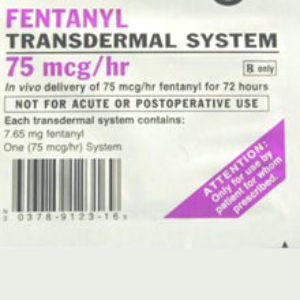
All federal fentanyl patch lawsuits filed against Watson Pharmaceuticals will be transferred to one judge for coordinated pre-trial proceedings, as part of an MDL, or multidistrict litigation.
According to an order (PDF) issued by the U.S. Judicial Panel on Multidistrict Litigation (JPML) on August 7, all wrongful death complaints filed in U.S. District Courts throughout the United States on behalf of individuals who suffered fatal injuries from Watson fentanyl patch will be consolidated before U.S. District Judge Matthew F. Kennelly in the Northern District of Illinois.
At least 22 Watson fentanyl patch lawsuits pending in 15 different federal district courts will be transferred into the MDL.
All of the complaints involve similar allegations that the generic pain patches, which are used to slowly release the potent painkiller fentanyl into patients’ bodies through skin contact, are defectively designed and have contributed to fentanyl overdose injuries and deaths.
Plaintiffs allege that the Watson pain patches feature a reservoir design, which is defective and prone to leak fentanyl gel directly onto the skin of users. Consolidating the cases for pretrial proceedings will eliminate duplicative discovery, prevent inconsistent pretrial rulings and help conserve the resources of the parties, the attorneys and the courts.
Fentanyl Patch Problems
The fenatnyl patch was originally introduced as the Duragesic pain patch, manufactured by a subsidiary of Johnson & Johnson. It is now widely available as a generic, and a number of drug companies make fentanyl pain patches, including Watson, ALZA and Mylan Pharmaceuticals.
Fentanyl is a painkiller that is about 100 times more powerful than morphine. While the patches are supposed to deliver the fentanyl gel through a membrane that is placed on the skin, a number of the products have been plagued by production problems and manufacturing defects.
The FDA has received hundreds of adverse event reports involving fentanyl overdoses and deaths associated with the use of pain patches, causing many to question whether they can be safely manufactured.
According to allegations raised in the Watson fentanyl patch lawsuits, plaintiffs indicate that the problems could have been avoided through the use of a matrix design to hold the fentanyl in the patch, which eliminates the risk of leakage. The complaints allege that Watson knew about the safer alternative design, but instead continued to use a reservoir design, which was prone to leakage, in order to save money.
Other Generic Fentanyl Patch Makers Excluded from MDL
Following a request filed in April by some of the plaintiffs involved in the litigation, Watson filed a response agreeing that the litigation should be centralized as part of an MDL. However, the drug maker argued that other manufacturers of fentanyl pain patches should also be consolidated in the same MDL.
Although plaintiffs took no position on this request, several manufacturers of other generic fenanyl patches filed responses opposing the inclusion of their cases in any broad fentanyl pain patch MDL.
The U.S. JPML denied the request to centralize all actions, irrespective of the manufacturer, indicating that each group of cases will involve unique product and defendant-specific issues, and that there are insufficient commonalities among the allegations made against the various defendants.
“Centralization of all actions against all manufacturers will add few efficiencies to the resolution of this litigation, especially given that the ALZA and Mylan defendants employ a matrix design on their fentanyl patches — the same design that plaintiffs contend is a safer alternative to the Watson reservoir design,” wrote Judge John G. Heyburn II, Chairman of the Panel on Multidistrict Litigation.
The Panel also denied a request by one plaintiff to be excluded from the Watson fentanyl patch MDL.
The creation of a Watson fentanyl patch MDL means that all lawsuits filed in federal court will be consolidated for pre-trial proceedings. However, if the parties fail to reach a settlement or otherwise resolve the litigation, the cases will be remanded back to their districts of origin and be resolved through individual trials.
Get more articles like this sent directly to your inbox.
"*" indicates required fields
1 Comments





monicaSeptember 7, 2012 at 7:21 am
I DID LOSE MY BELOVED DAUGTHER DUE TO THIS FACT MY LIFE AND MY FAMILY LIFE NEVER WILL BE THE SAME iCOME IMIGRATE THIS COUNTRY LEGALLY UNDER EMBASADOR AND DIPLOMATIC VISA 29 YEARS AGO SCAPING FROM A COMUNIST REGIME WHEN MY LITLE GIRL WAS BORN i WAS A WIDOW BUT I WAS STRONG AND SHE MAKE MYSELF KEEP GOING LATER A MARRY AND HAVE A BOY IN JANUARY 2010 MEDICATION WAS PRESCRIBE TO YESENYA SHE WAS 22 [Show More]I DID LOSE MY BELOVED DAUGTHER DUE TO THIS FACT MY LIFE AND MY FAMILY LIFE NEVER WILL BE THE SAME iCOME IMIGRATE THIS COUNTRY LEGALLY UNDER EMBASADOR AND DIPLOMATIC VISA 29 YEARS AGO SCAPING FROM A COMUNIST REGIME WHEN MY LITLE GIRL WAS BORN i WAS A WIDOW BUT I WAS STRONG AND SHE MAKE MYSELF KEEP GOING LATER A MARRY AND HAVE A BOY IN JANUARY 2010 MEDICATION WAS PRESCRIBE TO YESENYA SHE WAS 22 AND HALF YEARS OLD IT HAPEN SO FAST IT IS NO DAY I GO BY THAT i MISS HER i HOPE ATTORNEY WIN THIS CASE FOR ALL THE VICTIMS THIS HAVE CAUSE SO MUCH PAIN AND ALSO TO THEIR BABIES CHILDRENS SURVIVERS MY GRANDSON LIFE ALSO WAS CHANGE FOREVER God help us and my condolescens and rrespect to the other families who have lose a beloved one due to watson Fentanyl patch damaged.