Prenatal Blood Tests Often Mislead Pregnant Women, Endanger Their Pregnancies: Report
The report found that advertising brochures gave expecting parents a false sense of security in the accuracy of prenatal blood tests.
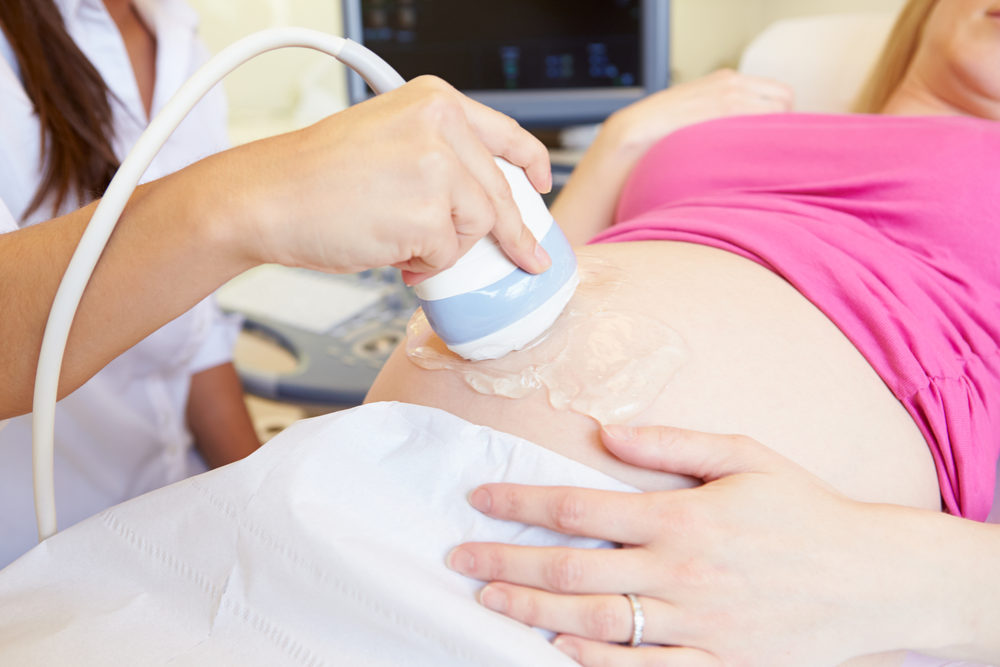
A new study warns that many prenatal blood test companies misrepresent the effectiveness of their tests in patient brochures, which could potentially endanger women’s pregnancies.
The brochures for non-invasive prenatal blood tests (NIPT) often advertise “peace of mind” and effective results for pregant women. However, a recent Hastings Center report evaluated information provided by 27 for-profit and five non-profit testing companies in 2019, and found that the information is often inaccurate.
Misleading prenatal testing results about birth defects and other conditions can cause pregnant women to make serious and erroneous decisions about the pregnancy, including termination, the report warns.
NIPTs are blood tests done between the ninth and 21st week of pregnancy, which are used to test for Down, Edwards, Patau sundry syndromes, as well as other chromosomal conditions. The tests cost between $500 and $800. However, prior research has indicated the tests are not as accurate as companies depict and can often provide false positives.
Noninvasive prenatal tests are marketed around the world as a solution for genetic testing without regulatory oversight. However, researchers from the Institute of Health Policy, Management and Evaluation at the University of Toronto warn that the brochures often mislead expecting parents.

Learn More About
Premature infants fed Similac or Enfamil cow's milk formula faced increased risk of necrotizing enterocolitis (NEC) or wrongful death.
Learn More About this Lawsuit SEE IF YOU QUALIFY FOR COMPENSATIONThe report indicated the brochures failed to meet most of the 15 standards set as a benchmark established by the Nuffield Council on Bioethics, and for-profit companies were less compliant overall.
In all, 52% of companies misrepresented the tests on brochures. Among those, 32% of for-profit companies were compliant compared to about 60% of nonprofit companies. None of the companies were compliant with all of the criteria, or even those designated by the council as “essential.”
Only four companies met 10 or more criteria. In fact, many companies failed to offer evidence in their brochures to support claims about their tests’ accuracy and provided no high-quality clinical data.
Most of the brochures included misleading language. The report found the language has the potential to confuse expectant parents about the results and could lead them terminate a healthy and normal pregnancy.
None of the brochures provided information or the likelihood of a test reporting a false positive, giving parents a false sense of security in the results. Many of the brochures failed to make clear that these are screenings, not definitive results, and should require follow-up appointments and genetic testing options before a parent makes a hasty decision.
Inaccurate and poor-quality information can lead an expecting parent to experience shock, distress and confusion. At times, parents may opt to terminate the pregnancy when they don’t understand follow-up diagnostic testing is required to confirm a positive result.
Furthermore, many doctors, OBGYNs, and maternal-fetal medicine specialists don’t have a full understanding of the tests and lack the ability to properly communicate what the results mean, despite 57% of brochures indicating the doctor could explain the test fully.
The tests are not nearly as accurate as companies make them out to be, and the majority of companies over-promise the accuracy, highlighting 88% to 99% accuracy rates, according to the report. There is no regulatory agency governing the market and no review of product brochures to indicate whether the informant provided to expecting parents is accurate.
Get more articles like this sent directly to your inbox.
"*" indicates required fields






0 Comments