FDA Issues New Draft Guidance on Children’s Use in Clinical Trials
Many drugs and medical procedures used on children have never been tested on children in clinical trials for safety and informed consent reasons.
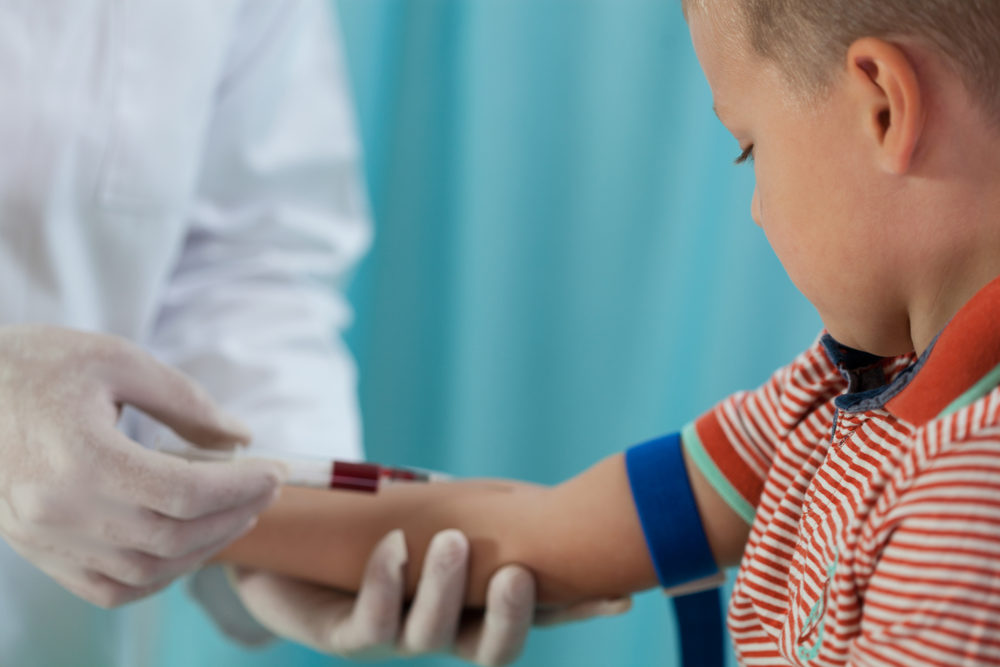
Federal drug regulators have issued new guidelines for including children in clinical trials, which seeks to balance between safety considerations and the need to include minors in human studies, so that safe and effective dosing and warnings can be provided for drugs and devices.
Historically, children have not been included in clinical trials, because researchers believed excluding them was the best way to protect them, since they could not offer consent and some drugs or devices may have harmful side effects. However, this has resulted in the approval of many medications and medical devices that are widely used among children, without any testing or pediatric-specific label information.
The FDA issued the new draft guidance to address this problem last week, titled “Ethical Considerations for Clinical Investigations of Medical Products Involving Children,” which provides guidelines on ethical considerations for including children in clinical trials and protecting them from unanticipated side effects.
“Children need access to safe and effective medical products and health care professionals need data to make evidence-based decisions when treating children,” Dr. Dionna Green, director of the FDA’s Office of Pediatric Therapeutics said in a press release. “However, children are a vulnerable population who can’t provide consent for themselves and are afforded additional safeguards when participating in a clinical investigation.”
The new guidance, if finalized, is intended to assist industry, sponsors and institutional review boards when enrolling children in clinical investigations of drugs, biological products and medical devices.

Did You Know?
Millions of Philips CPAP Machines Recalled
Philips DreamStation, CPAP and BiPAP machines sold in recent years may pose a risk of cancer, lung damage and other injuries.
Learn MoreThe report outlines an ethical framework for including children in research and protecting them. It also highlights concepts that should be considered when conducting clinical trials that include children, including:
- Whether including children in a clinical trial is scientifically necessary.
- How to weigh risks for interventions and procedures that will not directly benefit the child.
- How to evaluate whether an intervention or procedure will directly benefit the child.
- Assessing the risks and benefits of interventions and procedures that directly benefit a child.
- Analysis of the risks of components of interventions and procedures
- Potential for regulatory review of research not approved by an institutional review board.
- Parental or guardian permission and child consent.
The guidance was developed by the FDA’s Office of Pediatric Therapeutics with contributions from the Center for Drug Evaluation and Research, the Center for Biologics Evaluation and Research and the Center for Devices and Radiological Health.
The FDA is accepting comments from the public on the draft guidance until December 27. See the Federal Register notice for details on how to submit comments.
Get more articles like this sent directly to your inbox.
"*" indicates required fields






0 Comments