Fentanyl Patch Deaths from Accidental Exposure Lead Required Changes
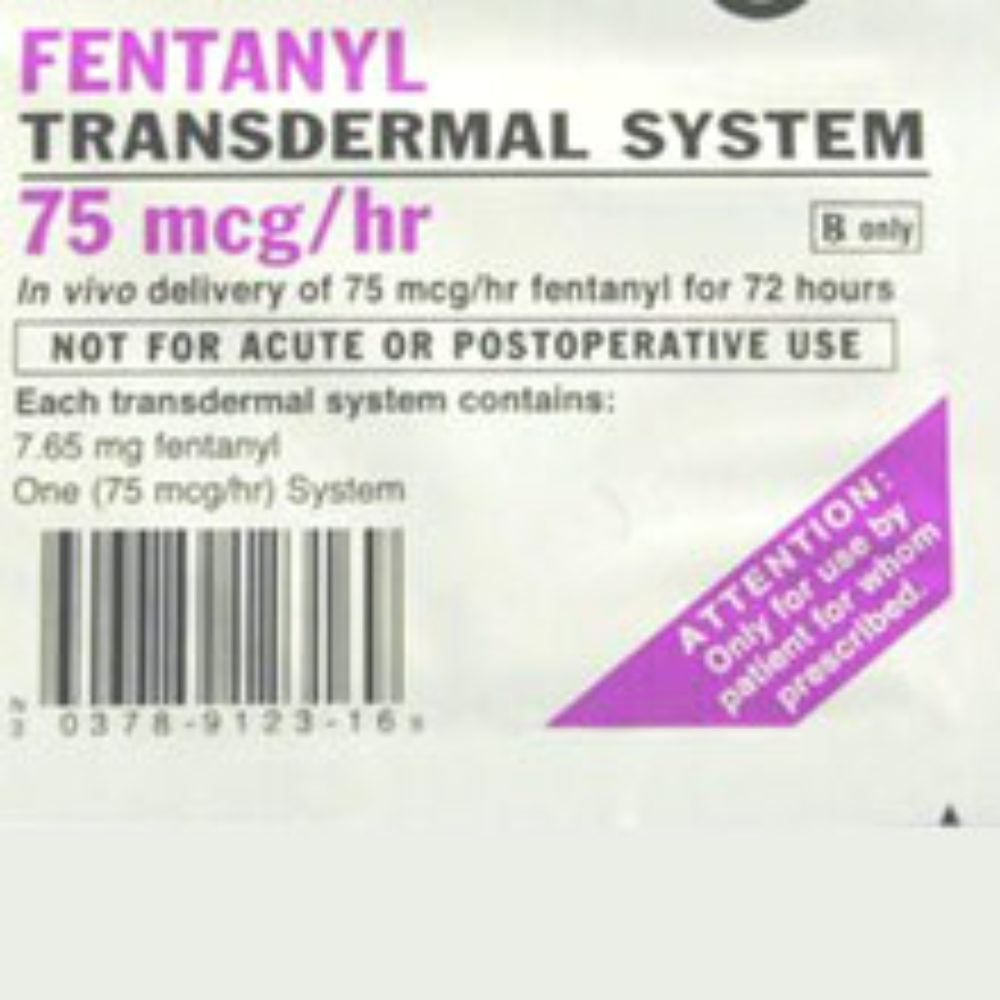
Following the deaths of several children from accidental exposure to fentanyl pain patches, the FDA is requiring more obvious warning labels and changes to the color of the Duragesic patch and other generic fentanyl patches.
On September 23, the FDA issued a drug safety communication announcing changes that will help emphasize that accidental exposure to used patches can cause death.
The FDA warns that the Duragesic and other fentanyl patches can still be deadly even after they are used and discarded, because of the high amounts of pain medicine still in them. If the fentanyl gel contained within the patch leaks out and comes in direct contact with the skin, it can cause a potentially fatal overdose.
The enhanced warnings come following the deaths of at least two children since the FDA’s last fentanyl patch warning in April 2012. One of the deaths involved improper disposal of the patch into the household trash, and the other fentanyl patch death resulted from the transfer of a patch from a parent to a child in close proximity, according to the FDA’s data.
Under the new requirements, patch manufacturers will put the name and strength of the drug on the patch in easily readable colors, made of long-lasting ink that will resist fading over time. The FDA is also warning consumers about proper disposal of the patches, indicating that they should be folded so that the adhesive sides stick together. The fentanyl patch should then be flushed down the toilet.
Child Fentanyl Deaths, Injuries
Between August 7, 1990, when the Duragesic patch was first approved, and April 16, 2012, the FDA is aware of at least 30 cases of pediatric exposure to fentanyl patches, 10 of which resulted in death. Of the cases, 28 involved children under the age of 10, with most being two years old or younger.
In April 2012, the U.S. Centers for Disease Control and Prevention (CDC) issued a report noting that the death rate among children due to prescription drug overdose has risen significantly over the last several years and accounted for 57% of all poisoning deaths of children between the ages of 15 and 19 as of 2009. That was up from 30% in 2000. The CDC found that as other causes of child death were on the decline, prescription drug overdoses were on the rise.
The fenatnyl patch was originally introduced as the brand name Duragesic pain patch, manufactured by a subsidiary of Johnson & Johnson. It is now widely available as a generic, and a number of drug companies make fentanyl pain patches, including Watson, ALZA and Mylan Pharmaceuticals.
The pain patch products contains powerful fentanyl gel, which is designed to be slowly delivered through the skin in a regulated manner. Fentanyl is a painkiller that is about 100 times more powerful than morphine.
Since it was introduced, the Duragesic patch has been plagued by a number of manufacturing problems, where some patches contained defects that allowed gel to leak out of the patch, posing a risk of fentanyl overdose.
The FDA has received hundreds of reports involving adverse events and deaths associated with the fentanyl patch, leading some critics to question whether they can be safely manufactured. A number of consumers have also filed fentanyl pain patch lawsuits as a result of injuries or the loss of loved ones.
In August 2012, all federal lawsuits involving the Watson fentanyl pain patch were consolidated for pretrial proceedings before U.S. District Judge Matthew F. Kennelly in the Northern District of Illinois. The U.S. Judicial Panel on Multidistrict Litigation refused to include lawsuits involving other patch manufacturers. Those lawsuits are proceeding as individual cases throughout the United States.
Get more articles like this sent directly to your inbox.
"*" indicates required fields




0 Comments