Stryker Hip Wrongful Death Lawsuit Filed After Fatal Revision Surgery Infection
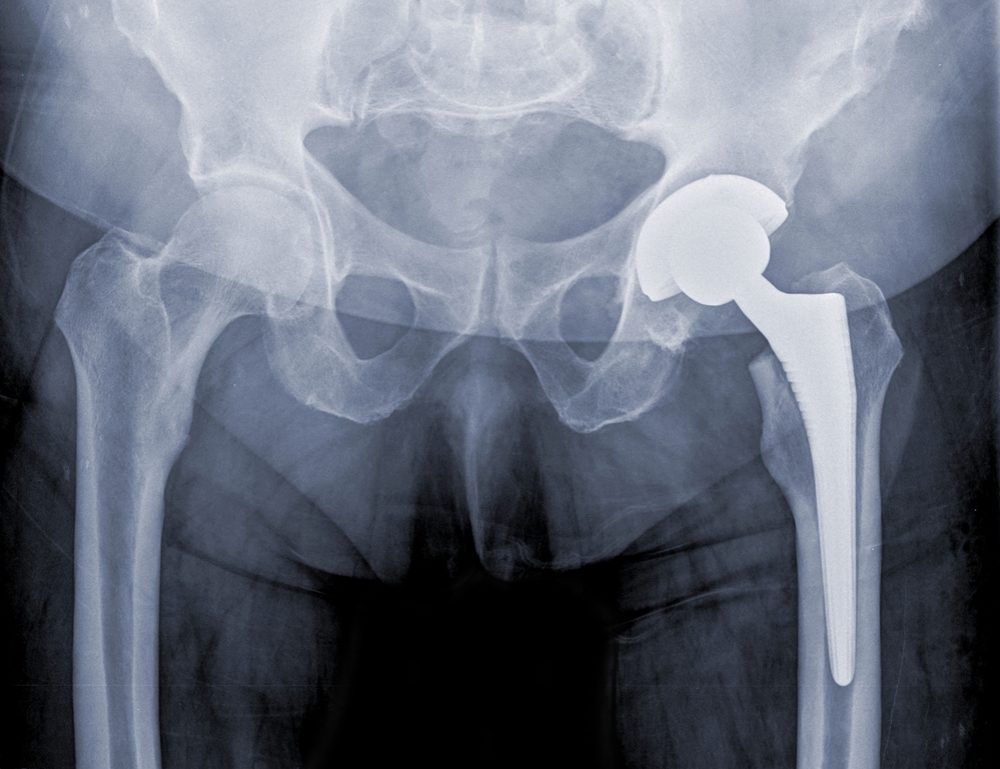
The family of a Maryland woman has filed a wrongful death lawsuit over problems with the Stryker LFit V40 and Accolade II Femoral Hip replacement system, which allegedly resulted in the need for revision surgery and the development of a subsequent infection.
The complaint (PDF) was brought earlier this month in the U.S. District Court for the District of Maryland, by the husband and two sons of Rosezetta Engle, who died due to a post-operative infection following revision surgery, which was allegedly needed because of design defects with certain Stryker hip replacement components.
Engle underwent a hip replacement in 2011, at which time surgeons used a Stryker Accolade II femoral stem with the Stryker V40 Taper LFit femoral head, which was subsequently recalled from the market due to a risk that the components may corrode and fail within a few years.

Learn More About
Lawsuits are being reviewed for several different dangerous and defective hip replacement systems.
Learn More About this Lawsuit See If You Qualify For CompensationIn January 2015, Engle required hip revision surgery to remove the implant, at which time her surgeon found “significant necrosis and fluid within the hip and gluteus minimus,” the lawsuit states. The surgeon noted that there was “corrosion and black staining under the ball and surrounding taper.”
Weeks later, Engle died on March 11, 2015, as a result of a post-operative infection.
The Stryker LFit v40 is a femoral head used in several hip replacement systems, which was removed from the market about a year after Engle’s death, amid a higher-than-expected number of reports involving hip complications and failures, including taper lock probems with the cobalt-chromium femoral head that may result in pain, inflammation, loss of mobility, disassociation and the need for risky revision surgery.
After receiving similar reports involving hip failures, a Stryker LFit v40 hip recall was issued by the manufacturer in August 2016, impacting certain large-diameter femoral heads. At that time, Stryker acknowledged that a higher-then-expected number of individuals were experiencing problems with trunnion failure, metal wear, adverse tissue reactions and other complications.
The case filed by Engle joins a growing number of other hip replacement lawsuits filed over the last year by individuals have suffered complications that resulted in revision surgery only a few years after receiving the implant. In particular, complications with the Stryker Accolade stem in combination with the LFit V40 femoral head have resulted in reports of excessive metallic debris, limited range of motion, disassociation, fractures and failed hip replacements.
The Engle family’s wrongful death lawsuit will be ccombined with other product liability cases filed nationwide over problems with the Stryker LFit v40 head, as the U.S. Judicial Panel on Multidistrict Litigation (JPML) established coordinated pretrial proceedings last year before U.S. District Judge Indira Talwani in the District of Massachusetts.
Get more articles like this sent directly to your inbox.
"*" indicates required fields






0 Comments