barre Syndrome. I am waiting to be transfers to an acute care rehabilitation facility and my recovery if successful may take several weeks, months, or even years. I was definitely affected by this vaccine in a terrible way, and I do not remember being told GSB was a possible side-effect.
MaxineAugust 27, 2020 at 3:23 am
Got the new shingles vaccine first dose .I got it August 21/20. I had a sore arm at injection site . That has subsided , however I got dome rash and extreme itching a week later . I’ve have been fatigued and have body aches . The itchy is waking me I middle of night . I hope these side effects go away soon !
lauraAugust 25, 2020 at 9:06 pm
I had my vaccinations yesterday I woke up and thought I was dying from the Corona virus have never experienced so much pain and fever and chills I am so upset that I did it I have side effects of Most medications IDK why the doctor would give it to me
GinnyAugust 18, 2020 at 7:47 am
I underwent my first shot of shingrix in May and had some side effects. I hv a high tolerance so could handle it. 3 months passed and I underwent my 2nd shot. Wow it gave me very terrible body ache which was like bone pain even in my thumbs, fingers and toes and I had a very difficult night with pain and chills. Took Tylenol for a few days to deal with it but it's been day 5 and I still feel leth[Show More]I underwent my first shot of shingrix in May and had some side effects. I hv a high tolerance so could handle it. 3 months passed and I underwent my 2nd shot. Wow it gave me very terrible body ache which was like bone pain even in my thumbs, fingers and toes and I had a very difficult night with pain and chills. Took Tylenol for a few days to deal with it but it's been day 5 and I still feel lethargic and on and off headaches which gives me a feeling of malaise. I used to exercise daily and walk for 45 mins every day or play tennis prior to the 2nd shot, but somehow I am feeling like too strained to do it since. I am not sure why and I hope this wears off soon.
ThereseAugust 15, 2020 at 1:52 am
I got my first dose of Shingrix about a month ago at the Pharmacy. I did get a mild case of shingles on my left side. I also had extreme fatigue for three days. My injection site was originally red, hard and sore. That evolved into itching and a small pimple. To this day the site is hard, warm and deep inside the middle of deltoid I have tenderness to my elbow. Also when using my arm I will exper[Show More]I got my first dose of Shingrix about a month ago at the Pharmacy. I did get a mild case of shingles on my left side. I also had extreme fatigue for three days. My injection site was originally red, hard and sore. That evolved into itching and a small pimple. To this day the site is hard, warm and deep inside the middle of deltoid I have tenderness to my elbow. Also when using my arm I will experience a deep stabbing pain in the same area. I am afraid to get the second dose. I have had my warning with the problems I have experienced to date. I hope nothing more serious happens to me.
RoselineAugust 14, 2020 at 9:32 pm
I had the shingle vaccine august 8th .Iam not going for the 2nd dose in sept 2020.I had terrible migraine headace fatigue no appetite up to this present moment I feel sick sleep and sleep.Several red spot patches under both arm pit also my hands itching .The left side I got vaccine itch swollen very painful.I got dizzy spell.Iam so worried .Dont like the way I feel.still have the terrible headace[Show More]I had the shingle vaccine august 8th .Iam not going for the 2nd dose in sept 2020.I had terrible migraine headace fatigue no appetite up to this present moment I feel sick sleep and sleep.Several red spot patches under both arm pit also my hands itching .The left side I got vaccine itch swollen very painful.I got dizzy spell.Iam so worried .Dont like the way I feel.still have the terrible headace .Iam diabetic
RoselineAugust 14, 2020 at 9:28 pm
I had the shingle vaccine august 8th .Iam not going for the 2nd dose in sept 2020.I had terrible migraine headace fatigue no appetite up to this present moment I feel sick sleep and sleep
DeborahAugust 9, 2020 at 8:43 am
I went to my PCP for an annual check up back in September 2019 and was advised that I should get the shingles vaccination, being over 50 years old. The office didn't have the vaccination available, so I was advised that I could go into my local pharmacy to get the vaccination. I went to the pharmacy on October 3rd and received the Shingrix vaccination, and of course was never told or read in the [Show More]I went to my PCP for an annual check up back in September 2019 and was advised that I should get the shingles vaccination, being over 50 years old. The office didn't have the vaccination available, so I was advised that I could go into my local pharmacy to get the vaccination. I went to the pharmacy on October 3rd and received the Shingrix vaccination, and of course was never told or read in the information what happened to me next...3 days later, I was in full blown shingles. I went to the ER because I was experiencing chest pains and a blister on my back started forming. I was told that I had contracted the shingles. The shingles covered the ban around my upper back/chest area, I was in excruciating pain due to this outbreak. I am still, to this day, experiencing post neuralgia pain and it affects my lifestyle tremendously.
EyobJuly 31, 2020 at 9:37 am
I had Shingrix vaccine in October 2019 and the second one in March this year. Since I had this vaccine my vision decreased and I have some other issues with my eyes. I go for a check up every year and when I was at my eye doctor last year April every thing was fine with my eyes. I know that Zostavax created a lot of health issues, Shingrix maybe has also some side effects.
LaneJuly 27, 2020 at 1:55 pm
First of all i am NOT an "anti-vaxxer" person that fabricates stories about reactions to vaccinations etc. I am a perfectly healthy 56 year old active male. I recently went in to my pcp for my annual physical. He suggested the shingles vaccine. Explained it is a 2 part deal will have some discomfort at the injection site blah blah blah. Sure, sounds reasonable plus i have seen what my 82 year ol[Show More]First of all i am NOT an "anti-vaxxer" person that fabricates stories about reactions to vaccinations etc. I am a perfectly healthy 56 year old active male. I recently went in to my pcp for my annual physical. He suggested the shingles vaccine. Explained it is a 2 part deal will have some discomfort at the injection site blah blah blah. Sure, sounds reasonable plus i have seen what my 82 year old Mother has gone through with shingles outbreaks recently. So the nurse administers the first vaccine. It was a little uncomfortable but nothing unbearable. Arm a little sore that evening, took some ibuprofen went to bed. Woke up about 2am chills, vomiting, every joint aching and a 102 fever. This lasted about 36 hrs. Not sure if i will go back for the 2nd round!!
HeidiJuly 16, 2020 at 3:09 pm
My 88 year old female friend had Shingrix shot in December & had horrible mouth ulcers for weeks. She could barely drink water & broth due to the pain, but we couldn’t figure out what it was & Dr’s had no remedy. One Dr literally shrieked when she looked in her mouth! She just got the 2nd shot a few weeks ago & they came back a few days later. That’s when we figured it out!
StaceyJuly 14, 2020 at 11:33 pm
My mom was given the first dose of Shingrix. Two months later, she contracted a viral ear infection- lab tests showed this virus was related to the shingles virus (?)... Her inner ears were so inflamed she lost 100% of her hearing in less than a day and had to be hospitalized for months with a wackload of other symptoms seemingly coming up out of nowhere. Diabetes type II, diabetes inspidus, pitui[Show More]My mom was given the first dose of Shingrix. Two months later, she contracted a viral ear infection- lab tests showed this virus was related to the shingles virus (?)... Her inner ears were so inflamed she lost 100% of her hearing in less than a day and had to be hospitalized for months with a wackload of other symptoms seemingly coming up out of nowhere. Diabetes type II, diabetes inspidus, pituitary gland tumour, lung nodule, and finally vasculitis. Treatment for the vasculitis was chemo (free), or an expensive alternative with fewer side effects... no brainer for our family. Forked out the money but there's no guarantee that this treatment would cure the disease. However, it was considered superior to the chemo. If it doesn't cure the vasculitis she has to do another round of it. [Aside: met a lady at the hospital drug administration unit during the first treatment dose. This lady had the same vasculitis disease as my mom and she takes this expensive drug every few years as maintence...]. 12% of her hearing came back, and she'll be getting cataract removal surgery soon to remove the cataracts that grew insanely thick after all the high dose steroid treatments she's had to be on. More money being forked out due to coverage for only the most basic treatment. Again, no brainer because the woman has been through enough.
On to the gameJuly 14, 2020 at 2:27 am
This is an unmoderated comment section filled with unsubstantitated injury claims. Anti-vaxxers seek out these forums to post messages claiming all sort of horrific injuries caused by a vaccine to scare people. The majority of these complaints are far-fetched and unattributable to the 2-part shingrix injection.
MichaelJuly 12, 2020 at 2:53 pm
Visited doctor for physical to receive diabetic meds. Doctor said I need the shingles vaccine. I told her not to give me anything that will mess me up. She said all I will feel is a slight headache and gave me a Shingrix shot. The next day I felts bad all day. Flu symptoms that night. Next day started itching in a few places. That night hives started covering my body. I have been fighting hives fo[Show More]Visited doctor for physical to receive diabetic meds. Doctor said I need the shingles vaccine. I told her not to give me anything that will mess me up. She said all I will feel is a slight headache and gave me a Shingrix shot. The next day I felts bad all day. Flu symptoms that night. Next day started itching in a few places. That night hives started covering my body. I have been fighting hives for three days. I hope this ends soon.
SteveJuly 12, 2020 at 1:49 am
Have hip surgery just four months ago before taking the Shingrix vaccine. It caused high fever and severe pain at the inner upper thigh joint area of the operative leg. Feel as bad as joint dislocation. Talked to doctor and no one seem to know of such side effect so far.
PaulJune 25, 2020 at 4:45 pm
My wife had a second vaccine and three hours later suffered a massive hemorrhage stroke
PatriciaJune 11, 2020 at 5:59 pm
2 weeks after receiving my 2nd Shingrix vaccine I developed debilitating joint pain to my L shoulder and bilateral knees. It came on suddenly and it's progressively getting worse. I was seen by my orthopedic MD who could find nothing on the x rays. I walk everyday 3-6 miles and now I am basically bed bound. I did not put 2 and 2 together since it did not appear immediately after the injection. [Show More]2 weeks after receiving my 2nd Shingrix vaccine I developed debilitating joint pain to my L shoulder and bilateral knees. It came on suddenly and it's progressively getting worse. I was seen by my orthopedic MD who could find nothing on the x rays. I walk everyday 3-6 miles and now I am basically bed bound. I did not put 2 and 2 together since it did not appear immediately after the injection. I was not warned of this. I would risk my chances with the shingles if i had known of this pain. It is life changing with no relief in sight. My MD was not aware of this side effect either. When I researched the side effects I have found on here the horrible stories.
CharlesMay 18, 2020 at 9:25 pm
I have a horrendous story to tell of what the Shingrix vaccine has done to me. (CDC says I’m the one in 6) Did not have a problem , it was recommended by my General MD and my life has been a physical hell the last 1 1/2 years with boils appearing all over my body where there are lymph nodes .
CecilMay 13, 2020 at 4:03 am
I received the first Shingrix shot on 15 Jan 2020 and my arm ached a month with flu-type symptoms, three weeks later my hands and knees developed arthritis swelling and pain barely managed with ibuprofen. That faded and I developed a few episodes of moderate sciatica which subsided around the end of March. This past week the sciatica returned with debilitating pain (10 of 10) and resulted in a me[Show More]I received the first Shingrix shot on 15 Jan 2020 and my arm ached a month with flu-type symptoms, three weeks later my hands and knees developed arthritis swelling and pain barely managed with ibuprofen. That faded and I developed a few episodes of moderate sciatica which subsided around the end of March. This past week the sciatica returned with debilitating pain (10 of 10) and resulted in a medical teleconference due to Covid-19. After recovering from the last event and with hindsight today I realized the source of my problems. I will not be getting the second shot.
DonnaMay 9, 2020 at 2:41 pm
I got my 1st shot of shingex on November 19 2019. Not long after that started having issues with not having contol of relieving myself and some really serious back pain. I couldn't make it to the bathroom in time on many occasions. I had to just wet on the floor beside my bed. I started keeping towels by my bedside. And while trying to keep a somewhat normal routine, I would just mess myse[Show More]I got my 1st shot of shingex on November 19 2019. Not long after that started having issues with not having contol of relieving myself and some really serious back pain. I couldn't make it to the bathroom in time on many occasions. I had to just wet on the floor beside my bed. I started keeping towels by my bedside. And while trying to keep a somewhat normal routine, I would just mess myself. Could not control my bowels. I assumed it was from by back issues...swelling, etc not having room inside from the swelling and full bowels and bladder. I did not tell anyone because I was embarrassed. Now to the back pain. I have had sciatic issues before but not this extreme. This was around Thanksgiving and Christmas, I called my neurologist, I received 3 shots to help with the pain. Hardly touched the pain i was having. Finally I coul not get any more shots. The doctor ordered exrays and MRI'S. They saw a cloudy area near my spine. They really didn't know what it was...som w kind of infection. They would not prescribe any meds until more test to determine what kind of infection. Finally got a antibiotic that would cover most any infection. Went back for more MRI'S , the cloudy area was gone. I was told it could been several different things nothing definite. I was told that I gave spinal stenosis. I had tons if blood work looking for cancer, etc. Nothing. I cannot even start to explain how much pain I was in. It was cramping muscles and would not stop. Felt kinda like someone had hold of my nerve and rubbing it together with sandpaper. I have cried and cried at home, nights, days, while at work..all the time. The major pain is gone after the antibiotics, but I still have pain in my lower buttocks. I didn't put 2 and 2 together until I read an article on the shingles vaccine.
MichaelMay 1, 2020 at 3:40 pm
I received my first shingles (shingrix) shot towards the end of August 2019. On our way down to Kentucky to visit our daughter a few days later, my legs began to develop red spots. This continued to worsen over the next several days, and I ended up going to a hospital emergency department in Lexington soon after we arrived. This turned into vasculitis and I had open sores on both legs, especially [Show More]I received my first shingles (shingrix) shot towards the end of August 2019. On our way down to Kentucky to visit our daughter a few days later, my legs began to develop red spots. This continued to worsen over the next several days, and I ended up going to a hospital emergency department in Lexington soon after we arrived. This turned into vasculitis and I had open sores on both legs, especially around my ankles. I had bandages on both legs for over three months. Along with that I had neuropathy (that pins and needles feeling) in both legs and it was very painful. The neuropathy finally went away, but after the sores healed (these looked like something out of a forensic pathologist's book), it left reddish scars that I still have. I called the attorneys who are handling the class action suit, but nobody every returned my call. Needless to say, I did not get my second shot, as it's administered in two stages.
WendyApril 25, 2020 at 4:53 pm
I received the first Shingrix vaccine in early November of 2019. Two weeks later to the day I completely lost hearing in my left ear. I have horrible tinnitus as well. I have been to an ENT specialist who prescribed oral steroids as well as steroid injections. I am still deaf in my left ear. I am writing this in April of 2020. I am 58 years old. This has been especially traumatic because I am a re[Show More]I received the first Shingrix vaccine in early November of 2019. Two weeks later to the day I completely lost hearing in my left ear. I have horrible tinnitus as well. I have been to an ENT specialist who prescribed oral steroids as well as steroid injections. I am still deaf in my left ear. I am writing this in April of 2020. I am 58 years old. This has been especially traumatic because I am a retired Language teacher. I currently work as an ESL tutor at a public middle school. This job brings me purpose and joy. I hope and pray that I will be able to continue.
LauraApril 21, 2020 at 5:58 pm
I had my first Shingrix in my left arm in May 2019. I had the expected side effects of fatigue and sore arm which I signed a consent acknowledging side effects. The sore arm has never gone away and now seems worse with numbness and tingling and limited range of motion due to pain in the bicep area. Maybe the needle went into the bicep instead of the deltoid or maybe the needle hit the tendon bene[Show More]I had my first Shingrix in my left arm in May 2019. I had the expected side effects of fatigue and sore arm which I signed a consent acknowledging side effects. The sore arm has never gone away and now seems worse with numbness and tingling and limited range of motion due to pain in the bicep area. Maybe the needle went into the bicep instead of the deltoid or maybe the needle hit the tendon beneath. I don't know. I feel a lump on the surface of the bicep. External rotation of my left arm is excruciatingly painful. My second Shingrix was in the right arm in August - no symptoms at all. Why would I have a sore arm for months and then a flare up like this????
MaggieMarch 26, 2020 at 5:35 pm
I received this injection October 2019. My life has been a living hell since then. My deltoid is still sore with a knot in it, after the injection it was pie-left and swollen for 8 weeks. I had flu like symptoms for 3 days with temp of 103-104 degrees, nausea, aching all over. Then to my shagrin I starting getting burning and tingling in my feet and sudden, severe low back pain. I now have constan[Show More]I received this injection October 2019. My life has been a living hell since then. My deltoid is still sore with a knot in it, after the injection it was pie-left and swollen for 8 weeks. I had flu like symptoms for 3 days with temp of 103-104 degrees, nausea, aching all over. Then to my shagrin I starting getting burning and tingling in my feet and sudden, severe low back pain. I now have constant ringing and buzzing in both ears and a strange vibration throughout my body. I am being evaluated now for chronic inflammatory demyelinating polyneuropathy. I would rather have the shingles! If I could go back in time I would never get this shot, what’s left of my life is a nightmarish hell! I am having to postpone neurologist evaluation until Coronavirus crisis has passed. I am having blurred vision and other visual disturbances. The numbness is in both feet but particularly great toe and second toe and the muscle twitching feels like electrical impulses traveling in my calves and feet. Do not get this shot! Has died down.
JohnMarch 16, 2020 at 3:36 pm
I had shingles in 2007, and did not want to have them again, so I was eager to get the new shingles vaccine. My pharmacy finally had availability in January, so I had the first shot one week before my 74th birthday, the 20th. On the 22nd, while showering, I felt the equivalent of an electric shock in my right ear, and I suffered a sudden hearing loss. My otorhinolaryngologist put me on steroids[Show More]I had shingles in 2007, and did not want to have them again, so I was eager to get the new shingles vaccine. My pharmacy finally had availability in January, so I had the first shot one week before my 74th birthday, the 20th. On the 22nd, while showering, I felt the equivalent of an electric shock in my right ear, and I suffered a sudden hearing loss. My otorhinolaryngologist put me on steroids, which reduced the hearing loss temporarily, but did not restore full hearing. After the hearing loss seemed to worsen, he put me on a virus suppressant, which again seemed to help while I was on it. Now the hearing loss continues, accompanied by loud tinnitus in that ear, and may be spreading to my left ear. As a semi-professional musician, I would much rather have shingles again than lose my hearing in either ear, much less both. Nothing warned against such a potential side effect, but testimonies on this site make clear it may be one.
DanielMarch 7, 2020 at 2:46 pm
I got my shingrix shot over month ago. The first night woke up with chills and flu like symptoms which lasted for a couple days-also stinging fingers--sore arm/shoulder,weird muscle aches.. I still have shoulder pain now after a month!!and stiff neck.. I am 66 ,not on any meds.I do yoga ,exercise, work out ,ride my bike and now massage my shoulder.. I wish I had read up on the side effects. hope[Show More]I got my shingrix shot over month ago. The first night woke up with chills and flu like symptoms which lasted for a couple days-also stinging fingers--sore arm/shoulder,weird muscle aches.. I still have shoulder pain now after a month!!and stiff neck.. I am 66 ,not on any meds.I do yoga ,exercise, work out ,ride my bike and now massage my shoulder.. I wish I had read up on the side effects. hopefully the shoulder pain will go away.I appear to be one of the lucky ones without more serious side effects so far....I will NOT get the second shot...
josephineMarch 1, 2020 at 1:28 am
I had no problem with first shingle shot had second one can’t hardly lift my left arm biggest problem my neck I have to hold it straight or pain shoots up back of neck been like this four days
laurieannFebruary 28, 2020 at 7:00 pm
I had my 1st and only Shingrx Vaccince 6/26./2019 no reaction at first. I had a stroke on 12/11/2019 with no none health risk ! is it possible that the vaccince had something to do with it ? Had to get TPA to break up the clot ended up in ICU. All follow up test have be negative for heart issuse , clotting , vascular, neuro. lost the whole right side of by body. Has this happen to anyone else ? No[Show More]I had my 1st and only Shingrx Vaccince 6/26./2019 no reaction at first. I had a stroke on 12/11/2019 with no none health risk ! is it possible that the vaccince had something to do with it ? Had to get TPA to break up the clot ended up in ICU. All follow up test have be negative for heart issuse , clotting , vascular, neuro. lost the whole right side of by body. Has this happen to anyone else ? Nobody can give me answer. Thank You
TeresaFebruary 18, 2020 at 1:15 am
My husband, who walked 2 miles a day and lifted weights three times a week at our local gym, contracted shingles 8 days after receiving the second Shingrex vaccination. He had varicella encephalitis two days later. Within two weeks, he got two brain bleeds. He was in two hospitals, and three rehab facilities but had been 3 months now and he still can’t walk, his bladder function is not under contr[Show More]My husband, who walked 2 miles a day and lifted weights three times a week at our local gym, contracted shingles 8 days after receiving the second Shingrex vaccination. He had varicella encephalitis two days later. Within two weeks, he got two brain bleeds. He was in two hospitals, and three rehab facilities but had been 3 months now and he still can’t walk, his bladder function is not under control, and he is unable to continue working. We also had to forgo a cruise to South America and Viking would not reimburse us on anyway.
ArtFebruary 11, 2020 at 8:54 pm
I recieved the Shingrix vaccine Oct 18 2019 and after 3 visits to the ER it was deterimined I had Shingles. After the 3rd day I had the Rash down my right arm and hand. I was treated with the antibiotics and after a few weeks the rash went away but the pins and needles pain has continued to this day. I have other symptoms including the feeling of frozen Toes in both feet, My vision in both eye's h[Show More]I recieved the Shingrix vaccine Oct 18 2019 and after 3 visits to the ER it was deterimined I had Shingles. After the 3rd day I had the Rash down my right arm and hand. I was treated with the antibiotics and after a few weeks the rash went away but the pins and needles pain has continued to this day. I have other symptoms including the feeling of frozen Toes in both feet, My vision in both eye's has gotten worse and my overall nervous condition has forced me to quit my retirement job two weeks ago and two years earlier than I planned. I will not be getting the 2nd dose. A report was sent to shingrix about my situation but they have not yet responded.
EileenFebruary 11, 2020 at 12:10 am
I received my first shingrex shot on May 23, 2019 with no side effects the second one I received on July 24. 2019. The second one has given me problems since getting it my arm was sore for weeks. The night I received the shot I was paralyzed in my bed when I could finally wake up I could barely stand up. I have never felt so horrible in my life my legs and every part of my body was weak and to th[Show More]I received my first shingrex shot on May 23, 2019 with no side effects the second one I received on July 24. 2019. The second one has given me problems since getting it my arm was sore for weeks. The night I received the shot I was paralyzed in my bed when I could finally wake up I could barely stand up. I have never felt so horrible in my life my legs and every part of my body was weak and to this day I have not regained my strength back.
TanyaFebruary 9, 2020 at 6:32 pm
Dad age 68 got his 1. shingrix vaccine end of Oct 2019. 10 days later the armpit lymph node grew to the size of an apple. End up in ER due to stomach pain and arm pit 'apple'. ER too CT said other lymph nodes enlarged. Recommended PET scan. In meanwhile, the armpit lymph node started leaking water for 2-3 days and the size and pain went down. PET scan reveled some radioactive trace uptake. And rec[Show More]Dad age 68 got his 1. shingrix vaccine end of Oct 2019. 10 days later the armpit lymph node grew to the size of an apple. End up in ER due to stomach pain and arm pit 'apple'. ER too CT said other lymph nodes enlarged. Recommended PET scan. In meanwhile, the armpit lymph node started leaking water for 2-3 days and the size and pain went down. PET scan reveled some radioactive trace uptake. And recommended thyroid ultrasound and hematologist for blood test. Thyroid ultrasound at 1. hospital reveled L 9 mm nodule, R 11 mm nodule (needs biopsy). Went to 2. hospital (after 1 wk). Took another ultrasound, L 7 mm nodule (too small to biopsy), R nodule VANISHED. 1. hospital fudged the results by playing with the ultrasound contrast. We went to the 2. hospital for evaluation of possible low grade lymphoma. No symptoms, no history of cancer, no fever, no rash, all blood work NORMAL even leukocytes normal and even went down from 1590 to 1300 in 2-3 weeks. Docs are covering up the shingrix side effects. Shingrix its a gift that keeps on giving to the Medical Industrial Complex.
LyndaFebruary 7, 2020 at 4:34 pm
Received the Shingrix vaccine on January 21, 2020. I have had chronic muscle aches in my arms, legs and neck since that time and chronic tiredness. I have had back pain that was at a level 2-3 and is now a 10 since this vaccine. I will not get the second dose. Hope all of this goes away and is not permanent.
MVFebruary 3, 2020 at 12:28 am
Had Flu like symptoms and my arm was very soar, also my Sciatica was so painful for almost 2 weeks...At this point I choose not to have the second vaccine and still have a bruise from the first one..had a hard time breathing, vision was totally impaired and f..k this!!! Lost the desire to not having the itch!
ThomasJanuary 31, 2020 at 4:46 pm
I got the first of two vaccine shots for Shingles on January 6th, 2020 and it did make my arm a little sore for a couple of days but nothing unusual. Then a couple weeks later I noticed that my tongue felt off, kind of numb, a few days later I started getting a pain here and there under my right ear, it was a semi severe pain but it only lasted for what seemed maybe 5 to 10 seconds and would go a[Show More]I got the first of two vaccine shots for Shingles on January 6th, 2020 and it did make my arm a little sore for a couple of days but nothing unusual. Then a couple weeks later I noticed that my tongue felt off, kind of numb, a few days later I started getting a pain here and there under my right ear, it was a semi severe pain but it only lasted for what seemed maybe 5 to 10 seconds and would go away. A few days later on the 27th my tongue felt even more numb, it was affecting my taste ability. It all seemed like no big deal and that it would go away at some point. Unfortunately this hasn't been the situation, the following day on the 28th I did some errands, came home and made dinner and everything was fine. Then about an hour later I started to notice that my lips felt kind of numb and things didn't feel right so I called a 24 hour nurse for a phone evaluation. She told me to get to an emergency room so I did and they were quick to get me in, my symptoms (I was told later) mimicked a possible stroke. I was admitted and run through a number of tests and seen by a number of doctors and admitted into the hospital. The end result was that I have Bell's Palsy. This is the worst thing I have had to deal with... the right side of my face is somewhat paralyzed and my speech is impaired. Since I left the hospital my speech has gotten worse. Could all this be a side effect of the shingles vaccine? I don't know for sure but a doctor said that it's possible, they all kept asking if I had been sick with any kind of cold or virus but I generally never get sick. So now I'm hoping that I'm one of lucky ones that recovers from this and I'm trying to stay positive but it's tough when I dwell on the thought of the idea that this could be permanent. At this point I can only wait day by day and hope that there will be future improvements. So it's extremely doubtful that I will get the second part of shingles vaccine. I will try and post an update in the future of my recovery.
DeniseJanuary 13, 2020 at 1:35 am
Had shingles years ago and got the Zostavax vaccine. The only side effect was a sore arm. Got the 1st new RZV shot in early November. Had 3 colds in December. Got my second RZV January 8, 2020. As I was given the shot I felt it travel up to my neck. I have had very sore muscles all over and chills. Also very fatigued last nite but couldn’t sleep until 4 am. Woke up this morning and went to the r[Show More]Had shingles years ago and got the Zostavax vaccine. The only side effect was a sore arm. Got the 1st new RZV shot in early November. Had 3 colds in December. Got my second RZV January 8, 2020. As I was given the shot I felt it travel up to my neck. I have had very sore muscles all over and chills. Also very fatigued last nite but couldn’t sleep until 4 am. Woke up this morning and went to the restroom an had blood in my urine. (I am 69 years old). Anyone else have this reaction?
SaraJanuary 2, 2020 at 8:22 pm
I was a healthy 76 year old female, not on any medication AT ALL. This vaccine was highly recommended by my PCP. Received the 1st Shingrix vaccination in Jan 2019, by the end of the month I could not even lift my arm to put deodorant on, throbbing pain at 10+, unable to sleep at night, numbness in hand and fingers, along with extreme fatigue. I suffer with pain up and down my neck. I was dia[Show More]I was a healthy 76 year old female, not on any medication AT ALL. This vaccine was highly recommended by my PCP. Received the 1st Shingrix vaccination in Jan 2019, by the end of the month I could not even lift my arm to put deodorant on, throbbing pain at 10+, unable to sleep at night, numbness in hand and fingers, along with extreme fatigue. I suffer with pain up and down my neck. I was diagnosed with “frozen shoulder”. I had to start wearing garments I could put on without using both arms. Received booster in 4/19 on other arm. I have had to see Neurologist, have CAT scan, be in PT for three months, see an Ortho for cortisone. I was given a a prescription of gabapentin (a habit forming medication). My range of motion is very, very limited now. As of today’s date I am scheduled for carpal tunnel surgery 1/6/20. Before this vacation, I had a yearly check up for the past nine years- that’s it! This vaccine is deadly! I also am contacting the CDC regarding this faulty vaccine! #MASS TORT!
CynthiaJanuary 1, 2020 at 6:50 pm
I was Vaccinated for shingles in September. About 12 hours later I woke up in the middle of the night with serious bed spins and Vertigo such that I began vomiting. I was unable to walk sit up straight or even stand. I was eventually diagnosed with Labyrinthitis. The cause of which was the introduction of the shingles virus which has introduced itself to my brain causing lack of balance. For th[Show More]I was Vaccinated for shingles in September. About 12 hours later I woke up in the middle of the night with serious bed spins and Vertigo such that I began vomiting. I was unable to walk sit up straight or even stand. I was eventually diagnosed with Labyrinthitis. The cause of which was the introduction of the shingles virus which has introduced itself to my brain causing lack of balance. For the 1st couple of months I was almost unable to do anything. I am 57 years old and I am extremely active and healthy. However now I feel like I'm totally disabled. While it has gotten somewhat better there are days when I can't even get off the couch. Here it is December and I'm still fighting this illness because of us simple vaccine that I was told I needed because I'm over 50. It's been terrible absolutely terrible living through this period and I'm told it might take an entire year before in better. I hope that I am able to return to my active ways however I'm very suspicious. I used to be able to do things like ride my horse, Kayak, exercise daily, hike, and scuba dive. I Feel robbed!
EmmaDecember 30, 2019 at 3:38 pm
I received the first Shingrix injection on my left arm on 9/9/2019, and experienced the horrible flu-like symptoms for three days. The arm pain was difficult to handle. On 11/8/2019, I received the second vaccine on my right arm. The pain on my arm has lasted for several weeks. There is a constant feeling of numbness from my armpit to my fingertips. It is very painful and has caused me to experien[Show More]I received the first Shingrix injection on my left arm on 9/9/2019, and experienced the horrible flu-like symptoms for three days. The arm pain was difficult to handle. On 11/8/2019, I received the second vaccine on my right arm. The pain on my arm has lasted for several weeks. There is a constant feeling of numbness from my armpit to my fingertips. It is very painful and has caused me to experience much sleep deprivation. My arm feels as if there is a ball of pain at the injection site.
PamelaDecember 29, 2019 at 1:15 pm
I had my second shingles shot the first week of November 2019. The injection site is still visible, itches and pain to touch all around the area. Sort of like it’s bruised under the skin. I have total body pain. I can only stand or walk for a few minutes before I feel exhausted. Even sitting can be painful. I see my provider for a physical in about 2 weeks and plan to go over this with her.
iviDecember 18, 2019 at 1:50 am
My first Shingrix shot was this Monday Dec 16. Within few hours I felt like getting cold or flu. At night I started to run fever, chills, today I had to stay at home because I felt sooooo bad, weak, everything hurts, joints, throat, back, nose is running, just horrible. Advil seems to help a bit but who wants to live on Advil. This vaccine should not have been approved with such side effects. [Show More]My first Shingrix shot was this Monday Dec 16. Within few hours I felt like getting cold or flu. At night I started to run fever, chills, today I had to stay at home because I felt sooooo bad, weak, everything hurts, joints, throat, back, nose is running, just horrible. Advil seems to help a bit but who wants to live on Advil. This vaccine should not have been approved with such side effects. I am going to report it to CDC. This is not right.
SusanDecember 16, 2019 at 8:28 pm
I had the first shingles vaccine in May 2019 and from the moment the shot touched my skin I was in pain. My arms was immediately sore and I suffered flu like symptoms for two days. Immediately after and to this day, 7 months later I have experienced an array of issues including digestive issues, throught issues, headaches and fatigue. Blood work all comes back fine but I have not been right sin[Show More]I had the first shingles vaccine in May 2019 and from the moment the shot touched my skin I was in pain. My arms was immediately sore and I suffered flu like symptoms for two days. Immediately after and to this day, 7 months later I have experienced an array of issues including digestive issues, throught issues, headaches and fatigue. Blood work all comes back fine but I have not been right since.
LisaDecember 7, 2019 at 5:25 am
I had the first Shingrix Shot November 2018 and I am still suffering from Trigeminal Neuralgia and many other debilitating symptoms. Right after receiving the Shingrix shot I could not use my arm due to severe pain and the next day I had fever and flu like symptoms. I could barely walk or function at all. Within the next few days I started getting severe headaches around my eye area and the righ[Show More]I had the first Shingrix Shot November 2018 and I am still suffering from Trigeminal Neuralgia and many other debilitating symptoms. Right after receiving the Shingrix shot I could not use my arm due to severe pain and the next day I had fever and flu like symptoms. I could barely walk or function at all. Within the next few days I started getting severe headaches around my eye area and the right side of my face went completely numb. My mouth constantly aches and feels like I have several absessed teeth where I don't have any teeth at all. My face is somewhat deformed and I have eye issues along with shocks and tingling in my face. In addition, my lower back is worse then ever and I can barely move my arms without pain. It is impossible tctively and am afraid I will no longer be employed. I did not have any of these symptoms prior to receiving the Shingrix shot and the pain has been constant for over a year now.......I have tried medications, acupuncture, and pain relief creams all of which have not helped. After reading some of the reviews on the internet, I wonder if I contracted internal Shingles after getting the shot however I have had little medical help in this area....I am not sure how much longer I can deal with this debilitating pain all over my body.........PLEASE HELP!!!o sleep at night and I am constantly fatigued and extremely DEPRESSED. I have not been able to do my job effe
DavidDecember 1, 2019 at 7:45 pm
I got 2 Shingrix shots. 1st was early June 2019 and 2nd Sept 2019. I have had left shoulder pain since June. I've been in physical therapy off and on since late June. I'm getting an MRI mid December 2019. The only thing I can think of for a history of this issue is the shot. I can barely lift my arm after a certain elevation. Range of motion is very limited. I've brought the shot up to providers a[Show More]I got 2 Shingrix shots. 1st was early June 2019 and 2nd Sept 2019. I have had left shoulder pain since June. I've been in physical therapy off and on since late June. I'm getting an MRI mid December 2019. The only thing I can think of for a history of this issue is the shot. I can barely lift my arm after a certain elevation. Range of motion is very limited. I've brought the shot up to providers as a possible source but nobody is really considering it yet.
BobNovember 15, 2019 at 5:31 pm
I’ve had severe rheumatoid arthritis since 2015. Dr doom told me to get my shingles shots since my blood tests showed me in near remission. First shot was 8/1/19. Injection site was very sore for a couple weeks and flu like symptoms. Got second shot 10/9/19. Next day have had bad flu symptoms to this day. My whole body started aching, loud ringing in ears, all my joints are in severe pain, can bar[Show More]I’ve had severe rheumatoid arthritis since 2015. Dr doom told me to get my shingles shots since my blood tests showed me in near remission. First shot was 8/1/19. Injection site was very sore for a couple weeks and flu like symptoms. Got second shot 10/9/19. Next day have had bad flu symptoms to this day. My whole body started aching, loud ringing in ears, all my joints are in severe pain, can barely walk now, rectal bleeding, very dry mouth and sore throat. RA and pain meds no longer affective. Set up an appointment with dr. doom, my rheumatologist. All he could do is report it to the Shingrix co. Took a complete blood count. All RA and inflammation markers are back in the upper red zone. WBC is back over 13. The only time I’m not in pain is if I’m sitting perfectly still. Wish I would have never got the injections. Life sucks now.
JohnNovember 1, 2019 at 10:42 pm
I received the first dose of Shingrix on July 2nd, 2019. After severe earache in August 2019, I was diagnosed with Ramsay Hunt Syndrome due to contracting shingles near my face and ear nerve. I had palsy on right side of my face, severe mouth drupe and could not shut right eye. Hearing in my right ear is reduced by half with severe ringing. Persistent vertigo prevents me from driving, now two mon[Show More]I received the first dose of Shingrix on July 2nd, 2019. After severe earache in August 2019, I was diagnosed with Ramsay Hunt Syndrome due to contracting shingles near my face and ear nerve. I had palsy on right side of my face, severe mouth drupe and could not shut right eye. Hearing in my right ear is reduced by half with severe ringing. Persistent vertigo prevents me from driving, now two months after the fact. Unreal. 54 years old.
MicheleOctober 29, 2019 at 4:04 pm
I received my first shot around 5 weeks ago. My arm was killing me for over a week. I too had flu like symptoms and now for the first time have developed sciatica nerve pain which was so bad I have to see a spine doctor to receive shots. The first round only worked for two days. Went back yesterday for round two if spinal shots and only a little better. Have to go for round three in two weeks and[Show More]I received my first shot around 5 weeks ago. My arm was killing me for over a week. I too had flu like symptoms and now for the first time have developed sciatica nerve pain which was so bad I have to see a spine doctor to receive shots. The first round only worked for two days. Went back yesterday for round two if spinal shots and only a little better. Have to go for round three in two weeks and I can tell you this that the booster shingles shot is never going to happen.
StephanyOctober 29, 2019 at 4:11 am
I got the first Shingrix vaccine in Aug. 2019. It is just a few days before Halloween and my shoulder still aches and is still very swollen. Seeing my doctor about this in a few days.
GregOctober 17, 2019 at 12:31 am
I got my shingrix vaccine October 10th 2018 same time I was going through dental work assumed it was related to that in the 10 months since then I've been to a neurologist and neurosurgeon acupuncture acupressure General Practitioners MRI MRI with contrast all told normal friend of mine told me she was getting ready to go get a vaccine but was afraid of the side effects I asked her what they were [Show More]I got my shingrix vaccine October 10th 2018 same time I was going through dental work assumed it was related to that in the 10 months since then I've been to a neurologist and neurosurgeon acupuncture acupressure General Practitioners MRI MRI with contrast all told normal friend of mine told me she was getting ready to go get a vaccine but was afraid of the side effects I asked her what they were and she read them off and it was exactly what I have numbness left side of face lips tip of my nose to my ear all on the left side taste buds can't taste anything I only got the one vaccine thank God I didn't get the other... I was told that these side effects or indicative of the first type of shingles vaccine and not the new 1 BS
KimOctober 11, 2019 at 10:52 pm
I recieved my second shot of Shingrix in September 2019. A day after I developed severe neurological problems that lasted a week. I'm so glad no permanent damage was done. And so glad that was my second shot!!
HerschelOctober 10, 2019 at 12:07 pm
I received the shingles vaccine on August 12, 2019 and loss my hearing out of my left ear. I went to a ear specialist he said I have nerve damage to the inter ear cause deaf in that ear. Both ears were perfectly normal before the vaccine, I’m 63yrs old. I will not get the 2nd dose not worth loosing my other ear. I am blessed that I can still work and drive without issues. Very concerned why this h[Show More]I received the shingles vaccine on August 12, 2019 and loss my hearing out of my left ear. I went to a ear specialist he said I have nerve damage to the inter ear cause deaf in that ear. Both ears were perfectly normal before the vaccine, I’m 63yrs old. I will not get the 2nd dose not worth loosing my other ear. I am blessed that I can still work and drive without issues. Very concerned why this happened to me. Concentration is very hard because the ringing noises that continues around clock. Learning to blockout the noise through other wind devices while sleeping.
MichelleSeptember 29, 2019 at 5:14 am
I.got the shringix vaccine on 9-19-19 broke out with severe shingles on 9-21-19. I was feeling like I was hit by a truck on 9-20-19 The virus is still running its course. I am in Hell.
MichaelSeptember 21, 2019 at 2:44 pm
I had a severe ear infection that lasted about 4 weeks. It was both bacterial and fungal. When the infection cleared up I had an audio test done to determine what type of hearing aids we were going to use. We did the examination and I made an appointment for 2 weeks later to get fitted. Everything was fine and then one week before I was to be fitted I took the shingles shot. Within 5 days my heari[Show More]I had a severe ear infection that lasted about 4 weeks. It was both bacterial and fungal. When the infection cleared up I had an audio test done to determine what type of hearing aids we were going to use. We did the examination and I made an appointment for 2 weeks later to get fitted. Everything was fine and then one week before I was to be fitted I took the shingles shot. Within 5 days my hearing deteriorated so much that they could no longer fit me for hearing aids until the severity of my hearing loss could be determined. I have taken steroids prescribed by my ENT to help regain some of my hearing but they are saying I will not regain what I had lost.
MaeSeptember 8, 2019 at 1:34 am
I received the Shingrix vaccine at North Seminole Family Practice & Sports Medicine on September 6, 2019. Within 24 hours the entire left side of my body felt like it was on fire, and my right middle toe was completely numb. I had to get blood drawn today at Quest labs, and i asked the phlebotomist to look at it and she told me to IMMEDIATELY go to the Florida Blue clinic to have them examine my l[Show More]I received the Shingrix vaccine at North Seminole Family Practice & Sports Medicine on September 6, 2019. Within 24 hours the entire left side of my body felt like it was on fire, and my right middle toe was completely numb. I had to get blood drawn today at Quest labs, and i asked the phlebotomist to look at it and she told me to IMMEDIATELY go to the Florida Blue clinic to have them examine my left arm. When the Nurse Practitioner examined me, she immediately notice the Shingrix vaccine was administered entirely too low on my left arm. She went on to tell me it should've been given in the upper deltoid area. The nurse practitioner informed me to follow up with my PCP, to see if I should get the second dose. If I need to to have it administered by my PCP ONLY AND NO ONE ELSE AT THAT OFFICE. I don't want to receive the second dosage EVER. I have been extremely fatigued and my whole left side of my body feels like lava is coursing through my veins. This pain is REALLY SERIOUS...OH MY GOD
AlliAugust 9, 2019 at 12:27 pm
I received the Shingrix vaccine at Walmart and it has caused severe pain in the top of my left arm. The first several weeks I could not lift my arm. It's been three weeks and it is still very painful and I can barely lift my left arm to brush my hair. It throbs during the night. I will not got the second shot. I hope my arm gets better.
CarolJuly 29, 2019 at 12:31 am
Caroline and Les ... I would like to chat with you about your experience. I also had a quick onset of sciatica after my second shot. Flu like symptoms day 2 after shot.. horrible sciatica day 3 and onward. I am finally nerve pain free, but have muscle cramps and tightness and pain in the same leg I had the sciatica. I received the second shot on June 7 ... so it’s been 7 weeks. If you are willing [Show More]Caroline and Les ... I would like to chat with you about your experience. I also had a quick onset of sciatica after my second shot. Flu like symptoms day 2 after shot.. horrible sciatica day 3 and onward. I am finally nerve pain free, but have muscle cramps and tightness and pain in the same leg I had the sciatica. I received the second shot on June 7 ... so it’s been 7 weeks. If you are willing I would love to contact you by phone or email.
CarolineJuly 13, 2019 at 2:28 pm
Can you update me on your sciatica problems. I also had a quick onset after my second vaccine. Wondering how long this will last and how it will go away abruptly. This has been awful
GenevaJune 24, 2019 at 8:51 pm
I got the Shingrix shot on July 5, 2018 at Walgreens. Had a horrible reaction to the sho t including swelling and redness, severe pain, flu-like symptoms and a headache for days. Could not use my left arm. Had continuous severe pain and still have pain in the injection site to this day. Cannot use that arm. I have pictures of my arm showing the reaction I had. I have experienced severe f[Show More]I got the Shingrix shot on July 5, 2018 at Walgreens. Had a horrible reaction to the sho t including swelling and redness, severe pain, flu-like symptoms and a headache for days. Could not use my left arm. Had continuous severe pain and still have pain in the injection site to this day. Cannot use that arm. I have pictures of my arm showing the reaction I had. I have experienced severe fatigue ever since. I am 72 years old. Could not use that arm after injection and still cannot. Has caused blurred vision. I had severe headache for days after the injection. I called Walgreens and reported the horrible reaction. They said they would report it and for me not to get the second shot. No way I was going to take that second shot. It has ruined my life..
TammyJune 22, 2019 at 6:27 pm
I received my first dose on Nov. 5th 2018. 6 months to that day I was diagnosed with Bells Palsy. How did I ever get this if the vaccine worked?!? Thank God the pharmacy was out of the second dose whenever I went back. I will not be getting the 2nd dose, and truly believe that the reason I got Bells Palsy is directly related to the vaccine. I am already out about $1500.00 and have an appt. wi[Show More]I received my first dose on Nov. 5th 2018. 6 months to that day I was diagnosed with Bells Palsy. How did I ever get this if the vaccine worked?!? Thank God the pharmacy was out of the second dose whenever I went back. I will not be getting the 2nd dose, and truly believe that the reason I got Bells Palsy is directly related to the vaccine. I am already out about $1500.00 and have an appt. with an EMT to consult for surgery. I feel compensation should be mandatory!!!
JulieJune 18, 2019 at 11:09 pm
My husband Troy got the shingrix vaccine for shipping gels and 3 wks later got shingles which doctors said also triggered Gout in his left arm and hand
DeannaApril 30, 2019 at 6:04 pm
I received my first shingrix shot in July 3018. Immediately after I got shingles in my pelvic area with horrible nerve pain. I was hospitalized to control the pain...
LesApril 28, 2019 at 9:59 pm
April 24,2019 I received round 1 of Shingrix. Within 24 hours I developed severe flu like symptoms and my Sciatica flared to a 10 level of pain. I had Sciatica prior to the injection but it had been reduced down to a 2 or 3 level of pain. I have read on another site the comments of 3 others that developed Sciatica from Shingrixx.
TerryApril 22, 2019 at 2:42 pm
I received the Shingrix vaccine in August of 2018. I broke out with a rash of hives 2-3 weeks later. I went to a Dermatoligist and she said it looked like shingles and took a bi-opsy witch came back negative for shingles and said I dont know maybe just a fluke. My pereferal vision has developed worse to almost nothing, hearing is worse. I have had lung and breathing problems since the vaccine. Aft[Show More]I received the Shingrix vaccine in August of 2018. I broke out with a rash of hives 2-3 weeks later. I went to a Dermatoligist and she said it looked like shingles and took a bi-opsy witch came back negative for shingles and said I dont know maybe just a fluke. My pereferal vision has developed worse to almost nothing, hearing is worse. I have had lung and breathing problems since the vaccine. After a doctor visit he advised me that the booster could be deadly and I had all ready told him I was not going to get the booster. I have had to take disability from my aviation carrier. I am 59years old.
PamMarch 20, 2019 at 12:56 am
Received my 1st dose Shingrix shot February 1st I have been suffering with some palsy effects on left side of face. Painful headaches and feeling exhausted. Went back to Drs and put on predizone. Nothing changed. Seeing a nureologist . I could cry that I agreed to this shot. I should of done my research.. I pray I get my once good health back being only 57 ...
DaraMarch 17, 2019 at 12:11 am
I received my first Shingrix shot 6/8/18, subcutaneous in my right arm. Swelling, pain, tenderness and rash on entire arm that lasted 3 weeks. Was told the reaction was because the shot was given incorrectly and I shouldn't have a problem with dose 2. Received dose 2 3/13/19, intramuscular in my left arm. Extreme pain in arm, headache that lasted 48 hours, fever, and now rash that covers half[Show More]I received my first Shingrix shot 6/8/18, subcutaneous in my right arm. Swelling, pain, tenderness and rash on entire arm that lasted 3 weeks. Was told the reaction was because the shot was given incorrectly and I shouldn't have a problem with dose 2. Received dose 2 3/13/19, intramuscular in my left arm. Extreme pain in arm, headache that lasted 48 hours, fever, and now rash that covers half of my upper arm.
GeorgeJanuary 20, 2019 at 1:06 am
I am a 71 year old white male blind in my right eye due to a childhood injury. I received the 1st Shingrix Vial injection on 04/12/2018 at CVS. I was diagnosed by my eye Dr Snead with Macular Degeneration in my left eye on 07/24/2018. I was referred to my retina Dr Ghuma on 07/31/2018 for treatment that is still going on. I received my 2nd Shringrix Vial injection on 08/03/2018 at Publix.
JimJanuary 14, 2019 at 8:15 am
I took a chance and got this shot and I am really sorry I did. Since then my glucose has been impossible to control, white cell and lymph count shot up, extreme and long periods of fatigue. The bicep pain lasted several days and soreness lasted close to a month. This shot may end up being the death of me. GlaxoSmithKline is an evil corporation with history of criminal convictions of lying about th[Show More]I took a chance and got this shot and I am really sorry I did. Since then my glucose has been impossible to control, white cell and lymph count shot up, extreme and long periods of fatigue. The bicep pain lasted several days and soreness lasted close to a month. This shot may end up being the death of me. GlaxoSmithKline is an evil corporation with history of criminal convictions of lying about the effectiveness of their drugs and their horrible side effects including death. The hyped-up trials GSK paid-for finding 95% effectiveness are probably just pure BS. And lots of people complaining on Internet about serious side effects of Shingrix. Just stick with Zostavax.
CarolynNovember 27, 2018 at 6:55 pm
I received the Shingrix both of the recommended dose. I have shingles worse now than i have had them in 30 yrs. Miserable. And my vision is blurry. I'm not sure if I would get it again. And now i can't take my meds for Rheumatoid Arthritis.
BettySeptember 9, 2018 at 11:32 pm
I had my second SHINGRIX shot and immediately became ill. I was hospitalized with upper lobe pneumonia and severe bronchitis. I’m on my third week and am still ill.