Infant Drug-Related ER Visits Dropped by Half After Cold Medicine Recalls
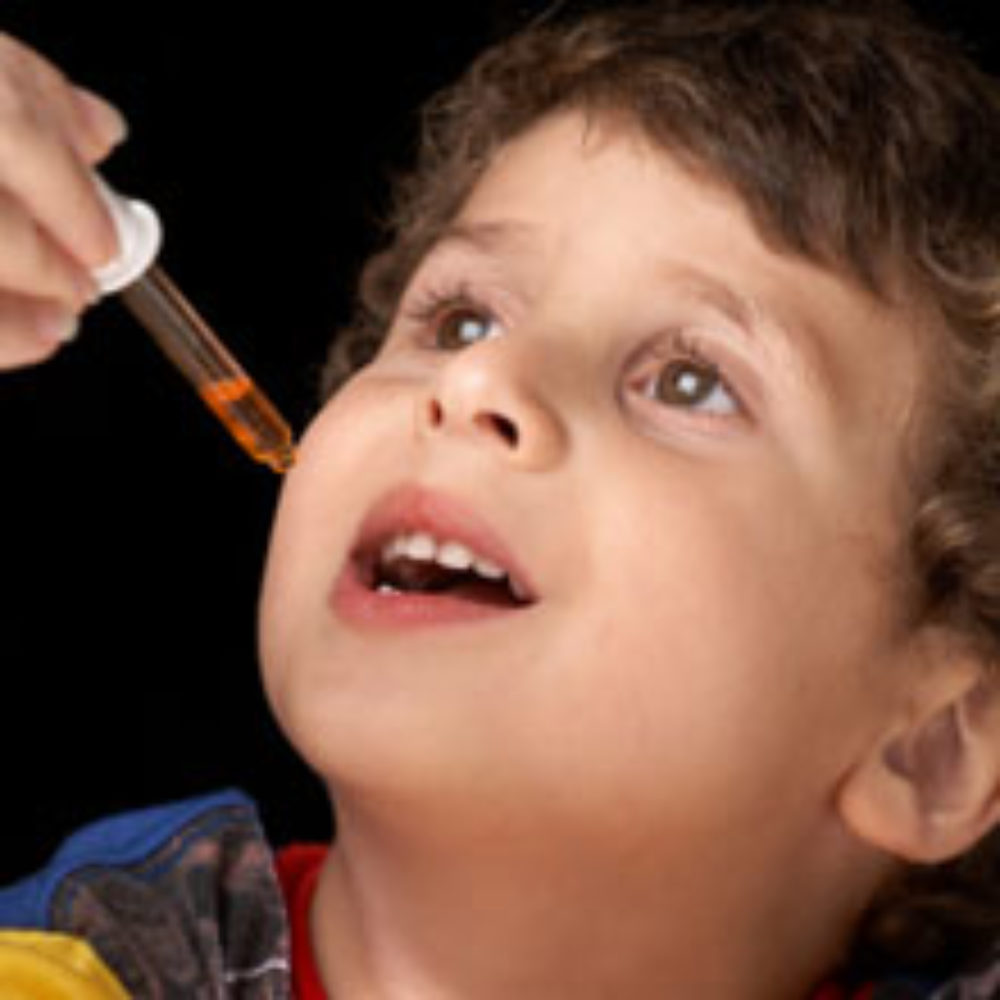
Government researchers report that when drug makers removed infant cold medicine from the market, the number of emergency room visits by children under 2 years old dropped by more than half.
The report, from researchers at the U.S. Centers for Disease Control and Prevention (CDC), was published online in the medical journal Pediatrics. Researchers found that two-thirds of emergency room visits involving children under the age of 12 were due to children taking medications they were not supposed to take.
In 2007, growing health concerns over infant cold and cough medications resulted in drug manufacturers agreeing to pull many over-the-counter cold medicines for children under two years old off the market. They later expanded the market withdrawal to cold and cough medications for children four and under.

Did You Know?
Millions of Philips CPAP Machines Recalled
Philips DreamStation, CPAP and BiPAP machines sold in recent years may pose a risk of cancer, lung damage and other injuries.
Learn MoreCDC researchers looked at admissions in 63 emergency departments across the U.S. in the 14 months following the market withdrawal and compared those numbers to the premarket admissions. They found that the number of infants admitted to the emergency room for cold and cough medicine-related adverse events dropped from 2,790 in the preceding 14 months, to 1,248 in the months that followed. The numbers for older children remained unchanged.
The FDA has found that many cold and cough medications can negatively impact children’s still developing respiratory systems, even when administered correctly. The agency has recommended that parents not give babies and toddlers any kind of cold and cough medicine.
Medical experts advise parents dealing with cold-stricken infants to use saline sprays and bulb syringes to fight congestion, consider the use of a humidifier, and to make sure the child drinks plenty of fluids. Children running a high fever can be given Tylenol, however pediatricians recommend that low fevers be left alone, since that is the body’s defense mechanism for preventing invading bacteria from replicating.
The study was published as Johnson & Johnson announced another recall of 4 million packages of children’s Benadryl and 800,000 bottles of junior-strength Motrin. The company says that the recalled drugs carry no health risk and can be used safely, and are not asking consumers to return them. However, they are pulling the remaining product off store shelves due to a failure of the products to meet manufacturing standards.
The recall affects all children’s Benadryl Allergy FASTMELT tablets in cherry and grape flavors, and all Junior Strength Motrin 24-count caplets. The recall comes as part of an ongoing review of McNeil Consumer Healthcare’s manufacturing processes. McNeil is a subsidiary of Johnson & Johnson.
McNeil and Johnson & Johnson have been under scrutiny since a massive recall on April 30 that affected 40 different liquid medication products. The recall affected 136 million bottles of children’s medications, and resulted in the shutdown of the company’s Ft. Washington, Pennsylvania, plant, and the suspension of the production of all of McNeil’s children medications. Following that recall, the FDA has received nearly 800 complaints, including at least seven reports of deaths associated with the medication. However, the FDA says its investigations so far have not directly linked any of the recalled products to any of the deaths.
Get more articles like this sent directly to your inbox.
"*" indicates required fields




0 Comments