DePuy Attune Knee Problems Linked To “Unusual” Number of Tibial Loosening Reports: Study
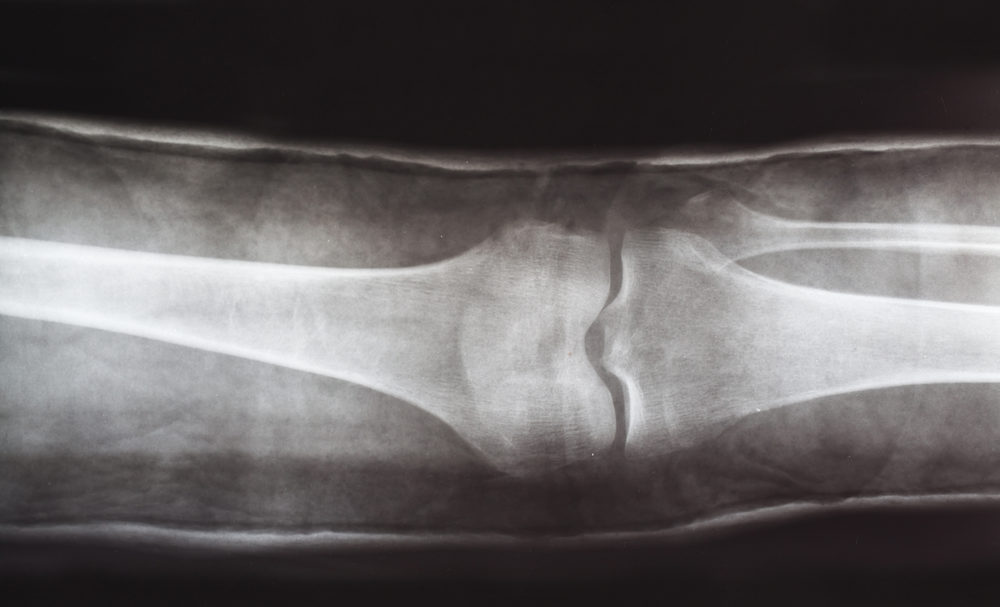
Knee replacement patients are experiencing a surprisingly high number of adverse event reports involving tibial loosening and debonding problems with the DePuy Attune knee system, according to new research.
In a study published recently in the Journal of Knee Surgery, researchers warns that the FDA is receiving an “unusual” number of reports in its Manufacturer and User Facility Device Experience (MAUDE) database involving DePuy Attune knee problems, and that a review of incidents at three hospital centers also identified a high number of asceptic early failures.
Researchers with the Bonutti Clinic in Illinois, and Sinai Hospital in Baltimore, Maryland, reviewed three hospital databases for patients who suffered tibial component loosening at the implant-cement interface, and also reviewed the FDA’s MAUDE database for similar cases.
Learn More About
Design problems with several types of knee implants have resulted in lawsuits for individuals who experienced painful complications.
Learn More About this Lawsuit See If You Qualify For CompensationThe study identified at least 13 patients at the three facilities, involving 15 knee replacements, each resulting in tibial loosening failures with the DePuy Attune knee. Researchers found dozens of similar cases on the MAUDE database, warning that the number of problems in the database were probably underreported, meaning many more incidents are likely undocumented.
The DePuy Synthes Attune Knee system was approved by the FDA in 2010, and the manufacturer has boasted about the system’s 1.3% revision rate over four years, which is below the 1.9% revision rate average for knee replacement systems. However, this year has seen a spike in reports of tibial loosening and failures, with the FDA receiving 21 reports in just a two-month period, according to researchers who now suggest there appear to be signs of a problem.
“In our experience of three community hospital centers, we have encountered unusually high numbers of the aseptic early failures. In addition, many reports have been filed in MAUDE database concerning tibial loosening at the implant–cement interface,” the researchers determined. “Numerous other tibial failures were reported, how-ever, the mechanisms of failures were not specified.”
Researchers found that the reports they were able to review contained similar mechanisms of knee replacement failure. All of the cases reported in the study had a failure of the cement-to-implant interface in a tibial component. However, those failures did not show up on radiographic evaluation, which researchers said may be because the failures are linked to the debonding of the implant-cement interface, as opposed to a structural failure.
“Although we report on only 15 cases, we believe that this complication is markedly underreported due to failure of radiographs to accurately assess loosening,” the study concludes. “In addition, MAUDE database reporting is not consistent by the company representatives and competing companies cannot provide data on the revised components.”
Researchers recommended that when patients with a DePuy Attune knee implant present with unexplained pain, healthcare professionals should do a thorough workup on the joint and not rely solely on radiographic analysis.
In recent months, a number of product liability lawyers have also begun investigating the potential for DePuy Attune knee replacement lawsuits on behalf of individuals who have experienced complications.
Get more articles like this sent directly to your inbox.
"*" indicates required fields





0 Comments