Belladonna Poisoning Risk from Hyland’s Teething Products Results in FDA Warning
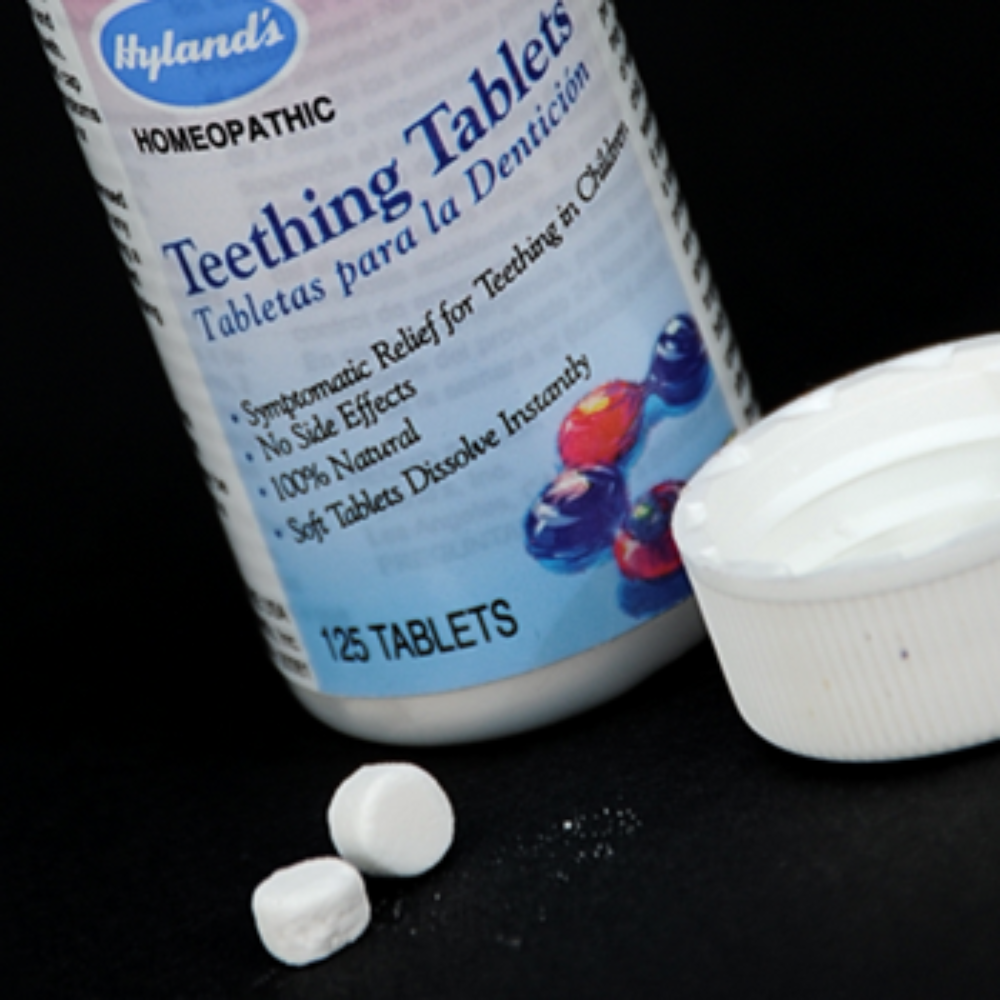
Federal health officials warn that Hyland’s homeopathic teething tablets have tested positive for excessive levels of belladonna, a toxic substance that could poison or kill infants.
The FDA issued a safety alert on January 27, indicating that infants face a risk of belladonna poisoning from Hyland’s teething products. The warning comes after the manufacturer failed to issue a voluntary recall for teething products that may still be in use.
The agency indicates that it is currently investigating at least 10 deaths of children who used teething tablets or gel products, as well as 400 adverse events reported over the last six years, which they suspect are associated with homeopathic teething that contain high levels of belladonna. So far, the deaths and adverse events have not been proven to be connected to the teething products .

Did You Know?
Millions of Philips CPAP Machines Recalled
Philips DreamStation, CPAP and BiPAP machines sold in recent years may pose a risk of cancer, lung damage and other injuries.
Learn MoreDespite the latest findings, the FDA indicates that Standard Homeopathic Company, the parent company of Hyland’s, has refused to conduct a voluntary recall at this time. However, three similar products by Raritan Pharmaceuticals were recalled last year, including two that were sold at CVS.
Belladonna is a chemical extracted from the berry plant Atropa belladonna, a perennial herbaceous plant in the Nightshade family Solanaceae. Belladonna extract has chemicals that can block functions of the body’s nervous system and has been used in the past in the make-up of ointments that are applied to the skin for joint pain or leg pain.
The use of the belladonna extract has been known to cause adverse health effects such as seizures, difficulty breathing, lethargy, excessive sleepiness, muscle weakness, skin flushing, constipation, difficulty urinating, and agitation.
“The body’s response to belladonna in children under two years of age is unpredictable and puts them at unnecessary risk,” Dr. Janet Woodcock, director of the FDA’s Center for Drug Evaluation and Research, said in a press release. ”We recommend that parents and caregivers not give these homeopathic teething tablets to children and seek advice from their health care professional for safe alternatives.”
The FDA noted that the teething products have not been evaluated or approved for any medical benefits, indicating that the manufacturer Standard Homeopathic Company illegally advertised these products to relieve teething symptoms in children.
The manufacturer has attempted to maintain that their homeopathic teething products are safe to use, but did discontinue sales in the U.S. late last year, after an earlier FDA warning about the belladonna poisoning risk in September 2016. The agency determined at that time that belladonna-containing products have not been proven to provide any health benefits.
Customers are being asked to stop using Hyland’s homeopathic teething products and to seek immediate medical attention if their children experience any adverse health consequences following the exposure of use of the products.
Get more articles like this sent directly to your inbox.
"*" indicates required fields





0 Comments