Stryker Hip Replacement Metal Blood Poisoning Lawsuit Filed Over Accolade, LFit v40 Components
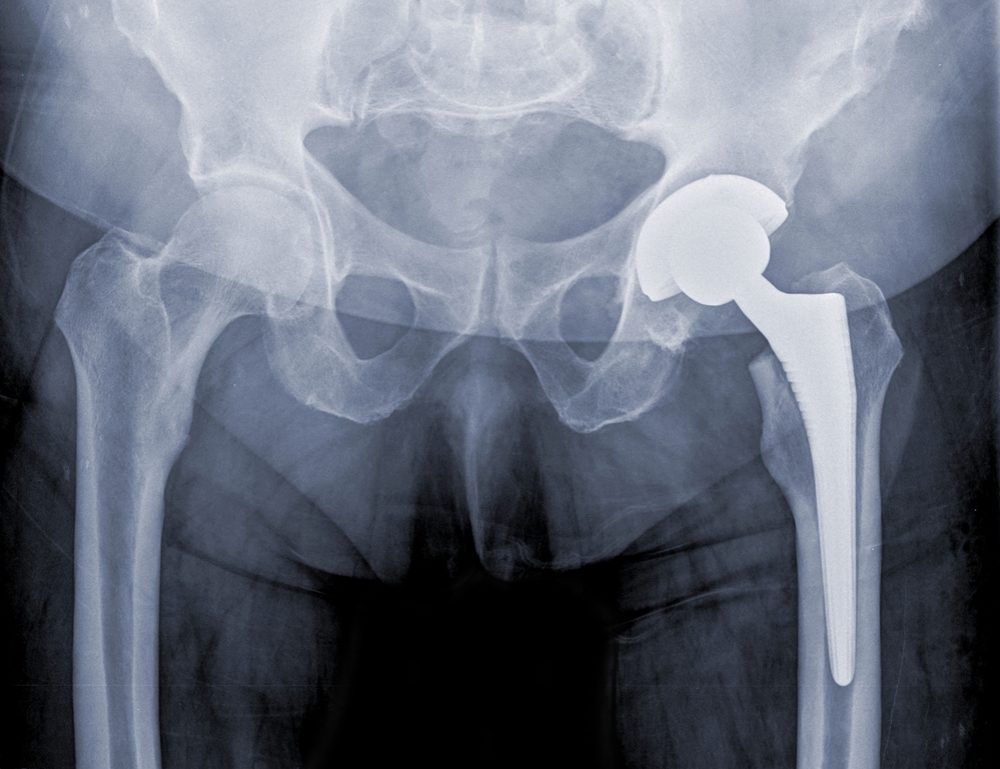
According to allegations raised in a product liability lawsuit filed by a woman who experienced problems with a Stryker metal hip, design defects with the Accolade hip stem and LFit v40 femoral head caused debris to be released into her body, resulting in metal blood poisoning and failure of the hip replacement less than ten years after it was implanted.
The complaint (PDF) was filed by Linda Regan in the U.S. District Court for the District of Massachusetts on February 22, indicating that Howmedica Osteonics Corp., doing business as Stryker Orthopaedics, manufactured and sold hip replacement components that were prone to release metal ions into patients’ blood. As a result, users face a high risk of catastrophic failures, often resulting in the need for risk hip revision surgery, according to the lawsuit.
Regan underwent hip replacement surgery in July 2007, at which time a Stryker Accolade TMZF Hip Stem was implanted together with a Stryker LFit Anatomic v40 Femoral head, which was ultimately recalled last year amid reports of metal blood poisoning problems.

Learn More About
Lawsuits are being reviewed for several different dangerous and defective hip replacement systems.
Learn More About this Lawsuit See If You Qualify For CompensationAfter the Stryker metal hip components were placed into her body, Regan indicates that she began to experience pain and discomfort. Diagnostic workup failed to find any evidence of hip loosening, infection, malposition or other explanation for her symptoms, according to the complaint. However, it was subsequently discovered that there were “markedly increased levels of metal ions” in Regan’s blood.
As a result of the Stryker hip replacement metal blood poisoning, and continued worsening of her symptoms, Regan underwent hip revision surgery in July 2014, at which point it was noted by her surgeons that there was significant metallosis and trunnionosis in her hip, resulting in soft tissue damage. The doctor also found that there was corrosion at the taper junction between the Stryker Accolade hip stem and Stryker L-Fit v40 chromium and cobalt femoral head.
Due to similar reports of problems, a Stryker LFit v40 hip recall was issued by the manufacturer last year, impacting certain large-diameter femoral heads sold before 2011. At that time, Stryker acknowledged that a higher-then-expected number of individuals were experiencing problems with trunnion failure, metal wear, adverse tissue reactions and other complications.
“Plaintiff is informed and believes, and thereon alleges, that the problems with the V40 head/Accolade Stem combinations are much larger than those referenced in the [Stryker recall notice] and are caused by the metallurgical formulation used to manufacture the TMZF Accolade Stem and/or the geometry of the Cobalt/Chromium V40 Head,” according to the complaint filed by Regan. “Plaintiff alleges that all V40 heads sold by defendants are defective and should recalled. Instead the Stryker defendants have intentionally downplayed the risk of harm and limited the scope of its recall in an effort to hide from surgeons, patients and the FDA the true extent of the problems with their defective hips.”
The case joins a growing number of similar Stryker Accolade hip replacement lawsuits filed in recent months over metal blood poisoning problems.
Although plaintiffs indicate that a simple, inexpensive blood test could be used to help determine whether individuals are experiencing the corrosive process that leads to metal poisoning and the recent recall, specifically the presence of elevated levels of cobalt, chromium or titanium in the blood, lawsuits indicate that Stryker failed to provide adequate warnings or instructions to surgeons.
As a result, hip replacement lawyers are not only pursuing similar cases for individuals who have required revision surgery, but are also reviewing claims for individuals with recalled Stryker hip components in their body, which may be releasing toxic levels of metal without any knowledge that their hip replacement is corroding.
Get more articles like this sent directly to your inbox.
"*" indicates required fields
2 Comments






JessiMay 3, 2017 at 5:40 am
My mother had two artificial hips. She got the first hip replaced in 1999, and the second hip replaced in 2003 (ish). She also had to have revision surgery on one of these, as at one point it became dislocated. She wound up suffering a great deal of pain in this region of her body (hips/back/legs) throughout the rest of her life. In 2015, my mom died two weeks after undergoing aortic valve replac[Show More]My mother had two artificial hips. She got the first hip replaced in 1999, and the second hip replaced in 2003 (ish). She also had to have revision surgery on one of these, as at one point it became dislocated. She wound up suffering a great deal of pain in this region of her body (hips/back/legs) throughout the rest of her life. In 2015, my mom died two weeks after undergoing aortic valve replacement surgery. She was only 67, in relatively good health (normal weight, no smoking, no drinking, no drugs, low blood pressure, etc) and had not been considered a 'high risk' patient. The surgeon said that in 30 years of performing the procedure, he had never encountered the kind of post-surgical symptoms like my mother exhibited, including extreme delirium, hypertrophy, joint pain, and muscle weakness. The autopsy report concluded that her death was not the result of surgical error, with the cause of death ultimately being attributed to 'multi-organ system failure'. Eventually, hoping to figure out what went wrong, and why my mom died, I collected a sample from her hairbrush, and sent it to a toxicology lab. What they discovered was so shocking that the lab receptionist told me that she could not release the test results to me until she consulted with an attorney, as the findings went 'significantly beyond normal parameters'. In total, 21 heavy metal poisons were detected in my mother's hair sample - eight of which were as much as SIX TIMES ABOVE the 'HIGH LEVEL' demarcation. Of all the toxic metals detected in her hair sample - CHROMIUM was among the highest. Cobalt was there as well, but at less dangerous levels (although I have since come to understand that toxins combine to have a 'synergistic' effect on the body). Although her body was cremated, I called the pathology lab where her body was autopsied and asked that they put a 'hold' on my mom's tissue samples pending investigation, and they said that they would do so. (As her husband - rather than myself - is listed as my mother's 'next of kin' in the hospital records, I was told that I would need a court injunction in order to get the samples tested). Unfortunately, the doctors conducting the autopsy were apparently solely concerned with the condition of the internal organs. The report makes no mention of her hips - or the two metal implants - at all. ( As the saline implants in her breasts ARE listed - this leads me to think that, rather than conducting a thorough examination, the pathologist(s) performing the autopsy probably figured that limited investigation would suffice). After a great deal of research on both the autopsy findings, and the toxicology results, and strongly believe that my mother's post-surgical symptoms, and eventual death, were the result of chronic heavy metal intoxication. Many of the findings in the autopsy report not only include symptoms unassociated with heart disease, but in some cases are in word-for-word alignment with descriptions of typical poison morphologies. I don't know what - if anything - can be done about any of this - but I do know that the poison found in my mother's hair sample came from somewhere. As heavy metal toxins are initially subtle and bioaccumulate over time, those exposed to such poisons rarely ever realize to what extent their health has been compromised. Should one of you attorneys out there know of some way I might go about getting this matter more thoroughly investigated - I am willing to consider any suggestions you may have. Thanks Jess
howardMarch 7, 2017 at 1:35 am
pain in left hip ,and lower back. dr natazerian did replacement march 3rd 2008. it was done at penna hospital (3Bs)