Zostavax Vaccine Lawsuit Filed By 22 Plaintiffs Over Persistent Shingles
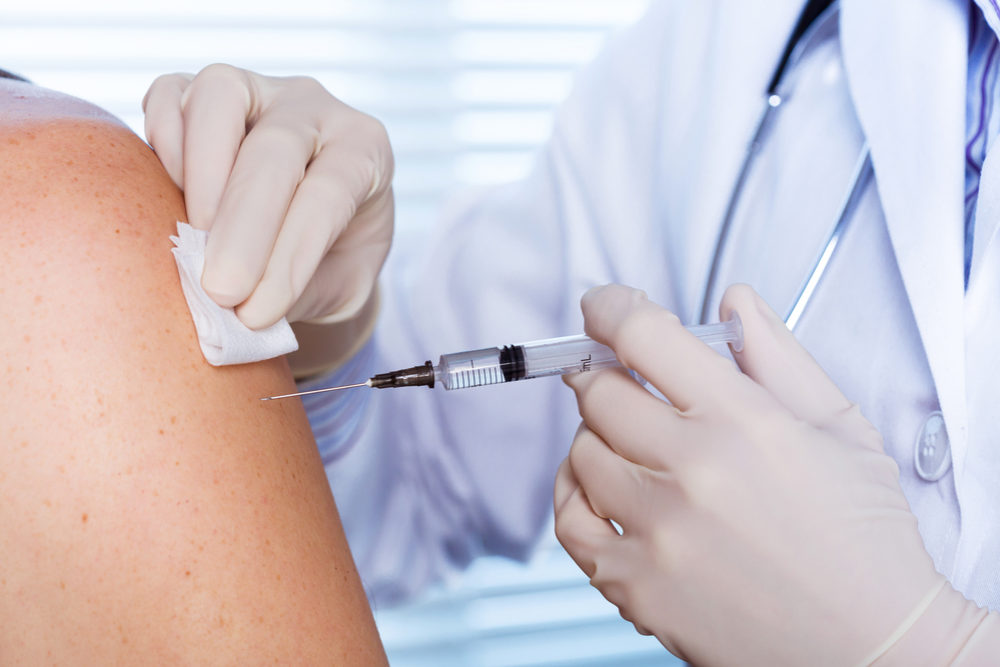
A product liability lawsuit filed by on behalf of 22 different people accuses Merck of manufacturing and selling the shingles vaccine Zostavax without first ensuring that it was safe and effective, resulting in all of the plaintiffs developing a persistent form of the skin condition.
The complaint (PDF) was filed in the Superior Court of New Jersey on August 17, indicating that the plaintiffs took the vaccine to avoid shingles, but instead developed a recurring strain of herpes zoster, which is more difficult to treat than usual.
Shingles is caused by the same virus, varicella zoster, which causes chickenpox. Later in life, the virus can be reactivated and cause shingles, which is a painful rash that usually appears as a stripe of blisters on the body. Pain can continue even after the rash is gone, which is known as postherpetic neuralgia.
Learn More About
Side effects of the shingles vaccine Zostavax may result in the development of a painful and persistent strain of shingles
Learn More About this Lawsuit SEE IF YOU QUALIFY FOR COMPENSATIONTo prevent shingles, adults can get a chickenpox or shingles vaccine. Zostavax was approved in May 2006, for the prevention of shingles among individuals ages 60 or older. It is a more potent version of the Merck chickenpox vaccine, Varivax.
The Zostavax vaccine lawsuit claims that Merck used an under attenuated live strain of the varicella zoster virus (VZV) in their product, which was not weakened enough to prevent reactivation of the virus. Instead of the body developing the proper immune response, the live virus combined with the old virus into a more virulent strain of shingles.
“At all times relevant hereto, the patient information sheet, as well as the label and prescribing information for Zostavax, did not adequately, if at all, address the risk of viral infection,” the lawsuit states. “All that was addressed was the concern that a rash and itching might develop at the injection site. This was despite the fact that shingles was a noted occurrence during clinical trials of the vaccine.”
The complaint indicates that by September 2015, there were already 1,111 reports of severe Zostavax problems identified among adverse event reports, including 36 deaths. Many of the reports were linked to viral infections of the central nervous system, resulting in side effects such as acute disseminated encephalomyelitis, peripheral neuropathy, myalgia, arthralgia, rashes, severe cutaneous disease, herpes keratis that caused vision loss, pneumonia, brain inflammation, facial paralysis and death.
Shingles itself can cause scarring, bacterial infections, encephalitis, hearing loss, vision problems and other complications.
A similar Zostavax lawsuit, filed on behalf of 15 plaintiffs, was filed in New Jersey state court in July. Some have estimated that thousands of future lawsuits may be filed in courts nationwide over side effects of the Zostavax vaccine.
Get more articles like this sent directly to your inbox.
"*" indicates required fields





0 Comments