A number of women have reported suffering serious and debilitating injuries from side effects of Mirena IUD, where the implanted birth control increases fluid pressure in the brain, causing migraines and permanent vision loss. There are also claims that it perforates the uterus, migrates to other areas of the body and causes infections or other problems.
MIRENA IUD LAWSUIT STATUS: Financial compensation may be available through a Mirena IUD lawsuit as a result of the manufacturer’s failure to adequately warn about the risks associated with the device.

Did You Know?
Millions of Philips CPAP Machines Recalled
Philips DreamStation, CPAP and BiPAP machines sold in recent years may pose a risk of cancer, lung damage and other injuries.
Lawyers are reviewing potential Mirena cases for women throughout the United States who have suffered:
- Pseudotumor Cerebri or Idiopathic Intracranial Hypertension
- Perforation or Puncture of the Uterine Wall
- Migration of the Mirena IUD Outside the Uterus
- Infections
- Surgical Removal of the IUD Due to Complications
MANUFACTURER: Bayer
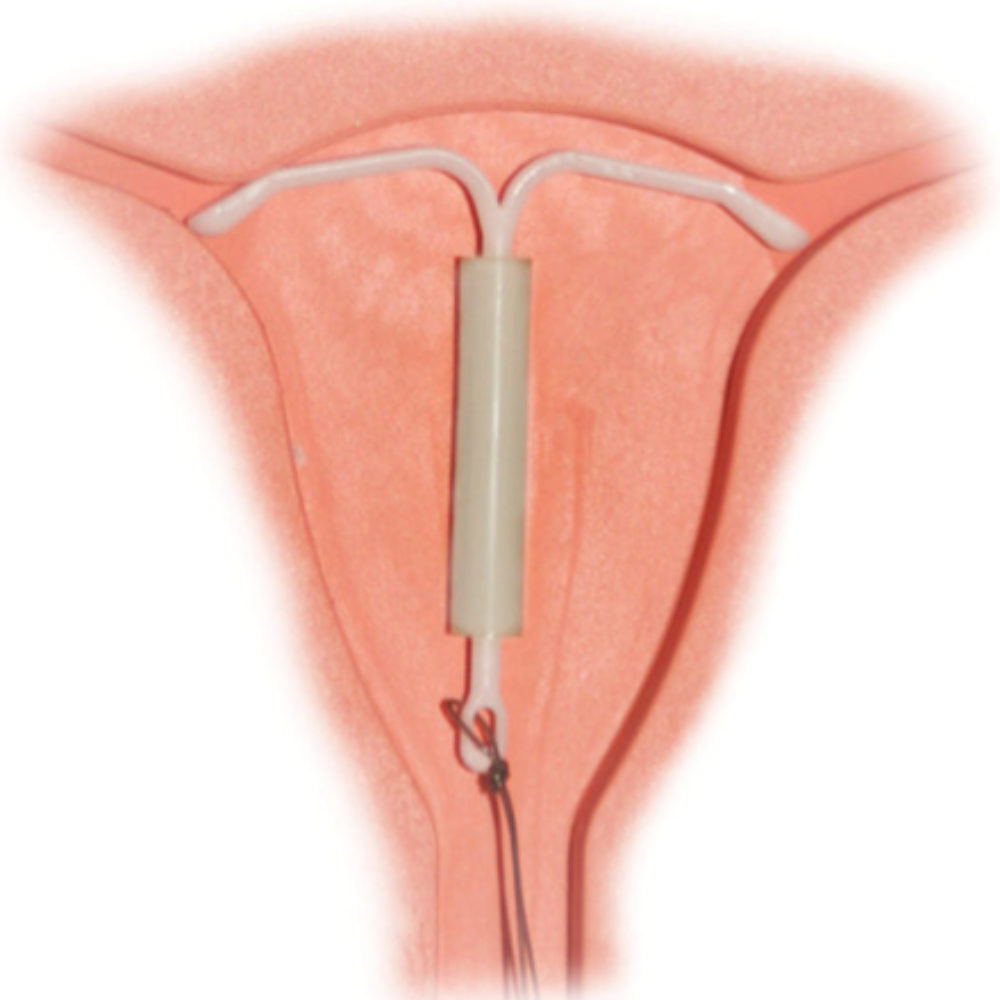
OVERVIEW: Mirena, which is sometimes referred to as an IUD (Intrauterine Device) or IUS (Intrauterine System) is a long-acting form of birth control that is placed into the uterus to prevent pregnancy. The IUD disrupts the connection between the egg and sperm, and Merina also releases levonorgestrel, a progestin that helps keep the woman’s ovaries from releasing eggs.
Mirena was approved by the FDA in 2000, and use has grown in recent years amid heavy promotion as a hassle-free form of birth control that lasts for 5 years or longer. The manufacturer has been attempting to promote Mirena IUD to appeal to younger and younger women and girls, despite serious risks that they may not be adequately disclosing or warning about.
MIRENA LITIGATION STATUS UPDATE: Although the Mirena warning labels do indicate that there is a risk of perforation, the information provided to consumers and the medical community suggests that such problems only occur during insertion of the IUD, which is misleading and inaccurate. A large number of women have experienced “spontaneous” Mirena IUD complications that may occur years after the device was inserted.
Lawsuits over Mirena migration problems have been centralized at the state level in New Jersey Superior Court in Bergen County as part of an MCL, or Multicounty Litigation. At the federal level, all Mirena IUD injury lawsuits have been centralized and consolidated for pretrial proceedings before U.S. District Judge Cathy Seibel in the Southern District of New York. In August 2016, Judge Seibel dismissed all federal Mirena IUD migration lawsuits, but state lawsuits and claims of other injuries, like pseudotumor cerebri, were unaffected.
However, in April 2017, all Mirena pseudotumor cerebri lawsuits were consolidated in a new MDL in the U.S. District Court for Southern New York before District Judge Paul A. Engelmayer
MIRENA IUD PROBLEMS: A number of potentially serious problems and complications have been associated with Mirena IUD side effects, some of which the manufacturer may not have adequately researched or warned about, including:
- Pseudotumor cerebri/idiopathic intracranial hypertension: An increase in cerebrospinal fluid which causes pressure on the skull, resulting in migraines, vision loss and other problems.
- Uterus Perforation: Mirena may puncture the uterus or embed in the uterine wall. Mirena IUD perforation can cause internal scarring, infection or damage to other organs and may result in the need for surgery.
- Migration: Delayed detection of a perforation may allow the Mirena IUD to migrate through the intraabdominal cavity, potentially resulting in intestinal peforations, intestinal obstruction, absecesses or adhesions.
- Expulsion: Partial or complete expulsion of Mirena IUD may occur. It may be more common among younger women using Mirena IUD, women who have not had children or women who have the device inserted immediately after childbirth or abortion.
- Infection: As a result of infection from Mirena IUD, women could suffer serious scarring that may deminish fertility or their ability to conceive, and in severe cases infection may be life-threatening and result in death from Mirena.
An analysis released in May 2013 by a private company found that the FDA had received more than 47,000 reports of Mirena IUD injuries and complications between November 1997 and late August 2012. Most of the problems involved the Mirena being expelled from women’s bodies, migrating to different locations, or causing vaginal hemorrhaging.
A report issued by the Institute for Safe Medication Practices in January 2018 found that the number of adverse events reported to the FDA involving the Mirena IUD outnumbered reports concerning all other leading birth control methods combined.
BAYER ADVERTISING WARNING: In December 2009, the FDA issued a warning letter to Bayer over its promotions of Mirena. The FDA accused the drug manufacturer of overstating the efficacy of Mirena, presenting unsubstantiated claims, minimizing the risks of Mirena and using false and misleading presentations.
The FDA accused Bayer of organizing live in-home Mirena promotional events that claimed that the use of the IUD would improve couples’ relationships and sex lives. They also blasted the company for telling women that using Mirena would help them “look and feel great.” The FDA said that there was no evidence the device did any such thing.
The FDA also pointed out that at least 5% of Mirena IUD recipients reported decreased libido in clinical trials, which would certainly not help a couple’s intimacy or sex life. They also noted that women using Mirena had reported side effects including irregular bleeding, ovarian cysts, back pain, weight increase, breast pain and tenderness, and acne. None of those things will help someone look or feel “great,” FDA officials said.
Image By Gloecknerd via Wikimedia Commons: Public Domain
471 Comments



GabrielaJuly 9, 2023 at 8:20 pm
I had the iud mirena inserted in me March 2022 it caused me sever cramps threw out the first 6months they were so hard they felt like contractions I got medicine to stop my period because I was on my period for 1 month . I had terrible raging mood swings I had postpartum and then 1 yr later in may 2023 I started getting a lot of bleeding and cramps and it lasted 1 whole month again so I’m June 20[Show More]I had the iud mirena inserted in me March 2022 it caused me sever cramps threw out the first 6months they were so hard they felt like contractions I got medicine to stop my period because I was on my period for 1 month . I had terrible raging mood swings I had postpartum and then 1 yr later in may 2023 I started getting a lot of bleeding and cramps and it lasted 1 whole month again so I’m June 2023 I went to pee and I had horrible pains in my lower back I felt like a needle was pocking my butt and then I passed two huge meat looking blood clots and between those balls was the iud it fell directly out of me ! I kept bleeding as if I just given birth! My doctor prescribed me provera for the bleeding and it went away but now I suffer from extreme abdominal pain and sex is painful idk anything of why this happens if it had moved or if it was embedded inside me I have no answers and I’m left with so many questions! Iud mirena is dangerous and very toxic to woman’s health!
GailMarch 29, 2023 at 1:43 am
I had the mirena Iud place in me May 4, 2022 . It was worst then giving birth when they inserted it into me . I bleed so bad for the next 2 months that when I went to get exam to see if it was in place all the test shown it wasn’t inside my uterus 7 month down line guess it Perforated my uterus, uterus, and now it’s flowing around in my lower abdomen area . Waiting on surgery now to have it remove[Show More]I had the mirena Iud place in me May 4, 2022 . It was worst then giving birth when they inserted it into me . I bleed so bad for the next 2 months that when I went to get exam to see if it was in place all the test shown it wasn’t inside my uterus 7 month down line guess it Perforated my uterus, uterus, and now it’s flowing around in my lower abdomen area . Waiting on surgery now to have it removed. Worst 7 months of my life
LatoshaMarch 1, 2023 at 10:56 am
Mirena caused me to have excess bleeding, severe pain,headaches, moods swings, and after the removal of the mirena it still causes me to have heavy bleeding, and severe menstrual pains to the point where I pass out I have been without the mirena for several years and it's still causing all kinds of trouble.....
CandaceFebruary 3, 2023 at 2:09 pm
I got the mirena in 2015 I think and I had it for 3 years. I suffered with severe pain, bleeding, blood clots, headaches, mood swings. I went to the er 2 or 3 times because of severe pain and bleeding and they didn’t know what was wrong and sent me home every time until I think it was my 3rd time at the er and they did an ultrasound. They told me that the iud was embedded in my cervix and I had t[Show More]I got the mirena in 2015 I think and I had it for 3 years. I suffered with severe pain, bleeding, blood clots, headaches, mood swings. I went to the er 2 or 3 times because of severe pain and bleeding and they didn’t know what was wrong and sent me home every time until I think it was my 3rd time at the er and they did an ultrasound. They told me that the iud was embedded in my cervix and I had to have surgery to have it removed. While I was in surgery they had a hard time getting it out of me and I was in a lot of pain afterwards. I still suffer with pain from it. It was supposed to help with my bleeding and keep me from having a baby for 5 years. Since I had to have it removed 2 years early I got pregnant 6 months later. It was a very tough pregnancy to say the least!
LaMyraFebruary 12, 2022 at 8:57 pm
I had my Mirena IUD inserted in 2015. Suffered several “possibly unrelated” infections since. Went to have it removed yesterday and the Gyn yanked without any success. She pulled at it to the point “it broke” and now I have to wait a week to be sedated and have it removed. I’m in so much pain and taking Motrin. Please keep me in your prayers.
ShaunonAugust 22, 2021 at 11:03 pm
I had the mirena for 3 1/2 years. I didn’t realize how much it was affecting my health. I’ve recently had it removed July 13 2021. The doctor sent out the mirena to be tested it came back saying it had grown out a bacteria but most woman did not require treatment.. Well I did require it. I had some antibiotics that I just started taking because I do not have health insurance. I would like to get t[Show More]I had the mirena for 3 1/2 years. I didn’t realize how much it was affecting my health. I’ve recently had it removed July 13 2021. The doctor sent out the mirena to be tested it came back saying it had grown out a bacteria but most woman did not require treatment.. Well I did require it. I had some antibiotics that I just started taking because I do not have health insurance. I would like to get the word out of how much damage this has caused me mentally and physically
ANTONIAMarch 23, 2021 at 9:07 pm
my mirena was in for over ten years, kept asking my gynae to revove it. turns out it was embeded. he pulled so hard for over a minute, i could feel my womb and ovaries being dragged down. he tore my uterus. i bled heavily for 3 days so went to doctor who gave me antibiotics. my gynae ignored me for 2 half years, ive been in constant pain, and he ruined my repair, now have prolapse and lump on cerv[Show More]my mirena was in for over ten years, kept asking my gynae to revove it. turns out it was embeded. he pulled so hard for over a minute, i could feel my womb and ovaries being dragged down. he tore my uterus. i bled heavily for 3 days so went to doctor who gave me antibiotics. my gynae ignored me for 2 half years, ive been in constant pain, and he ruined my repair, now have prolapse and lump on cervix. they wont even look at my smear pap test as im negative for hpv. im suing, LOOKING FOR A LAWYER
JessicaMarch 2, 2021 at 2:51 am
My Mirena migrated into the left portion of my body cavity and I had to have surgery to have it removed
BrendaAugust 3, 2020 at 4:03 pm
I had the mirena put in August 2017. I was having heavy bleeding issues and my Dr felt this would be better then a ablation. I wish I'd gotten the ablation. Since the placement I've gained close to seventy five pounds. Still gaining. I've done everything to lose weight. Can't. I keep gaining instead along with the weight gain,my emotions are all over the place, I can't focus or stay focu[Show More]I had the mirena put in August 2017. I was having heavy bleeding issues and my Dr felt this would be better then a ablation. I wish I'd gotten the ablation. Since the placement I've gained close to seventy five pounds. Still gaining. I've done everything to lose weight. Can't. I keep gaining instead along with the weight gain,my emotions are all over the place, I can't focus or stay focused on one task so working has been let's say very hard very challenging. Along with all that I just got out of the Hospital for pancreaitis and had to have my gall bladder removed. I feel that all my problems I'm having are due to the mirena. Everything I stated and even more issues all began within two weeks of getting this thing. The only good thing is im not bleeding anymore. Which is why I still have it in. I'm struggling to find a Dr who can remove it and give me a hysterectomy or a ablation. In scared of the crash I hear will happen after wards. Im already dealing with emotional issues that started after I had it put in. A crash of my hormones would be very devastating to me I feel. Don't any one get this thing! It's terrible. Oh ya and my sex drive is negative zero since having it put in.
MeghanJuly 23, 2020 at 3:09 pm
I had my Mirena IUD placed for first time use on June 8, 2020. I had it removed July 23, 2020. First day of placement, I was crippled with a migraine that lasted 3 days (never get migraines) and followed by a pulsated windy sound in my right ear. I would continue to get a lingering headache every week and the ear became constant and worse over time. I also got an ache in my left leg and vision see[Show More]I had my Mirena IUD placed for first time use on June 8, 2020. I had it removed July 23, 2020. First day of placement, I was crippled with a migraine that lasted 3 days (never get migraines) and followed by a pulsated windy sound in my right ear. I would continue to get a lingering headache every week and the ear became constant and worse over time. I also got an ache in my left leg and vision seemed blurry once in a while. My doctor thinks I got a reaction to the hormone and had it immediately removed after my 6-8 week check up. I consider myself a healthy person but I did not feel myself while using this IUD.
DesireeJune 24, 2020 at 5:16 am
I had mirena twice. I got it re.oved the first time because I had bad cramps and mood swing. I got pregnant shortly after and almost lost my son as i was induced for 5 days for and emergency delivery and my cervix would not dilate at all. I demanded a c section and i got another mirena put in smh worst decision ever. Now 4 years later i have been teyong to get ot removed for 9 months. I have migra[Show More]I had mirena twice. I got it re.oved the first time because I had bad cramps and mood swing. I got pregnant shortly after and almost lost my son as i was induced for 5 days for and emergency delivery and my cervix would not dilate at all. I demanded a c section and i got another mirena put in smh worst decision ever. Now 4 years later i have been teyong to get ot removed for 9 months. I have migranes blurred vision. I have increased csf pressure and a large cyst in my pineal gland in my brain. I have an irregular heartbeat that has an "unexplained" cause and i also have repeated infections. I believe all caused by mirena. Now my cervix still wont dilate or open enough for it to be removed, strings cant be located, & my uterus is detaching from my uteran wall which will eventually cause it to prolapse. I hate bayer and i hate my choice to trust in mirena
CaseyJune 14, 2020 at 6:42 am
I had mirena iud and my memory loss is unbelievable.. I struggle to remember alot. Once I had it removed my uterus was torn during removal.. I am still trying to figure out how to file a lawsuit against them.. any help??
KatriceMarch 20, 2020 at 3:33 pm
I got the Mirena IUD in 2014. After gaining about 60 pounds and loosing most of my hair I went to have the IUD removed. When I went to my doctor, initially they could not find it. I was sent for two different ultrasounds (abdominal and vaginal) and they still could not locate it. After this my physician insisted that I expelled the IUD. However I knew that my body was still reacting to the hormone[Show More]I got the Mirena IUD in 2014. After gaining about 60 pounds and loosing most of my hair I went to have the IUD removed. When I went to my doctor, initially they could not find it. I was sent for two different ultrasounds (abdominal and vaginal) and they still could not locate it. After this my physician insisted that I expelled the IUD. However I knew that my body was still reacting to the hormones. My physician then sent me for an X-ray. The X-ray revealed that the IUD had migrated and was now in my lower abdomen. In order to have the IUD I had to have the removed I had to receive surgery. I was put to sleep and put in a breathing machine with tubes down my throat. I had two incisions on in my belly button and one on the right side of my stomach. Since having the device removed I am still struggling with my weight and my hair is extremely thin. My body has never been the same
MaeMarch 7, 2020 at 1:13 pm
I had my implant around nov2014-Jan2015 and can’t remember exactly when it was removed a year after or April of 2015. I had never had any complications prior to it just didn’t want to get pregnant 6years down the road with a surprise. My periods almost completely stopped and on Easter Day I had a horrible migraine and abdominal pain. Went to the er and nothing was said to me. I had it removed imme[Show More]I had my implant around nov2014-Jan2015 and can’t remember exactly when it was removed a year after or April of 2015. I had never had any complications prior to it just didn’t want to get pregnant 6years down the road with a surprise. My periods almost completely stopped and on Easter Day I had a horrible migraine and abdominal pain. Went to the er and nothing was said to me. I had it removed immediately as possible afterwards by the dr who put it in and she didn’t want to remove it but I made them. Since then I have not had those migraines but I have had heavy flow with a Diagnosis Of Adenomyosis now. Which is very painful and I have to take medication to help it. I don’t recommend it for someone who may want children later in life at all.
TaniaFebruary 15, 2020 at 7:50 am
I have had the Mirena IUD for a little over a year, and it has been one of the worst experiences in my life. Literally only a few days in and my stomach was more bloated than usual. Within 3-4 months I had gained over 30 pounds. I have always maintained the weight 110-115 pounds for 6 years. Now it has been over a year and I weight almost 190 pounds which is devastating. I have no idea why I’m gai[Show More]I have had the Mirena IUD for a little over a year, and it has been one of the worst experiences in my life. Literally only a few days in and my stomach was more bloated than usual. Within 3-4 months I had gained over 30 pounds. I have always maintained the weight 110-115 pounds for 6 years. Now it has been over a year and I weight almost 190 pounds which is devastating. I have no idea why I’m gaining this much weight, cause I have never been this unhealthy. I have tried dieting and drinking water only but I’m still gaining weight. Recently I went to the ER because of chest pains that I have consistently for 2 weeks, like if my heart kept skipping beats and have rapid heart rates. I have had a sore throat for almost 2 months, just in general health problems. They said my thyroid (TSH) count was a little above average. I asked if it was my IUD causing it and they said it was unlikely but I feel in my heart that this is causing my problems. The reason I wanted the IUD is because of my bad cramps. But this caused way more abdominal pain and psychological suffering. It does have benefits like having light periods or none at all, Or not having to worry about getting pregnant. The reason I still have it because the doctors said it’s not my IUD causing this, or maybe it’s just a phase my body is experiencing. I need more doctor’s opinions on this before taking this device out, cause I feel like it is not normal.
Desire'February 4, 2020 at 4:08 pm
I got the Mirena inserted in 2017 and still have it implanted. But since then my eye sight has decreased causing me to have to wear glasses now, I have extreme back pain, I have a lot of mood changes, Depression, Headaches/migraines, EXTREME loss of interest in sex, missed periods, and I have gained AT LEAST 80 lbs if not more since the implantation (even dieting and exercising but I'm still gaini[Show More]I got the Mirena inserted in 2017 and still have it implanted. But since then my eye sight has decreased causing me to have to wear glasses now, I have extreme back pain, I have a lot of mood changes, Depression, Headaches/migraines, EXTREME loss of interest in sex, missed periods, and I have gained AT LEAST 80 lbs if not more since the implantation (even dieting and exercising but I'm still gaining weight!).
IeshaJanuary 28, 2020 at 6:10 am
I had got the IUD stop my heavy periods but it came out and left damaged my insides
RebeccaJanuary 11, 2020 at 7:07 pm
My 12 year old daughter has PCOS, her doctor recommended that we do a birth control to help control her periods and to help with her hormone imbalance. We went to her post opp appointment after her Mirena was placed on November 1, 2019, they did an ultrasound to check the placement and her IUD is sideways with one arm of the BC bent backwards and pushed into her uterine wall. The radiologist state[Show More]My 12 year old daughter has PCOS, her doctor recommended that we do a birth control to help control her periods and to help with her hormone imbalance. We went to her post opp appointment after her Mirena was placed on November 1, 2019, they did an ultrasound to check the placement and her IUD is sideways with one arm of the BC bent backwards and pushed into her uterine wall. The radiologist stated he has never seen anything like this in his career. The doctor is removing the Mirena, they don’t know if it has perforated her uterine wall or not. She has had excessive bleeding since placement. I would not recommend anyone for this product. She is already going to have issues having children in the future, if this product damages her uterus to the point where she can’t have children at all, it will be devastating.
JenniferJanuary 9, 2020 at 7:49 am
I had the Mirena inserted For about 5 months and one day I passed out at work because I started bleeding horrifically got to the hospital and found out that not only had I gotten pregnant but my IUD I had perforated my uterus and they couldn’t stop the bleeding I was .02% away from having to have full blood transfusion because I had bled nearly to death. Needless to say I lost the baby and almost [Show More]I had the Mirena inserted For about 5 months and one day I passed out at work because I started bleeding horrifically got to the hospital and found out that not only had I gotten pregnant but my IUD I had perforated my uterus and they couldn’t stop the bleeding I was .02% away from having to have full blood transfusion because I had bled nearly to death. Needless to say I lost the baby and almost my life and had to have surgery to stop the bleeding remove the deceased fetus and save my life also to remove the IUD I was severely infected and have had this type of thing happened to me two times since then both times I tried to get pregnant I almost bled to death because of the damage that the IUD did to my uterus and cervix. My doctor says I will never have children and even though I can get pregnant I will never be able to carry to term and I will always lose the baby in a very dangerous manner all thanks to the Mirena iud
SarahDecember 24, 2019 at 5:04 am
I got a Mirena iud in fall of 2019, to help with very heavy periods that started coming 2/month. I immediately had spotting only, and then no periods at all. This is more convenient, though I am uncertain as to whether this is healthy. And my blood pressure has gone from consistently healthy to very high nearly daily. My head throbs and I can feel my pulse throughout my entire body. I also have v[Show More]I got a Mirena iud in fall of 2019, to help with very heavy periods that started coming 2/month. I immediately had spotting only, and then no periods at all. This is more convenient, though I am uncertain as to whether this is healthy. And my blood pressure has gone from consistently healthy to very high nearly daily. My head throbs and I can feel my pulse throughout my entire body. I also have very strange intermittent blurred vision that keeps me from working for days. I am a photographer so this is an unacceptable side effect. Wondering if it’s related to the HBP... and the Mirena. This string of comments while anecdotal, is beyond alarming.
ChristinaDecember 22, 2019 at 12:47 am
I had IUD inseted. Having health problems tried 3 different doctors in 12 months 4 attempts for removal no luck. Had surgery and surgery unsuccessful. Now to remove I need to have a full or partial hysterectomy. Never was warned these horrible situations. Just percentage of becoming pregnant not ever being able to bare children again let alone the many health issues. Doctor is saying that if I get[Show More]I had IUD inseted. Having health problems tried 3 different doctors in 12 months 4 attempts for removal no luck. Had surgery and surgery unsuccessful. Now to remove I need to have a full or partial hysterectomy. Never was warned these horrible situations. Just percentage of becoming pregnant not ever being able to bare children again let alone the many health issues. Doctor is saying that if I get a full are partial hysterectomy surgery that I could Hemorrhage to death.
AngelaOctober 5, 2019 at 11:30 pm
By the time I paid attention to what was happening to my body it was too late. I lost all body hair; legs, arms, fingers smooth as if they were waxed, and most of all my eyebrows are completely gone they use to be so thick, and the hair in the top of my head got very thin.
SaraJuly 23, 2019 at 4:55 am
I started using the Mirena to stop my periods. My periods were always so harsh on me. I’ve been on it for a year now and seems that my hair has grown shorter. My hair had stopped growing and the longest hairs had fallen out. I can’t seem to grow them back. Many people have noticed. Will my lost hair ever come back?
ErinApril 20, 2019 at 1:39 pm
After not being able to get to a DEPO shot appointment, due to an employer not allowing me the time off for a medical appointment(that supervisor was released of their duty for the denial) I suffered for months with horrific periods. I was not able to be put back on depo, due to what they say was high blood pressure. OK, fine. What were the other options, and my Dr at the time said the Mirena IUD [Show More]After not being able to get to a DEPO shot appointment, due to an employer not allowing me the time off for a medical appointment(that supervisor was released of their duty for the denial) I suffered for months with horrific periods. I was not able to be put back on depo, due to what they say was high blood pressure. OK, fine. What were the other options, and my Dr at the time said the Mirena IUD would be ideal. I did not and do not want kids, and it was explained that because I have not had kids, the insertion would be painful, and there would be some pain, and bleeding for a few months-and then all should be good - this was done 7/5/17. Well, flash forward almost 2 years - bleed super heavy for the first 8 months, and was told that was "normal" and it should balance out. OK, Dr knows best. Since then I have irregular, and break through bleeding EVERY month. I get the start of migraines, cramping at times. The acne is my tale-tale sign that my cycle is going to start. My vision has gone down hill, I would say over the past 2 years as well. Made an appointment, with my new Dr., to have the IUD removed, and see what other options I have available to me -
FernandaApril 18, 2019 at 2:37 am
I started with this Mirena birth control in December after 2 kids I decided to start with BC, this was the worst decision I’ve ever made. I had to go to an emergency room yesterday from a crucial pain it almost felt like I was giving birth, after hours in the hospital and all the testing doctors found out I had an ectopic pregnancy and it had burst so I had to go through surgery to get the fetus r[Show More]I started with this Mirena birth control in December after 2 kids I decided to start with BC, this was the worst decision I’ve ever made. I had to go to an emergency room yesterday from a crucial pain it almost felt like I was giving birth, after hours in the hospital and all the testing doctors found out I had an ectopic pregnancy and it had burst so I had to go through surgery to get the fetus remove and Fallopian tube. THIS IS THE WORST WAY TO PREVENT PREGNANCY. Please women out there don’t do it it’s not effective at all!!!!
ChasityFebruary 20, 2019 at 9:39 pm
I’ve always had some sort of birth control due to being fertile myrtle. I’ve been on numerous types of BC pills, nuva ring, depo shot, patches, etc. After my son was born I got the mirena put in 2015, the first year was fine. After that I’ve had nothing but problems. I have been to my obgyn several times and they would just blow it off like it’s normal! They would claim that they could take it o[Show More]I’ve always had some sort of birth control due to being fertile myrtle. I’ve been on numerous types of BC pills, nuva ring, depo shot, patches, etc. After my son was born I got the mirena put in 2015, the first year was fine. After that I’ve had nothing but problems. I have been to my obgyn several times and they would just blow it off like it’s normal! They would claim that they could take it out, but the mirena is the most affective to not get pregnant, and that I wouldn’t find another form of BC as goood as the mirena and many other things! I should have switched drs then but I had never been to another dr, and this same dr delivered my child and I had seen this dr multiple times, so I listened to the dr, blew off what I was feeling, and did not get it removed. I’ve not had a period in 3 years, but after sex I always cramp and hurt horribly and start spotting. I have migraines all the time, my boobs hurt all the time, I rarely want sex, I’m moody, my vision is blurry, I’ve gained 40 + pounds, My thyroid is messed up, acne is ridiculous and have had to start seeing a dermatologist, my hair is thinning and falling out, and now I can’t find the mirena. I am going to a new dr next week to see what is going on. I don’t know if it’s fell out or dislodged and moved somewhere else! 2 ppl that I know have had to have theirs surgically removed in the last year due to the mirena moving and getting lodged in their uterus. I am only 27 and I hope the mirena has not ruined my body! This product should be took off the market! And I honestly hope everyone that reads these comments listens to what is being said! Mirena is no good! Stay away!!!!
CourtneyFebruary 6, 2019 at 3:17 am
I got the Mirena put in March of 2017 had lower abdominal pains so dr had sonograms done, nothing was wrong, Jan of 2018 felt for the strings they were gone but I found it on the floor, 2weeks later I was pregnant along with a new skin disease hydranitis superativa which I never had before the Mirena so now I’m pregnant and have painful boils under my arms, Valentine’s Day comes I miscarry the sad[Show More]I got the Mirena put in March of 2017 had lower abdominal pains so dr had sonograms done, nothing was wrong, Jan of 2018 felt for the strings they were gone but I found it on the floor, 2weeks later I was pregnant along with a new skin disease hydranitis superativa which I never had before the Mirena so now I’m pregnant and have painful boils under my arms, Valentine’s Day comes I miscarry the saddest day ever, 6 months later boom pregnant again soon as I missed my period and got excited took three test to make sure 2 days later miscarriage again sadness all over again, now feb 2,2019 missed period that day and took four test until today and just like that I’m bleeding and losing the baby yet again, let alone I’m still battling the Hs apparently it came from the hormone imbalance from the Mirena, but I’m so hurt and can’t believe the Mirena did all this to me, I’ve never had a miscarriage in my life til this Mirena, I want answers and want to be able to actually go full term with a pregnancy which seems impossible because I’ve been pregnant in an year span three times and haven’t made it more than five weeks. And my vision got worse but didn’t know that was a side effect just thought that work and computers made it worse til I read all these stories. This bc needs to b taken off the market and they need to help all the women that are suffering from their product.
KayJanuary 27, 2019 at 6:03 am
I first started out with the Skyla iud in 2017, I had it for a year and my periods were terrible !! Once I stopped bleeding for months , I got my period regularly and it lasted 2 weeks every month. In October 2018 , I was recommended to switch my iud to the mirena. Worse decision of my life !! The doctor put it in wrong the first time and the second time she put it in she struggled for a good 15 m[Show More]I first started out with the Skyla iud in 2017, I had it for a year and my periods were terrible !! Once I stopped bleeding for months , I got my period regularly and it lasted 2 weeks every month. In October 2018 , I was recommended to switch my iud to the mirena. Worse decision of my life !! The doctor put it in wrong the first time and the second time she put it in she struggled for a good 15 minutes to find it....that same month i got the iud , I discovered a lump which I’ve been told is my lymph node in my “groin area”. I went back & forth to my obgyn from October-December , I think in mid November or early December I discovered the second swollen lymph node in my groin area. The obgyn could not tell me what it was , on January 28th I switched obgyn’s again and now I have a Bartholin cyst in my uterus which is painful asf btw and two swollen lymph nodes. After reading so many horror stories I am ready to just be done with birth control all together !
MoiraOctober 15, 2018 at 4:59 am
I had Mirena put in 7 months ago and started having some weird tingling and shocking pains in my wrists and neck/shoulders the same day. My doctor didn't believe me and told me to give it a few months. At first the cramps were bad, there was a lot of pain and bloating but things seemed fine for the first month while I was mostly taking Ibuprofen every day. Then, I started to stink. The cramps beca[Show More]I had Mirena put in 7 months ago and started having some weird tingling and shocking pains in my wrists and neck/shoulders the same day. My doctor didn't believe me and told me to give it a few months. At first the cramps were bad, there was a lot of pain and bloating but things seemed fine for the first month while I was mostly taking Ibuprofen every day. Then, I started to stink. The cramps became worse than ever, I became anxious and extremely emotional. My libido was nonexistant, but that may just be because all of the side effects were making me feel disgusting. If I did ever have sex, I bled and cramped a lot afterward. After a few months my knees started hurting during and after runs. Then my left knee became so bad that it's hard for me to lower myself onto a toilet, or lift my leg up onto the bed. I can walk and stand but can't bend or get up easily. If I need to kneel down at work, I cannot get myself up without assistance (it's embarrassing). Now I'm in my 7th month with this IUD. I have an appointment in a few weeks to get it removed even though my doctor doesn't think these things are related. In the past two months I've started experiencing numbness/tingly in my feet and hands. Pain in my back, neck and shoulders. Soreness in the tendons behind my knees and in my forearms and terrible pain in my knees. I also have pain in my finger joints when writing by hand, sewing, or typing. I got bloodwork done just in case and everything looked good except for a slightly low vitamin D3 level, so I started taking multivitamins with more Vitamin D but nothing has changed.
ShannonOctober 7, 2018 at 9:01 am
I got the marina in March 2010 a few months went by no issues. The at my 6 month mark I felt a stabbing pain on my right side. Went to the ob and was told there's nothing wrong. The pain continued and my period started to get heavier, so I went to another dr they said the same thing. I found out I had cyst on my ovaries in 2012. But yet another dr told me that it was fine and normal. When I got [Show More]I got the marina in March 2010 a few months went by no issues. The at my 6 month mark I felt a stabbing pain on my right side. Went to the ob and was told there's nothing wrong. The pain continued and my period started to get heavier, so I went to another dr they said the same thing. I found out I had cyst on my ovaries in 2012. But yet another dr told me that it was fine and normal. When I got together with my fiance in 2013 he told me that he could feel the coil. That summer I was in bed the whole time. Went to see yet again another dr and they again told me that it was in place. So I dealt with this pain till March of 2015 when removed it was inbeadded in my uterus. He just ripped it out. I bleed for 3 months after it was finally removed. The pain was still there but even worse. Around my 4 month after finally getting it removed I found out I was pregnant with my son. The pain was still there but not as severe. After my son was born the pain came back full force and I was diagnosed with pcos. Been treating this for 2.5 years. I didnt have any issues before I got the marina. I just found out that I might have endrmetroices too. I'm scheduled for a historectimy in November. Praying this takes away this pain 9 years of dealing with this has caused me to have severe anxiety, severe depression,anxiety, migraine headaches have gotten a lot worse,and PTSD due to the pain, and other things that has happened to me. I want to know how I go about getting involved with this lawsuit. I'm tired of this pain. I cant work, bearly able to do normal tasks.
CrystalAugust 23, 2018 at 5:32 pm
I started Mirena right after my son was born in April 2008; May 2010, I noticed a lump in my throat and was diagnosed with Thyroid Cancer in October 2010. I was never made aware of any negative side effects from this contraceptive. I had my thyroid removed and have been on several types of medications ever since. The side effects from this have been a lot to deal with. It's unfortunate that the[Show More]I started Mirena right after my son was born in April 2008; May 2010, I noticed a lump in my throat and was diagnosed with Thyroid Cancer in October 2010. I was never made aware of any negative side effects from this contraceptive. I had my thyroid removed and have been on several types of medications ever since. The side effects from this have been a lot to deal with. It's unfortunate that the warnings are out there.
TeresaJune 17, 2018 at 2:28 am
I am 26 years old. Just had my second iud removed 3 days ago due to hair thinning. I was and am still not sure if it is the leading cause but when I googled "mirena with hair thinning" it was all over the internet. Not to mention I saw a derm and was told my cause did not seem to be one from hereditary. I had the mirena for 9 years. I also suffered constant discomfort in my abdominal area through[Show More]I am 26 years old. Just had my second iud removed 3 days ago due to hair thinning. I was and am still not sure if it is the leading cause but when I googled "mirena with hair thinning" it was all over the internet. Not to mention I saw a derm and was told my cause did not seem to be one from hereditary. I had the mirena for 9 years. I also suffered constant discomfort in my abdominal area throughout the years but really never put two and two together until now that my stomach actually feels normal. During my first yr with the iud I brought up the discomfort to my obgyn and she ordered a sono where she noticed cysts that "erupt when I ovulate, but are small", that the discomfort was normal, so I started to think maybe I was just over thinking it. Another kid at my age was not an option so I stuck it out. At 19 I started to notice hair thinning and never associated it with the mirena until 6 years later that it is noticeably worse and no doctor can explain the reason behind it. I also experienced constant joint pain and mood swings with it but I'm not certain if the mirena is to blame. All in all I loved my Mirena IUD, up until my thin hair took a toll on my self confidence. I just hope I don't end up with PCOS.
DanielleMay 5, 2018 at 11:39 pm
I had the mirena put in after my first child when I was 19. I had it removed two years later due to pain, and it came out just fine. For about 4 years, I suffered through horrible cramps, extremely heavy and inconsistent periods, and painful intercourse. Finally fed up with all of this, I told my doctor he absolutely needed to check me out, and I refused to take no for an answer. Come to find out,[Show More]I had the mirena put in after my first child when I was 19. I had it removed two years later due to pain, and it came out just fine. For about 4 years, I suffered through horrible cramps, extremely heavy and inconsistent periods, and painful intercourse. Finally fed up with all of this, I told my doctor he absolutely needed to check me out, and I refused to take no for an answer. Come to find out, I had developed PCOS, which I had never had prior. A few months after this discovery, I was having more pain than usual, and scheduled an appointment. Two weeks later, I am having my left ovary removed at the age of 25. The PCOS, which my doctor said was likely caused by having had the mirena, had developed a cyst so large, it engulfed my entire ovary. I am now 27, I have only one ovary left, and I'm experiencing the same issues all over again. My doctor told me when he removed my ovary that it was highly likely it would happen to the remaining one. Because of something I was told was safe, I will now how to suffer through this pain until they remove my remaining ovary, after which I will be forced to go through menopause long before I should have.
ReginaSeptember 28, 2017 at 1:43 pm
Had Mirena put in and with no change in diet, I put on 60 pounds in 6 months. Prior to Mirena my weigh had stayed at a steady number. My Doctor said it was not the IUD. I have not gained any more weigh since I had it removed. The wost hing is I have not lost the 60 pounds either. I had a gastric bypass. I can not eat large volume of food. I am also fighting an autoimmune disease!
laurelSeptember 23, 2017 at 2:47 pm
I had the Mirena placed 8 weeks post-partum. WIthin 3 weeks I was suffering from unmanageable heart burn, bouts of nausea, diarrhea. Within 6 weeks I was unable to eat. I struggled to keep down food and even water. I was so nauseous I would throw up stomach bile all through out the day and be up with diarrhea in the middle of the night. I had sever fatigue, dizziness, and felt unbalanced and clums[Show More]I had the Mirena placed 8 weeks post-partum. WIthin 3 weeks I was suffering from unmanageable heart burn, bouts of nausea, diarrhea. Within 6 weeks I was unable to eat. I struggled to keep down food and even water. I was so nauseous I would throw up stomach bile all through out the day and be up with diarrhea in the middle of the night. I had sever fatigue, dizziness, and felt unbalanced and clumsy. I could barely pick up my son- every task was exhausting. It was hard for me to concentrate and I felt like I was in a permanent state of fogginess. I lost 30 lbs in 3 months. Eventually getting down to a mere 93.5 lbs. I made an appointment to have the Mirena removed and my symptoms gradually got better over a period of 2 months. I struggled to gain back weight and feel normal again. The Dr who removed my Mirena had blood tests done. My liver was showing signs of extreme failure. After multiple ultra-sounds, an endocrinologist, gi doctor and internist I still don't have a definitive answer to my problems but have had many things ruled out. Eventually my liver blood enzymes returned to a normal, non-failing result. Unfortunately I am now showing signs of auto-immune disorder. All of this occurred directly after the Mirena insertion and improved directly after its removal. Use extreme caution with this IUD. It is dangerous!
KarenAugust 18, 2017 at 12:48 am
I had the Merina put in October 2015, after I had it put in I started having pain in my lower left side, I saw my OB doctor about it several times he said there might be a cyst on my ovaries and it should go away on its own, the pain wouldnt go away so i went back he said maybe it was an infection he gave me antibodicts didnt work, he then did a pelvic ultrasound and a vaginal ultrasound and di[Show More]I had the Merina put in October 2015, after I had it put in I started having pain in my lower left side, I saw my OB doctor about it several times he said there might be a cyst on my ovaries and it should go away on its own, the pain wouldnt go away so i went back he said maybe it was an infection he gave me antibodicts didnt work, he then did a pelvic ultrasound and a vaginal ultrasound and didn't find anything wrong. so finally in November of 2016 I had him remove it, it hurt so much he looked surprised when it came out and said it was because it had attached itself to the wall of my uterus. I've had pain daily in the same spot on my left side ever since. haven't been able to exercise the same because it'll start hurting to much, it hurts if I lay certain ways. I definitely wouldn't recommend getting one.
ReynaJuly 20, 2017 at 8:23 am
Okay so I had my son now 3 yrs old June 2014. I got the Mirena put in at my six week check up!!!! I bled alot almost for a week I couldn't bend over because I would feel poking! That was for like 2 weeks. Eventually it stopped. 1yr. 1/2 later I got pregnant with the Mirena!!!! Mu Dr said maybe it fell and I didn't notice it. So around 5 months I got a ultrasound and it didn't show I had it so I t[Show More]Okay so I had my son now 3 yrs old June 2014. I got the Mirena put in at my six week check up!!!! I bled alot almost for a week I couldn't bend over because I would feel poking! That was for like 2 weeks. Eventually it stopped. 1yr. 1/2 later I got pregnant with the Mirena!!!! Mu Dr said maybe it fell and I didn't notice it. So around 5 months I got a ultrasound and it didn't show I had it so I thought okay maybe while I'm delivering it will appear. But I was wrong no IUD now my baby is 7months, and for the past 2 months I haven't been myself!!!! I have really bad mood swings, headaches, dizziness, some memory loss, Lower pain, pain on one side, and lower back pain. I have to wait until September to go into the Dr.
ElizabethJuly 2, 2017 at 2:41 pm
Hello I'm 50 years old been on mirena twice for 8 years total starting back in 2005 and again in 2012. When I first got on mirena it was for birth control, I was a very healthy, active woman no health problems. Then after the insertion came weight gain, depression, high blood pressure, headaches, acne, brain zaps (mini seizures), insomnia and 3 years later, miscarriage. I didn't know I was pregnan[Show More]Hello I'm 50 years old been on mirena twice for 8 years total starting back in 2005 and again in 2012. When I first got on mirena it was for birth control, I was a very healthy, active woman no health problems. Then after the insertion came weight gain, depression, high blood pressure, headaches, acne, brain zaps (mini seizures), insomnia and 3 years later, miscarriage. I didn't know I was pregnant. I had it removed on the 2010 and was put on birth control pills,I didn't stay on them but I noticed my periods was extremely heavy to the point of no sleep, using bladder control pads, exhaustion,I couldn't go anywhere, I even slept with towels under me if I could sleep. So after months of this happening I told my doc and she suggested mirena back in 2012,I took it in..... Again. Later that year I ended up in er with extreme pain that even morphine couldn't stop turns out I had ovarian torsion, if I didn't have the emergency surgery I could've died but they didn't take out the mirena. A year later I had my first bout with gout. No matter how much water I consumed I was dehydrated plus I was taking hbp pills. 2015 extreme hbp highest was 220/100, almost died again, still on mirena. I told the doctors I'm on it and I want it out. I was ignored. On the pamphlet it says if the patient has extreme high blood pressure remove the mirena, nothing. I had to beg to get it out!! It was taken out and I was back to the extreme heavy bleeding again. So on March 20,2017, I had a hysterectomy. Thank you Bayer for ruining my life. P.S I also have kidney stones.
TiffanyJune 18, 2017 at 2:47 pm
I am a 42 year old who had 2 Mirena's in a 10 year time span. Two years ago I began to have hot flashes (thought I was having a heart attack). Testing revealed I was postmenopausal and was diagnosed with atrial fibrillation (irregular heart rhythm). This was a shock because I was in physically good health and had no prior warning as perimenopause. In the last two years, I have gained middle weight[Show More]I am a 42 year old who had 2 Mirena's in a 10 year time span. Two years ago I began to have hot flashes (thought I was having a heart attack). Testing revealed I was postmenopausal and was diagnosed with atrial fibrillation (irregular heart rhythm). This was a shock because I was in physically good health and had no prior warning as perimenopause. In the last two years, I have gained middle weight, suffer with hot flashes, joint pain and extreme fatigue. I had no reproductive problems prior to the Mirena and had 2 healthy pregnancies with no trouble getting pregnant and regular mens cycles every 28 days since being a teenager. Something has affected my body and think that the Mirena was the culprit! I have been suffering tremendously for the last two years. I cannot believe so many others have similar symptoms!
PaulaApril 23, 2017 at 1:37 pm
What about cardiac arrythmia? I expericienced this and has been an ongoing problem since my second mirena insertion. I have found many women in forums with the same complaint. Thousands of dollars in exploratory surgeries to find the cause but nothing. I even have an implanted loop recorder now. I'll never know if I'm actually having a heart attack because the symptoms are the same. Recently, a ne[Show More]What about cardiac arrythmia? I expericienced this and has been an ongoing problem since my second mirena insertion. I have found many women in forums with the same complaint. Thousands of dollars in exploratory surgeries to find the cause but nothing. I even have an implanted loop recorder now. I'll never know if I'm actually having a heart attack because the symptoms are the same. Recently, a new gyno was surprised that my cardiologist had not recommened mirena removal as it was a known issue yet I have found no warning to this effect.
josieMarch 30, 2017 at 9:55 pm
My daughter who is now 23 yrs old had Mirena insertion about 2 years ago. She's relatively in good health but recently from her PCP routine visit they found that her kidney functions was about 30% due to the high creatnine level. A kidney biopsy was ordered along w/ many blood works but doctros found nothing unusual. She doesn't take any type of medication and nothing has change in past 3 yrs exce[Show More]My daughter who is now 23 yrs old had Mirena insertion about 2 years ago. She's relatively in good health but recently from her PCP routine visit they found that her kidney functions was about 30% due to the high creatnine level. A kidney biopsy was ordered along w/ many blood works but doctros found nothing unusual. She doesn't take any type of medication and nothing has change in past 3 yrs except for the Mirena insertion. Nephrologist suspected acute insterstitial nephritis so she was prescribed to take high dose steroids. It has been almost 3 weeks on high dose but labs shows kidney functions to be the same. Since the diagnosis was inconclusive our next step to have her undergo another biopsy and see if there's anything we missed. I strongly believe that this could have been from the Mirena for there was nothing else that she've done differently.
BarbaraMarch 25, 2017 at 5:44 pm
I'm on the 2nd mirena and I've had elevated high cholesterol, severe pain in my back, severe swelling in my left leg and constant burning as well, unexplained knee burning, insomnia, and now full post menopausal result. I've had several test to explain the leg swelling but nothing found for the cause of it. I'm wondering if the mirena side effects are the cause of my pain. I've also lost my sex dr[Show More]I'm on the 2nd mirena and I've had elevated high cholesterol, severe pain in my back, severe swelling in my left leg and constant burning as well, unexplained knee burning, insomnia, and now full post menopausal result. I've had several test to explain the leg swelling but nothing found for the cause of it. I'm wondering if the mirena side effects are the cause of my pain. I've also lost my sex drive, have mood swings, have had 3 fractures within the time frame of having the mirena, lack of energy to do daily routines, vision impairment, no periods for 10 years, weight gain. I'm due to get the mirena out this year. I've requested to get Pap smears done while on the mirena but was told I couldn't get one for 4 years due to the recommendation of the hospital policy. I've had to take sleeping pills, diet pills, water pills to help counter the side effects. I'm really concerned on what issues I will encounter once it comes out in ages months.
SylviaJanuary 1, 2017 at 10:24 pm
I had the mirena placed about 7-8 weeks after my second child. The first month was fine. Then the second month came the painful intercourse and just to touch my abdomen was torture, i was always In so much pain. Always felt bloated, had hot flashes, nauseand headaches. I went to my OBGyn, about 2 months after she inserted it, To figure out what was going on. During the exam she asked if I wanted i[Show More]I had the mirena placed about 7-8 weeks after my second child. The first month was fine. Then the second month came the painful intercourse and just to touch my abdomen was torture, i was always In so much pain. Always felt bloated, had hot flashes, nauseand headaches. I went to my OBGyn, about 2 months after she inserted it, To figure out what was going on. During the exam she asked if I wanted it removed and I said yes. She was not able to find it during the exam and wanted me to schedule ultrasound to see if it had moved. During this visit I left urine sample to rule out infection. And this is where I found out I was pregnant again. She explained the risks of ectopic pregnancy now bc of Mirena is still somewhere in there and they needed to get it out. She said if I felt any pain to go to ER. The next morning sure enough I was In the ER. Pelvic pain that dropped me to my knees while taking a shower. Did ultra sound and immediately after I was being prepped for emergency surgery. My Fallopian tube burst and I was having internal bleeding. I lost the baby and my right Fallopian tube, during surgery they also removed my IUD. It has been one year now and I have not been able to get pregnant and I'm still having Pain and irregular periods. I have never had problems getting pregnant before, I've never had irregular periods before the Mirena. I have always been healthy and now I'm not sure if I'll ever get pregnant again.
CarlaNovember 8, 2016 at 10:30 am
I had the mirena inserted in spring 2008. I was a year into it when my problems started. Episodes of low blood sugar, extreme fluctuations in fluid retention (8-10 pounds in a day). I had to go to the emergency room because I couldn't breathe. I was diagnosed with pleurisy. The doctor didn't believe that I was 10 pounds lighter the day before. I now believe it was a fluid issue around my lungs[Show More]I had the mirena inserted in spring 2008. I was a year into it when my problems started. Episodes of low blood sugar, extreme fluctuations in fluid retention (8-10 pounds in a day). I had to go to the emergency room because I couldn't breathe. I was diagnosed with pleurisy. The doctor didn't believe that I was 10 pounds lighter the day before. I now believe it was a fluid issue around my lungs. About 2 years into the iud my breasts became so severely sore they were untouchable. At that time I cold not feel the strings of the iud anymore. I went to the doctor and described all of my problems and was told everything was fine. No one could explain the sore breasts that were continuous. I took pregnancy tests for a few months that were all negative and then just believed I was crazy. I started bleeding at the end of November. By this point I was over going to seek medical help because I had been told so many times nothing was wrong with me. On December 28th, 2010 I took a pregnancy test and it was positive. I was told to go to the ER. At this point I had been bleeding for more than 5 weeks. The ultrasound showed something on my tube. I was taken into emergency surgery where they looked to see if I had an ectopic pregnancy and took out the iud. When I woke up the first feeling I had was that my breasts were not sore for the first time in 9 months! I lost that baby. My body bled for about 8 more weeks. My period was never "normal" again, but I was pregnant by June. After that pregnancy (I was 34). My body never returned to normal and was told I was in the early stages of menopause. I now have periods for 10 days out of every 21 on a good month. I truly believe the mirena ruined my body. I would tell any person that is not done having children to not put it in their body.
AmberSeptember 30, 2016 at 7:05 pm
Hello, My name is Amber and I got my Mirena put in July 2012 when I was in the Marine Corps because I had very bad, heavy and painful periods. When I go it put in it hurt because I never had kids but after a few month my periods just stopped which I was told was one of the side effects. Dont get me wrong I love not having a period. Occasionally I will get severe cramps tho and it sucks because it [Show More]Hello, My name is Amber and I got my Mirena put in July 2012 when I was in the Marine Corps because I had very bad, heavy and painful periods. When I go it put in it hurt because I never had kids but after a few month my periods just stopped which I was told was one of the side effects. Dont get me wrong I love not having a period. Occasionally I will get severe cramps tho and it sucks because it feels like im being stabbed in my lower abdomen. I have also had a a severe drop in my libido and I really dont want sex at all. When I do it is extremely painful no matter the position. Since I got it in I have also been having moderately severe vertigo. Ill loose all hearing in my left ear and Ill get a long-lasting migraine with dizziness and nausea. The doctors cant figure out what is wrong. I have gone for check ups recently since I go out of the Corps and they cannot find my Mirena now. I was told it may have fallen out but I am waiting for an appointment for an ultrasound. I really hope this isnt the cause of my constant fatigue either. I really wanted to get it replaced for another 5 years coming this July but Im not sure if i should now. What do you guys think I should do??
amiAugust 5, 2016 at 9:03 pm
Had the mirena coil put in August 2011 for 5 yrs. Noticed symptoms first a year later after insertion. Always felt nausea and fatigue. Have two children so still constantly on the go but felt exhausted and like a zombie. As time went on became more irritable and developed anxiety. Snapped at the slightest of things and felt overwhelmed eventually being like this for a few years i went to my G[Show More]Had the mirena coil put in August 2011 for 5 yrs. Noticed symptoms first a year later after insertion. Always felt nausea and fatigue. Have two children so still constantly on the go but felt exhausted and like a zombie. As time went on became more irritable and developed anxiety. Snapped at the slightest of things and felt overwhelmed eventually being like this for a few years i went to my GP and was put on Fluoxetine i think it is like prozac it was supposed to help with mood swings and anxiety. I just didnt understand why i became like this. I am quite a calm and relaxed person. I eat healthy most of the time. I dont drink or smoke. Before the coil i didnt find it very hard to lose weight but i noticed with the coil it would take so long and practically not eat anything. if i didnt exercise for the week i would gain about 4lbs. My stomach was always bloated and when i told the nurse who removed it said it can cause bloating. It has only been a few days it as been removed but I do feel more energetic. To sum up my symptoms, weight gain, reddish face looks light rash, anxiety, mood swings, fatigue, nausea and had to be put on medication. After reading others symptoms i would never have it again!
MichelleJuly 24, 2016 at 6:43 pm
In 2007 I had it placed, within six months I had extreme fatigue, very pale skin, extremely painful inner course, finally I went to the ER, I was in so much pain, hurled over in a fetal position. They did a ultra sound, the Mirena was all the way over to the right side of my uterus, along with a lot of scar tissue that they could see. They said the scar tissue was most likely from the mirena movin[Show More]In 2007 I had it placed, within six months I had extreme fatigue, very pale skin, extremely painful inner course, finally I went to the ER, I was in so much pain, hurled over in a fetal position. They did a ultra sound, the Mirena was all the way over to the right side of my uterus, along with a lot of scar tissue that they could see. They said the scar tissue was most likely from the mirena moving around so mucg.They weren't able to remove the Mirena at the ER. I had to wait for appointment with my OBGYN. He removed it and asked if I wanted a new one I firmly said no. Since then my periods are irregular, I have scar tissue that grows out side of my uterus, my new OBGYN says it from mutilation from inside of my uterus. When I seen the law suits advertised on TV I think in 09 I contacted them and I told them what happened and they said I don't quifiy for a law suit. Everyday I'm in pain. Please ladies don't ever get a IUD.
ElieJuly 22, 2016 at 12:26 pm
I GOT THIS AFTER HAVING MY CHILD APRIL OF 2014. I HATED THIS BC. WAS ALWAYS A PROBLEM EVEN WITHOUT INTERCOURSE. I was only 17 at the time and I felt very uncomfortable with it . Then I couldn't feel it so I had to go to the doctors and they didn't feel it. I guess it fell out on its own! Even when it was still in I had very painful sex and still do to this day. I still have the same partner so not[Show More]I GOT THIS AFTER HAVING MY CHILD APRIL OF 2014. I HATED THIS BC. WAS ALWAYS A PROBLEM EVEN WITHOUT INTERCOURSE. I was only 17 at the time and I felt very uncomfortable with it . Then I couldn't feel it so I had to go to the doctors and they didn't feel it. I guess it fell out on its own! Even when it was still in I had very painful sex and still do to this day. I still have the same partner so nothing has changed. This is ruining my life the pain is actually very painful every single time. I hate this birth control. I feel as if it's ruined my insides forever.
ellenMay 26, 2016 at 3:37 pm
I am having a very similar experience - enlarged pituitary gland, weight gain, exhaustion al lthe time, Want this thing OUT!!!
NicoleMay 18, 2016 at 4:20 pm
I had the mirena loud for about 2 years. I barely had any periods and my abdominal was so swollen , I was wearing sweats and stretchy short due to it. I suffered for infection after infection. So I finally had it removed Oct 2013... after it was removed , I had a very heavy period for about 8 days, then I was back to wearing my size 14 jeans. I went from an 18 to14 in 8 days. Crazy right? On Vale[Show More]I had the mirena loud for about 2 years. I barely had any periods and my abdominal was so swollen , I was wearing sweats and stretchy short due to it. I suffered for infection after infection. So I finally had it removed Oct 2013... after it was removed , I had a very heavy period for about 8 days, then I was back to wearing my size 14 jeans. I went from an 18 to14 in 8 days. Crazy right? On Valentine's day 2014 ,4 months later I found out I was pregnant. She was born Oct 2014. Out of three kids , she was the only one born with ear tags. My ex sister in laws daughter was conceived about three months after her iud was removed , and she was born with the same ear tags. Coincidence? I don't think so...
TiffanieFebruary 19, 2016 at 3:47 pm
I had Mirena placed in July 2014. By August I had gained 10 pounds (which continued every month for about 5 months) and started feeling seriously depressed had NO energy. By February of 2015 I finally decided to get blood work done and it was discovered that my thyroid was seriously malfunctioning to the point that I could have slipped into a coma. TSH should be 0.3-3.0 but mine was 175. They imme[Show More]I had Mirena placed in July 2014. By August I had gained 10 pounds (which continued every month for about 5 months) and started feeling seriously depressed had NO energy. By February of 2015 I finally decided to get blood work done and it was discovered that my thyroid was seriously malfunctioning to the point that I could have slipped into a coma. TSH should be 0.3-3.0 but mine was 175. They immediately put me on medication. At which point my Endocrine specialist put me on too high a dose. I ended up with a TSH of 0.007 and a heart condition called hashitoxicosis. After all I have read online I'm beginning to think the start of all these health issues was Mirena.
JuliaDecember 15, 2015 at 9:41 am
I am 35 yo, up at 2am with night sweats as I type this after Mirena removal 8 months ago. Had two of these devices in from 2008-2012 and 2012-early 2015. I had it removed because of reading about health risks associated with long term use and break through bleeding with the second one. I'm convinced the second one caused a cervical erosion or ectropion which has lead to over a year of post-coit[Show More]I am 35 yo, up at 2am with night sweats as I type this after Mirena removal 8 months ago. Had two of these devices in from 2008-2012 and 2012-early 2015. I had it removed because of reading about health risks associated with long term use and break through bleeding with the second one. I'm convinced the second one caused a cervical erosion or ectropion which has lead to over a year of post-coital and breakthrough bleeding. I'm feeling so worried that my hormones are permanently out of whack because of this device. I too experienced feeling severe unexplained depression, loss of libido, and anxiety when I had the device. That is all much better now, but hopefully not because I'm perimenopausal. Doctor has suggested hysterectomy and synthetic hormone therapy, or birth control pills none of which I am very thrilled about. I've paid $1,000 so far to diagnose what they thought was cervical cancer but are actually permanent cervical changes brought on by Mirena. If I have to do a hysterectomy, who knows how much that will cost. If you are reading this, do NOT get this device. There are thousands of women regretting buying in to the Mirena craze. Remember-- If it seems to good to be true, it usually is.
aliciaNovember 2, 2015 at 4:29 pm
I will never get this this again or recommend it. I had it in for only 5 month after having my 3rd child. I went to the er with stomach pain and because I had the iud they didn't do a pregnancy test and just said I must have a viral infection and left it at that. I ended up calling my doctor about a month later with the same pains my doctor did a mess of test and called about a hour later to tell[Show More]I will never get this this again or recommend it. I had it in for only 5 month after having my 3rd child. I went to the er with stomach pain and because I had the iud they didn't do a pregnancy test and just said I must have a viral infection and left it at that. I ended up calling my doctor about a month later with the same pains my doctor did a mess of test and called about a hour later to tell me i was pregnant and made a appointment to get the iud removed. Then 2 days later with the pain getting worse i called my doctor again to see if we could do it sooner thedoctor on call told me to go to the er right away so after work I went. They did both ultrasounds in which I heard the heart beat. Then the er doctor came in told me I was 2 months 2 months now remember I only had the iud for 5 months anyway and they said I had to have emergency surgery cause the baby was in my tube and it could rupture at any moment. So in the end I had surgery where they had to remove my whole tube abort the baby and oh yea I'm allergic to the meds to knock you out so I stopped breathing during surgery. Never again.
ChristinaSeptember 25, 2015 at 12:03 pm
I had the mirena put in 4-5 months ago. Since I have had spotting most of the month, never sure when I'm having a period or getting a period because I'm much of the month bleeding. I have tingling, numbness and pain in several parts of my body including my hands, feet, knees, nose. My balance is off when I walk because my feet are numb. I can barely drive can't grasp the steering wheel because my [Show More]I had the mirena put in 4-5 months ago. Since I have had spotting most of the month, never sure when I'm having a period or getting a period because I'm much of the month bleeding. I have tingling, numbness and pain in several parts of my body including my hands, feet, knees, nose. My balance is off when I walk because my feet are numb. I can barely drive can't grasp the steering wheel because my hands hurt so bad or are numb. I'm a hairstylist/ Esthetician I use my hands for everything but I drop my tools because everytime I use my hands they fall asleep. I cannot pick up my children because I am too weak. I've had blood work for every test I can have them run only a vitamin D defiency has shown up which I've never had before. I have had X-rays in my neck and back. Have an upcoming internal medicine appt. Was referred to physical therapy by my dr. Asked if it could be the mirena one dr. Told me I have no idea if it's mirena related he ddnt knw much about the device. I complained to another about my spotting and bleeding all the time she said that was normal with mirena it could take 6 mos to regulate. I just want my body to function right am afraid I'll be in a wheel chair soon. I have an appt. In a week to have the mirena removed.
KennedyAugust 3, 2015 at 9:57 pm
Has anyone every had a stroke form the IUD? I was just released form the hospital Friday because I had a stroke that ultimately caused me to have a seizure. I am only 24, very healthy; no risk factors for stroke or seizures. They took some blood and had it sent off to make sure I don't have some type of blood disorder. I was also told I needed to get my IUD removed ASAP; does anyone have any adv[Show More]Has anyone every had a stroke form the IUD? I was just released form the hospital Friday because I had a stroke that ultimately caused me to have a seizure. I am only 24, very healthy; no risk factors for stroke or seizures. They took some blood and had it sent off to make sure I don't have some type of blood disorder. I was also told I needed to get my IUD removed ASAP; does anyone have any advice...
StaceyJuly 9, 2015 at 10:15 pm
I had Mirena for 5 years and had no periods during that time. I had it removed a year ago, and my period never returned. I have now been told that I am menopausal! I am only 44! This device is the devil!
JenniferJune 12, 2015 at 8:46 pm
I had my Mirena inserted in January of 2005. I had a regular period 3 months following then only spotting at random times, then nothing. I had it 5 years longer than recommended because I had no insurance and removed was $300. I have been experiencing multiple symptoms of menopause and everyone told me I was too young it must be something else. Well Monday of this week I finally had it removed. I [Show More]I had my Mirena inserted in January of 2005. I had a regular period 3 months following then only spotting at random times, then nothing. I had it 5 years longer than recommended because I had no insurance and removed was $300. I have been experiencing multiple symptoms of menopause and everyone told me I was too young it must be something else. Well Monday of this week I finally had it removed. I addressed my concerns for menopause with my doctor. She order a test to measure FSH levels. I received my results today. I'm post menopausal at 33!!
StephanieJune 3, 2015 at 12:57 pm
I had the mirena IUD put in around 2004 and from the moment I got it I started having excruciating cramps, irregular heavy periods to just spotting, mood swings, fatigue, lack of patience and irritability and lost the urge to have sex and when I did it was very painful. I consulted doctor and was told the symptoms are "normal". I had it removed a year early because I could not tolerate the pain [Show More]I had the mirena IUD put in around 2004 and from the moment I got it I started having excruciating cramps, irregular heavy periods to just spotting, mood swings, fatigue, lack of patience and irritability and lost the urge to have sex and when I did it was very painful. I consulted doctor and was told the symptoms are "normal". I had it removed a year early because I could not tolerate the pain I was experiencing any longer. When I had the device removed it was very painful but came out with extra effort the first time with out surgery, luckily. I had a history of PID but doctors didn't seem too concerned with that issue when the IUD Was put in. Now, some 7 years later I am having very heavy periods with hemmoraging and clots every two weeks for 3 months then it subsided to 3-4 weeks in between cycles and my last one was actually 5 days late after waiting 31 days for period. (2 weeks ago). I am also experiencing a small protrusion or soft tissue mass on my left lower abdomen in pelvic area and fluttering or twitching maybe pulsating in that area constantly for last 2 weeks. I am concerned it could be cancer, cysts or fibroids or maybe bad scarring from IUD placement of 4 years. These could also be related to perimemopausal as well for I am 45. I am worried that the IUD could have caused ovarian cancer. I plan to see a doctor soon. Has anyone else experienced this and could I have a possible Lawsuit against mirena, I feel I was deceived and cheated out of 4 years of a "normal" healthy life because of their deception and now some years later am starting to experience the long term Effects of this device. Bad bad business ethics!
LaDonnaMay 7, 2015 at 4:30 pm
What about premature menopause brought on by this device? After having the device removed, I always suspected it sent me into premature menopause. I was barely in my 40s when it was removed, having had it placed after the birth of my daughter at 39. I've just researched this issue on the web, and you'd be amazed at the data and inquiries surrounding just this particular issue. I felt absolutel[Show More]What about premature menopause brought on by this device? After having the device removed, I always suspected it sent me into premature menopause. I was barely in my 40s when it was removed, having had it placed after the birth of my daughter at 39. I've just researched this issue on the web, and you'd be amazed at the data and inquiries surrounding just this particular issue. I felt absolutely horrible when I had the device -- always wondering if I was pregnant due to tender breasts, bloating, fatigue, nausea -- and still to this day have not returned to my pre-Mirena health. Only had the device for 4-1/2 years because I couldn't take it anymore, but period NEVER returned, not even spotting. I think this issue should be considered in the lawsuit claims because premature menopause is not just the absence of periods; there are more significant health issues involved -- higher risk for stroke, cerebral aneurysm, heart disease, osteoporosis, loss of teeth. Yep, this explains it.
DaniellaApril 13, 2015 at 11:05 pm
My name is Daniella Sandoval, i got the i.u.d Mirena on June 26, 2010 . i had gotten the five year plan , i had a lot of complications since , an hope i can still conceive after i get the surgery to get removed . I am scared do to the views an claims i have seen an hope since my five years is up my body is not as messed up as i think it is . please i hope someone can help.
JordanMarch 11, 2015 at 4:33 am
Hello, I'm 17 and I've been on the IUD since October of last year, when I first got it the pain was unbearable, I missed days of school, I cried, I didn't eat, I was in so much pain the only thing I could do was lay with a heating bad. I have been constantly bleeding and cramping, I'm exhausted, my hips hurt, my back hurts. I'm 17, I shouldn't hurt like this. I talked to my doctor she said my uter[Show More]Hello, I'm 17 and I've been on the IUD since October of last year, when I first got it the pain was unbearable, I missed days of school, I cried, I didn't eat, I was in so much pain the only thing I could do was lay with a heating bad. I have been constantly bleeding and cramping, I'm exhausted, my hips hurt, my back hurts. I'm 17, I shouldn't hurt like this. I talked to my doctor she said my uterus was still swollen and me being only 17 I believed her, she put me on some kind of pill that stopped the period for two weeks in December, then it came back, I called her and told her maybe I should take it out and she said no I just needed to regulate my bleeding. She put me on birth control pills on top of my IUD, my grandmother is concerned that this is going to hurt me and ruin my chances of having children, I don't know what to do anymore.
SaraMarch 8, 2015 at 4:23 am
I have been on the mirena for over 8 years. I actually had a second one put in. I've been battling weight, depression, and adrenal fatigue along with other concerning issues. I just discovered it's because of mirena. Shame on this product. There should be a shorter time limit on it. I'm so worried about how long it will take my body to recoup.
BrandiFebruary 19, 2015 at 6:31 am
I have had mirena for 5 years. First couPle of years were ok (especially due to not having periods) after that reoccurring ovarian cysts acne joint pain absolutely no sex drive SEVERE nausea abdomInal pain pain during intercourse depression anxiety Unable to lose weight. ( exercise all the time, always have) Breast cysts migraines this birth control is very effective not only because of the horm[Show More]I have had mirena for 5 years. First couPle of years were ok (especially due to not having periods) after that reoccurring ovarian cysts acne joint pain absolutely no sex drive SEVERE nausea abdomInal pain pain during intercourse depression anxiety Unable to lose weight. ( exercise all the time, always have) Breast cysts migraines this birth control is very effective not only because of the hormones , but from how wonderful it makes you feel, and because you never want to have sex. I have never suffers symptoms like this before mirena. I've been to the doc and e.r. countless times and I've given them the mirena theory and they treat my like I'm crazy I'm sorry but I know my body, and I know what my life was like before mirena. The worst decision of my life. I'm glad everyone is sharing their stories to get the word out. nobody should lied to and nobody should have to live like this !!
carolineDecember 16, 2014 at 2:47 pm
I have had the mirena out since 12-9-13 and its Bern a year now still no baby. I really hope one day I will be able to be a mother again.
autumnDecember 13, 2014 at 4:34 pm
Hi, im autumn and i got my iud Bck in 2009 well at first i didnt really have any problems until abt 2 and a half years later.. Well i started having wierd,annoying,pains in my top abdominal well i went to the er numerous times to make sure it didnt move well every time i went they would do a pap smear and test to make sure i didnt have any diseases and always came bck negative wich i already new [Show More]Hi, im autumn and i got my iud Bck in 2009 well at first i didnt really have any problems until abt 2 and a half years later.. Well i started having wierd,annoying,pains in my top abdominal well i went to the er numerous times to make sure it didnt move well every time i went they would do a pap smear and test to make sure i didnt have any diseases and always came bck negative wich i already new that but i also experienced very painful sex,loss of sex drive,fatigue,nausea,tender breast,,back pain,pelvic pain etc.. Well i had it removed abt a year later hoping that maybeall the trouble would stop well it didnt i still had the symptoms still but it is getting worse i have went to doctors plenty of times and they still can not understand why im having so much trouble so then they sent me to a nother doc to check if i had ovarian cycst supposedly i dont well then they put me on birth control for months said it would help with the discomfort when i have sex and increase my chances to have another child well it didnt help nor did i conceive and me and my husband have tried for years now with my first child i conceived him a month after i stop my bc some im pretty fertile... I just want answers for all the pain and stress it has caused me and i want a few more kids and i dont think i can have anymore.
AnnGSeptember 24, 2014 at 6:05 pm
The above sounds very similar to my issues since having the Mirena installed 12/2011 and removed 8/2012. I’ve been sick for four years now and still struggling. I lost 30 lbs in less than three months back in 2012 and continued to lose weight unexplained. I’m down 68 lbs, granted I was overweight to begin with and am thankful for that now. I had adrenaline rush/hot flush surges , nausea, and f[Show More]The above sounds very similar to my issues since having the Mirena installed 12/2011 and removed 8/2012. I’ve been sick for four years now and still struggling. I lost 30 lbs in less than three months back in 2012 and continued to lose weight unexplained. I’m down 68 lbs, granted I was overweight to begin with and am thankful for that now. I had adrenaline rush/hot flush surges , nausea, and fatigue. I saw the Gastroenterologist and all tests came back negative. I had tests done (cortisol), which caused me to be recommended to an Endocrinologist. I was diagnosed with hypothyroidism back in August 2013 and have had 3 CTs and a MRI of the pituitary gland to see if there were any issues with it. They found what they’re calling an anomaly, but nothing that requires further follow up. I was put on anti-nausea meds because of the nausea I get. My primary referred me to a holistic doctor to get another view on things. He ran a saliva test that revealed adrenal fatigue/exhaustion and premature ovarian failure. I’m showing the majority of the signs for Addison’s Disease. I was healthy before the Mirena was installed and haven’t been right since. It was a traumatic and stressful experience for me (I had adrenaline rushes w/hot flushes during the install & bled for 8 months; constant stress on the body/mind can cause the onset of adrenal issues). I struggle every day and have good and bad days. I now deal with ovarian cysts, back and pelvic pain, exhaustion, hair loss, lack of sex drive, can’t handle stressful situations, mood swings, and more.
AubreySeptember 23, 2014 at 6:29 pm
6 dr visits later, 3 ultra sounds, 1 MRI and multiple failed retrieval attempts, I am told it must of feel out. So now they need to xray my tummy to double check. What did they see in the ultra sounds then? and before when I came in on my very first appt more than 2 months ago for the replacement?
AshleySeptember 12, 2014 at 3:00 pm
I'm February 2014 my Mirena had fell out after being in for almost a year (April 2014) I called my doctor immediately after noticing it took two weeks to get an appointment. I went in and the doctor did an ultrasound and said that my Mirena was still there. Since I did not physically still have my Mirena with me there was nothing to prove it was not there. Now I am currently pregnant because of al[Show More]I'm February 2014 my Mirena had fell out after being in for almost a year (April 2014) I called my doctor immediately after noticing it took two weeks to get an appointment. I went in and the doctor did an ultrasound and said that my Mirena was still there. Since I did not physically still have my Mirena with me there was nothing to prove it was not there. Now I am currently pregnant because of all of this.
TiffanyAugust 31, 2014 at 5:18 am
I just got the Mirena put in on Wednesday August 27th. Since I got it put in, not even a week ago, I've been exhausted all the time, dizzy, nauseous, have had pain in my lower back, pain in my upper abdomen and sometimes in my sides. I've never felt like this before so I called the OB office and told them what was going on and they said "since you're not having and lower pelvic pain, it's not the [Show More]I just got the Mirena put in on Wednesday August 27th. Since I got it put in, not even a week ago, I've been exhausted all the time, dizzy, nauseous, have had pain in my lower back, pain in my upper abdomen and sometimes in my sides. I've never felt like this before so I called the OB office and told them what was going on and they said "since you're not having and lower pelvic pain, it's not the Mirena". I need to know if I should just get to removed now, before things get worse for me.
AmandaAugust 26, 2014 at 9:15 pm
I had the Mirena inserted in October 2010. At my last OBGYN visit the doctor could not see my strings, but did an ultrasound and claims IUD is still in my uterus. My recent blood work at my PCP revealed that I probably have an autoimmune disease because my ANA came back positive. She referred me to a rhumatologist for further testing.Many other women(according to internet research) also seem to ha[Show More]I had the Mirena inserted in October 2010. At my last OBGYN visit the doctor could not see my strings, but did an ultrasound and claims IUD is still in my uterus. My recent blood work at my PCP revealed that I probably have an autoimmune disease because my ANA came back positive. She referred me to a rhumatologist for further testing.Many other women(according to internet research) also seem to have auto immune issues since having the Mirena. I have had many odd symptoms, and I am now convinced that the Mirena is the cause.My symptoms include, ovarian cysts, acne, back and pelvic pain, tingling/numbing in various parts of my body, hair loss, lack of sex drive, constant fatigue, lack of patience, constant feeling of being overwhelmed, and extreme moodiness Prior to having the Mirena placed I never had any of these problems or side effects. I was healthy. I am currently 31 years old and am making an appointment to have this IUD removed!
DenishaAugust 26, 2014 at 3:10 am
Hi! After I had my son about a year ago 2013 , I had quickly made an appointment to the midwife to have a Mirena (IUD) put in. And after a year and a half I notice I was feeling some sort of cramps almost similar to contractions. So I went to the hospital and the doctors told that I had an affection , so I told them to take it out. After I still haven't got my period. I'm starting to wonder if so[Show More]Hi! After I had my son about a year ago 2013 , I had quickly made an appointment to the midwife to have a Mirena (IUD) put in. And after a year and a half I notice I was feeling some sort of cramps almost similar to contractions. So I went to the hospital and the doctors told that I had an affection , so I told them to take it out. After I still haven't got my period. I'm starting to wonder if something is seriously wrong because I can't get pregnant and I've tried many times and have token many test and each one always came back negative. After looking up answers I noticed some women were getting the same problems as me and they soon filed for a lawsuit. I'm at that point where I think the IUD messed up my uterus and maybe the infection that the mirena cause could be the to why I can't get pregnant , I'am at lost for words and need help right away!!! Because I am ready to conceive again but I don't know if I can try any more.
MelanieAugust 22, 2014 at 9:04 pm
I had the IUD inserted back in 2007, and I was in so much pain after then. I always had to lay down whenever the pain starts, I kept headaches, Lower adomen pain, it was very painful during and especially after intercourse. IUD has cause me so much pain while I had it in, til now it would not even become an option if I was asked again about receiving another one. I wouldn't recommend it to anyone [Show More]I had the IUD inserted back in 2007, and I was in so much pain after then. I always had to lay down whenever the pain starts, I kept headaches, Lower adomen pain, it was very painful during and especially after intercourse. IUD has cause me so much pain while I had it in, til now it would not even become an option if I was asked again about receiving another one. I wouldn't recommend it to anyone to use.
JessicaAugust 21, 2014 at 12:57 am
Hi I've also tried the marina ever since I've had it in I've had nothing but aerious problems. I'm 23 years old and i had a c-section on December 10, 2013 with my daughter in which i ended up getting a yest infection in my incision do to my allergic reaction to the staples. I ended up getting pergnant again right after in which i had to have an abortion do to medical issues. Which was April 16, 20[Show More]Hi I've also tried the marina ever since I've had it in I've had nothing but aerious problems. I'm 23 years old and i had a c-section on December 10, 2013 with my daughter in which i ended up getting a yest infection in my incision do to my allergic reaction to the staples. I ended up getting pergnant again right after in which i had to have an abortion do to medical issues. Which was April 16, 2014 and this same day i got the merina. Ever since then ive been having multiple problems like depression, short temper, constantly feeling confused emotions, abdominal pain on my right side, ive went to the clinic for a check up on my marina n explained everything to the physician and she told me to seek mental health and that the marina will not cause any of the problems i was have but yet i had a yest infection and a UTI Infection. And here i am now having chunks of meat that looks like pieces of raw vainy chicken pass every time i go pee i even had a chunck sitting on my underwear when i went to the restroom. Can someone give me advise as to what i should do please. I have 3 children, one thats going to be 3, a 1 1/2 year old and my baby thats 8 months and its really sucks feeling irritated All the time when i should be. So please give me advise because i was told i would be able to have the marina before 6months with medical reasoning or up to a year. Is that true?
Laura 13August 13, 2014 at 7:29 pm
At the time I got Mirena, I was 10 years old. I needed it for heavy periods and my doctor said it was completely safe, painless, and that I'd recover quickly. Fast forward to after the operation. I was in a lot of pain and felt very sick. I remember waking up crying before even getting in the recovery room because it hurt so badly. The pain was like severe period cramps that would never stop. I h[Show More]At the time I got Mirena, I was 10 years old. I needed it for heavy periods and my doctor said it was completely safe, painless, and that I'd recover quickly. Fast forward to after the operation. I was in a lot of pain and felt very sick. I remember waking up crying before even getting in the recovery room because it hurt so badly. The pain was like severe period cramps that would never stop. I had to stay in the hospital for 6 hours after the operation it was that bad... When I got home from the hospital, it wasn't really any better... In fact I never recovered from it. I was always in pain and couldn't eat because of it. Eventually my doctor said that the Mirena should be removed, as I had what may have been an allergic reaction to it. I got it removed, and I felt so much better and felt no pain for a change. Mirena ruined my life. If you'd like to know, I'm 13 as of now.
passion3flowerAugust 1, 2014 at 3:49 pm
There's a very informative site with lots of information on how to lose weight and get rid of the mirena iud side effects and find other natural birth control options. Check out: http://mirenadetox.com/mirena-weight-gain/ Her detox has also been immensely helpful to my recovery and I seriously recommend it
StephanieJuly 18, 2014 at 3:58 pm
I had the IUD for almost 3 years and for the first year it was great but then after that I gained 20 pounds I even tried a new diet to see if it would help but it didn't,I was really depressed to the point I didn't want to leave the house and I had horrible cramping all the time.I was in the hospital more then anything due to ovarian cystsand they also said I had a blood clot in the vein by my ova[Show More]I had the IUD for almost 3 years and for the first year it was great but then after that I gained 20 pounds I even tried a new diet to see if it would help but it didn't,I was really depressed to the point I didn't want to leave the house and I had horrible cramping all the time.I was in the hospital more then anything due to ovarian cystsand they also said I had a blood clot in the vein by my ovary.This is great if you like being in pain all the time.But after all that couple days ago it fell out and I didnt know it until I felt something weird and I went to the hospital and it was just sitting there inside.
TasiaJuly 15, 2014 at 8:53 pm
While using the Mirena I was always sick, depressed , sleepy ,and gained weight. I was always dizzy and nauseous which all came at one and I would always begin to vomit I was actually rushed to the hospital once and was later sent home because they could not figure out why I was having these symptoms. Time went by and I finally found a doctor that accepted my Insurance which was Medicaid and she r[Show More]While using the Mirena I was always sick, depressed , sleepy ,and gained weight. I was always dizzy and nauseous which all came at one and I would always begin to vomit I was actually rushed to the hospital once and was later sent home because they could not figure out why I was having these symptoms. Time went by and I finally found a doctor that accepted my Insurance which was Medicaid and she removed it. Upon removal I was in really bad pain and I asked why so she did a vaginal exam and sated my ovary was sitting on the Mirena and it was swollen which was the reason I always had pains. So she scheduled an ultrasound but I was given a run around from the clinic because it was a low cost clinic. Finally I went to a private PCP who rescheduled the ultrasound with a private facility. The day after having the ultrasound done I was rushed back to ER where I was in pain that I wouldn't wish on anyone I began all the symptoms of vomiting dizziness etc. again so eventually they rushed me into surgery due to my ovary being without blood flow for to long and I was also given a blood transfusion while hospitalized. I've never experienced anything of this sort before having the IUD placed inside of me and I'm relieved to no longer have this horrific thing inside of me any longer I've been great ever since getting it out.
NicoleJuly 10, 2014 at 12:53 am
I had the mirena for 6 years I was getting really bad infections to the point they wanted to do surgery so I had it removed, then I got pregnant after a year with my little girl who was born blind :"( I have no idea why this would happen to me I did everything right..
CrystalJune 18, 2014 at 6:57 am
I got mine in 2006. I had the miserable three months and then things started to be normal except for a extremely terrible pain in my right hip (and sometimes left hip). I had just been pregnant and so I though it was because of that. A doctor told me later, that the Mirena had caused all my grief. I also started gaining weight like crazy and I brought this up to the doctor. I was told the Mirena h[Show More]I got mine in 2006. I had the miserable three months and then things started to be normal except for a extremely terrible pain in my right hip (and sometimes left hip). I had just been pregnant and so I though it was because of that. A doctor told me later, that the Mirena had caused all my grief. I also started gaining weight like crazy and I brought this up to the doctor. I was told the Mirena had nothing to do with it. I thought that was just me too and so I started spending money on exercise training, natural supplements (which worked on everyone but ME, it seems). Seeing practitioners and trying to see what i could not lose weight. I even starved myself. NOW Mirena claims some people may have weight problems. This may not be important to you but, I lost a lot of money on this. Not to mention how it has hurt my lifestyle. People are not kind to those who are less than pretty/thin. I have been insulted to my face. This does not help me.
FeleciaJune 14, 2014 at 4:49 pm
I only had the IUD for 2&a half months. I had to get it removed because I was having really bad headaches, back pain, pains in my stomach, pelvic pain, anxiety, no energy, vaginal discharge, vaginal odor, mood charges. I also had to get it removed because it stated moving it self around. It was bad. I'm still having the pains after having it out for a couple days. I don't recommend this product to[Show More]I only had the IUD for 2&a half months. I had to get it removed because I was having really bad headaches, back pain, pains in my stomach, pelvic pain, anxiety, no energy, vaginal discharge, vaginal odor, mood charges. I also had to get it removed because it stated moving it self around. It was bad. I'm still having the pains after having it out for a couple days. I don't recommend this product to anyone.
BIOWAMay 21, 2014 at 4:46 am
I had the IUD for three years. No side effects and no pregnancies. Three months ago I had the following symptoms: sepsis kidney failure Fatty Live (I don't drink) Increase of bacteria in my blood and abdominal area OB told me it was due to the IUD and I need to have it removed asap. Then she told me it caused CYST. Low and behold: I have CYSTS on my ovaries. CA 125 levels suggest cancer. Why is [Show More]I had the IUD for three years. No side effects and no pregnancies. Three months ago I had the following symptoms: sepsis kidney failure Fatty Live (I don't drink) Increase of bacteria in my blood and abdominal area OB told me it was due to the IUD and I need to have it removed asap. Then she told me it caused CYST. Low and behold: I have CYSTS on my ovaries. CA 125 levels suggest cancer. Why is this not told to people? I trusted my doctors and my doctors trusted the FDA from researchers. Didn't people report this? I am very confused.
AShley GMay 6, 2014 at 7:17 pm
I have had my IUD for almost 2 years. During these 2 years I have had weight gain, anxiety, migraines, no sex drive, no energy, abdominal bloating pain, pelvic pain and heaviness, abnormal vaginal discharge, vaginal odor. I have cysts on both ovaries that seem to over grow and cause a lot of pain, lower back pain that radiates down my legs. In the past I spoke with my OBGYN's PA about all my sym[Show More]I have had my IUD for almost 2 years. During these 2 years I have had weight gain, anxiety, migraines, no sex drive, no energy, abdominal bloating pain, pelvic pain and heaviness, abnormal vaginal discharge, vaginal odor. I have cysts on both ovaries that seem to over grow and cause a lot of pain, lower back pain that radiates down my legs. In the past I spoke with my OBGYN's PA about all my symptoms and she states that Mirena has the lowest hormonal out put, that she does not see why it would cause all these problems, she has the IUD herself and does not have any problems. After that I figured it was just me until I researched this and started seeing other people with similar symptoms. I was just diagnosed with Group B Streptococcus in my urine. I have never had any issues until I had this IUD put in. I have an appointment to have the IUD removed before it causes in more permanent damage if it has not already.
LISAApril 22, 2014 at 11:39 pm
i got pregant with the iud, i was alomst 2 months when i found out, i was rushed to the hospial where they told me i had to get a er surgury, i fainted in the lobby because the pain was so unreal!! doctor said if i had waited even 5 mintutes, i would have died!!!!
CseaApril 13, 2014 at 8:59 am
hi im 21 turning 22 this year i had 3 csections in a row and with my last child the doctors tld me dae had to place a iud in me becoz i cant get pregnant again becoz its a big risk for me my new born is 3 mnths goin on 4 months nd i want to remve mine tomorrow becoz drs a copper string hanging from the outside of me i feel fine doe bt im confused i thort da iud wud b betr dn da jab bt im wrng hpe [Show More]hi im 21 turning 22 this year i had 3 csections in a row and with my last child the doctors tld me dae had to place a iud in me becoz i cant get pregnant again becoz its a big risk for me my new born is 3 mnths goin on 4 months nd i want to remve mine tomorrow becoz drs a copper string hanging from the outside of me i feel fine doe bt im confused i thort da iud wud b betr dn da jab bt im wrng hpe things al work out for me i thought it was juc me bein depressed nd cryin im to young to be goin thru dis shouldve sed no
BetsyMarch 31, 2014 at 9:31 pm
After reading these comments I see the side effects I experienced could have been much worse. However, after having the Mirena removed it is still giving me problems. My Mirena was inserted shortly after the birth of my second child, like most, I was told that it was a great form of birth control. In the begining I didn't experience much more than I was already warned about, discomfort and bleedin[Show More]After reading these comments I see the side effects I experienced could have been much worse. However, after having the Mirena removed it is still giving me problems. My Mirena was inserted shortly after the birth of my second child, like most, I was told that it was a great form of birth control. In the begining I didn't experience much more than I was already warned about, discomfort and bleeding. Eventually those symptoms wore off and for about 6 months my life was pretty accurately dipicted by the Mirena commercials. 6 months of easy periods was not a worthy exchange for what I suffered from the Mirena though. I started bleeding so heavily that I was on the verge of animia. My doctor advised that I should have the Mirena removed because I had developed endometriosis. After removing the IUD I continued to bleed and my gyno tried numerous treatment for the endometriosis, none of which worked. For nearly a year I suffered from the bleeding until finally I couldn't take it anymore (I was a personal trainer, which is not a condusive job for soaking a pad hourly) My doctor finally gave me 2 options, a hysterectomy or a surgery which cautorizes (sp) the lining of the uterous called ablasion (sp) I chose the surgery because I wanted to keep all of my parts and hoped to have more children. Slim chance, shortly after, I continued to have pain although the bleeding had stopped. I went back to the doctor and they discovered I had ovarian cysts. 3 years later and my husband and I still cannot concieve. So, yes, Mirena worked for me as a birth control. I would just suggest you remove all you lady parts instead of using it. I would imagine that would be easier.
deborahNovember 1, 2013 at 10:14 pm
im a very upset client of the i u d not being told all the risk that were involved as well as side effects , miss manning
NadineSeptember 24, 2013 at 4:08 pm
I got merina back in 2009 as it was pitched to me to be a great idea. since then I have had acne like crazy massive abdominal pain,hot and cold sweats my moods are all over the place. I still have it in and experience a few other things as well. I was told by my doctor at my last pap that it looked fine. I'm certain merina is to blame.
RosannaAugust 12, 2013 at 6:53 pm
I had my first daughter in 2008. Then had my second daughter in 2009. My girls are 11 months apart. I was consulted about the iud and had it for almost 2 years after my 2nd daughter was born. everything was great until one day about 2 years later I started having lower pelvic pain and pressure on my left side mainly but over all fullness in the lower pelvic area. I went to my doctor and they told [Show More]I had my first daughter in 2008. Then had my second daughter in 2009. My girls are 11 months apart. I was consulted about the iud and had it for almost 2 years after my 2nd daughter was born. everything was great until one day about 2 years later I started having lower pelvic pain and pressure on my left side mainly but over all fullness in the lower pelvic area. I went to my doctor and they told me everything looked fine after a normal Pap smear; no eyebrows were raised by them but I still had pain and a lump on my left side. The following week, I went back to my doctor and told them I wanted it removed. Immediately after removal I felt relief! the following week, I had sever lower abdominal, pelvic and back pain. I called my doctor back and they suggested maybe it was the hormones leaving my body! They told me I would be able to conceive again. It's been almost 2 years after removal and without any other birth control and not preventing pregnancy, I still have not gotten pregnant. I am now 27 years old and my girls are now 4&5. Two weeks ago I went to the Er for the same pain in my lower pelvic region with the same lump. After an ultra sound and a urine sample, they told me I had fiborous mass involving my ovary and uterus. It wasn't cancerous at this time but would need to have a regular check up every 6 months. I was also told I may not conceive again. I'm certain merina is to blame.
MaganAugust 8, 2013 at 4:03 am
I had mirena inserted in 2009 after the birth of my 2nd child. I never had a period. Maybe some LIGHT bleeding for 1 day...not a period! After suffering from the same horrible side effects as many of you, I had it removed and had my tubes tied. I was suicidal! I had to do something. I had my surgery a year ago and my periods still haven't returned. I am experiencing menopause symptoms. I'm 27...I'[Show More]I had mirena inserted in 2009 after the birth of my 2nd child. I never had a period. Maybe some LIGHT bleeding for 1 day...not a period! After suffering from the same horrible side effects as many of you, I had it removed and had my tubes tied. I was suicidal! I had to do something. I had my surgery a year ago and my periods still haven't returned. I am experiencing menopause symptoms. I'm 27...I'm convinced I'm in early menopause!
MariahJuly 10, 2013 at 8:09 am
Shortly after having my son in 2006 , I got the iud method of birth control inserted in me , jus recently I got it removed because I was having bad cramps all day every day so I went to the hospital to see what was going on with me . I don't remember the exact thing they said but the iud was causing me pain so I got the iud removed 10/2012. I'vebeen trying to gget pregnant since I had it removed b[Show More]Shortly after having my son in 2006 , I got the iud method of birth control inserted in me , jus recently I got it removed because I was having bad cramps all day every day so I went to the hospital to see what was going on with me . I don't remember the exact thing they said but the iud was causing me pain so I got the iud removed 10/2012. I'vebeen trying to gget pregnant since I had it removed but no luck. I'm afraid thst I won't be able to have another child. Please help.
TESSAJune 29, 2013 at 2:46 pm
i had my first coil in august 2010 it was fine but after two years it started to come out doctor didnt think it had been fitted correctly in the first place so had it removed had second one fitted in jan this year after 2 months started to get weird metallic taste in my mouth and then numbness pins and needles in arms and legs weird sensations in my head nausea dizziness generally feeling unwell w[Show More]i had my first coil in august 2010 it was fine but after two years it started to come out doctor didnt think it had been fitted correctly in the first place so had it removed had second one fitted in jan this year after 2 months started to get weird metallic taste in my mouth and then numbness pins and needles in arms and legs weird sensations in my head nausea dizziness generally feeling unwell wet feeling down below all the time had lotsof different tests all neagative call it womens intuition had a feeling it could be the coil my husband and family were quite worried about me contacted the doctor who fitted my coil she agreed to remove it the same day also told me these side affects are known but as they are not common they are not listed i hope my comments have been of some help to others experiencing the same effects
MichelleJune 12, 2013 at 6:09 pm
I had the Mirena inserted on June 3 as part of a treatment for endometriosis. In less than 2 weeks I have had huge crying jags that lasted for hours, the feeling of being overwhelmed and will cry at the drop of a hat - I was no where close to like this before. The past couple days I have now been experiencing numbness in my hand/arm (elbow) down on and off. Plus just a general feeling of unwell[Show More]I had the Mirena inserted on June 3 as part of a treatment for endometriosis. In less than 2 weeks I have had huge crying jags that lasted for hours, the feeling of being overwhelmed and will cry at the drop of a hat - I was no where close to like this before. The past couple days I have now been experiencing numbness in my hand/arm (elbow) down on and off. Plus just a general feeling of unwell. I have discovered all of these posts and I just called my OB/GYNE office to report these side effects and told them I would like it removed. My plan is to have it removed ASAP. I would rather deal with the pain of my endo than keep experiencing what I have been or worse, develop more as others have. Ladies, get them removed and all the best health and recovery to you.
RebeccaJune 11, 2013 at 5:55 pm
After the I.U.D i bled for months... eventually things got better and life went back to normal... a year ago i got it in.... for the past three months, its been bad. I bled badly, and i had to remove it. Ever since... i bleed four times a month, painfully.... and cant have intercourse at all with out aggravating it. The people who convinced me to get it in lied to me and said that there were no ri[Show More]After the I.U.D i bled for months... eventually things got better and life went back to normal... a year ago i got it in.... for the past three months, its been bad. I bled badly, and i had to remove it. Ever since... i bleed four times a month, painfully.... and cant have intercourse at all with out aggravating it. The people who convinced me to get it in lied to me and said that there were no risks at all and that it was 100% incapable of allowing pregnancy. this was a youth pregnancy place. What should i do?
VirginiaJune 11, 2013 at 5:19 pm
Wow I had the Mirena inserted and about three months later had started getting shooting pains in my arms and legs, moodiness. VERY sore breasts, fatigue, numbness in arms etc! The doctors also told me this could not possibly be from the Mirena so I believed them. Well three years or so later my life is not worth living. I hated to wake up in the morning. I never thought it was the Mirena until my [Show More]Wow I had the Mirena inserted and about three months later had started getting shooting pains in my arms and legs, moodiness. VERY sore breasts, fatigue, numbness in arms etc! The doctors also told me this could not possibly be from the Mirena so I believed them. Well three years or so later my life is not worth living. I hated to wake up in the morning. I never thought it was the Mirena until my sister found this site last October! We were amazed at how many women had the same symptoms. My doctors told me that nothing was wrong and that I needed counseling! After seeing all these women with the same issues I demanded that it be taken out the very next day. I hoped that all my symptoms would disappear. Well they did not. I still have horrible symptoms during my cycle and when I ovulate. I have multiple groths in my uterus golfball size and I need a partial hysterectomy!! For years these doctors denied my symptoms saying there was no reason for me to have them...I cannot believe that this has happened to so many women and still they do not recognize publicly what is causing this!! I am not the same person at all!! Geez I just want to feel my age wich is 42. As it is right now I feel 70! I had hoped last October when I had it removed that I would feel good again....NOT!
LindaJune 9, 2013 at 11:37 pm
I am presently awaiting ultrasound to find the missing Myrena coil, I went for smear and asked GP to check my stomach as it felt weird, she felt a hard edge to my liver; I must get scan on pelvic regjon and liver, and a liver function test. I am terrified I have cancer in my liver, or the coil is stuck in my liver, I don't know which is worse scenario. I now have to wait and see, has anyone heard [Show More]I am presently awaiting ultrasound to find the missing Myrena coil, I went for smear and asked GP to check my stomach as it felt weird, she felt a hard edge to my liver; I must get scan on pelvic regjon and liver, and a liver function test. I am terrified I have cancer in my liver, or the coil is stuck in my liver, I don't know which is worse scenario. I now have to wait and see, has anyone heard of it reaching the liver, if so, what happened?
jordynnMay 21, 2013 at 8:52 pm
I am a young 19 year old when I was fourteen I got pregnant and had a little girl at 15 I had a mirena. it was the most terrifying experience I ever had I would not wish it on my worse ememy.
CourtneyMay 16, 2013 at 10:49 pm
I have been trying to figure out what is wrong with me, the blurry vision, cloudy thoughts, abdominal pain, acne, and fatigue, I would have never thought it was mirena. After reading these comments I went directly to their website, downloaded the full prescribing information and read through it all. Nowhere does it list any of these symptoms, and this is the only medication change I've made so it [Show More]I have been trying to figure out what is wrong with me, the blurry vision, cloudy thoughts, abdominal pain, acne, and fatigue, I would have never thought it was mirena. After reading these comments I went directly to their website, downloaded the full prescribing information and read through it all. Nowhere does it list any of these symptoms, and this is the only medication change I've made so it has to be the issue. I know for a fact I did not sign anything that stated any of these when the IUD was placed as well. I attend college and maintain an A average, I had no problem with homework before, but now I find myself sitting for hours trying to figure something simple out, because I cannot think! I have always had perfectly fine vision as well and everything has been going blurry, I thought both of these meant I wasn't getting enough sleep, but even after 8 hours I still found it hard to do anything. I know thats not right. I don't want to go into everything else, I still have the IUD in though, I am calling the doctors ASAP tomorrow to get it removed. I hope that my vision is not permanently damaged and everything will go back to normal, its been just over a month. I'm glad I didn't go for a year or more thinking something else was wrong.
LavettaMay 13, 2013 at 11:04 pm
I had the mirena put in on 7/2010 and @ first I thought it was doing what it was supposed to do then I started bleeding on & off, sometimes heavy & sometimes light. After about a year my period would come on without warning so I went back to the Dr that put the mirena in & they sent me for an ultra sound & they said that I had fibroids and the mirena was in place. I said to the DR [Show More]I had the mirena put in on 7/2010 and @ first I thought it was doing what it was supposed to do then I started bleeding on & off, sometimes heavy & sometimes light. After about a year my period would come on without warning so I went back to the Dr that put the mirena in & they sent me for an ultra sound & they said that I had fibroids and the mirena was in place. I said to the DR that I wanted the mirena taken out & they kept saying give it more time & see if the bleeding stops & that it was caused by the fibroids. To make a very long story short after I did all this complaining telling them that I wanted the mirena removed because I thought that it was causing me more problems & I would rather deal with the heavy periods & them saying no.They are now telling me that the mirena is in my abdomen & it have to be surgically removed. I am very pissed off because if they would have listened to me & removed the damn thing I wouldn't be having the problems that I'm having.
DionMay 6, 2013 at 4:47 pm
ive had the mirena iud since july 17th 2012 and i dont have any pains or anything but i think that im pregnant my doctor told me that i wouldnt get pregnant but in all honesty i think that i am. how am i to go about all of this if i am
LoganApril 24, 2013 at 5:58 am
I got the Mirena in February 2012. I began to experience all of the symptoms that I expected such as heavy cramping and constant bleeding. About three weeks later those went away and I thought that was the end of the discomfort. Over the next five months I began to develop joint and muscle pains in my hips and legs. I am a runner and at first running was able to relieve the pain. As time went on[Show More]I got the Mirena in February 2012. I began to experience all of the symptoms that I expected such as heavy cramping and constant bleeding. About three weeks later those went away and I thought that was the end of the discomfort. Over the next five months I began to develop joint and muscle pains in my hips and legs. I am a runner and at first running was able to relieve the pain. As time went on it progressed to the point that nothing would stop the pain. The joint pain was debilitating. I couldn't sit or stand for more than ten minutes at a time and had to constantly be changing position or the pain became overwhelming. I am a very health individual so none of this made sense. It was like I was developing the symptoms of rheumatoid arthritis except that the pain started in the large joints of my hips and knees. One day it dawned on me that the pain started around the time that I got the Mirena. I did a google search and realized that other women had experienced similar symptoms. I made an appointment with my healthcare provider the next day. When I went in to talk to her I told her what was going on and that I wanted the Mirena out. She didn't believe the Mirena was causing this but she agreed to take it out. I walked into the clinic that day hunched over and barely able to walk. Literally an hour after the removal 90% of the pain was gone. This was June of 2012. It took a few months for the pain to be completely gone, but I am lucky I realized as soon as I did what was causing my distressing symptoms.
AriannaApril 23, 2013 at 9:55 pm
I have had the Mirena for a little over 2 years. I just recently started having serious issues like bleeding heavily with big huge blood clots for 2 weeks. went in for ultrasound 5 times at first i was told the IUD is there and then I got a second opinion and that said I didn't have one. Back n forth and back and forth. went to a different doc at a different hospital and it confirmed it's not ther[Show More]I have had the Mirena for a little over 2 years. I just recently started having serious issues like bleeding heavily with big huge blood clots for 2 weeks. went in for ultrasound 5 times at first i was told the IUD is there and then I got a second opinion and that said I didn't have one. Back n forth and back and forth. went to a different doc at a different hospital and it confirmed it's not there at all no where in my body. I have a cousin that had the same symptoms so here it goes: shaky, jittery feeling, not feeling well over all. weak, fatigued, abdominal pain both lower and high and pain retreated to the lower mid back. Contractions also in both high and low abdomen. She had a few seizures after a few months and was told too that they couldn't find the mirena with ultrasound and xray.I have had 2 xrays and says that of no IUD. After she had her seizures they did a Catscan and low and be hold it was there but she had to have 2 laproscopic surgeries to find it the first time they couldn't. SO I may be going through the same thing. Would like to warn other and let my story be heard. SOmetimes its not as easy to detect the Mirena for some odd reason but I hope they find whats going on with me if its not the mirena which I really think it is.
AnnaApril 17, 2013 at 7:51 pm
I had the Mirena for 3 yrs. march 23rd 2013 I went to the ER because I was I So much pain I thought maybe my cyst had burst. The doctor told me & my husband that I was pregnant. It was an ectopic pregnancy so I had to have emergency surgery because my left tube had burst, I was also internally bleeding. The doctor removed the Mirena while I was in surgery. It was one of the most scariest thi[Show More]I had the Mirena for 3 yrs. march 23rd 2013 I went to the ER because I was I So much pain I thought maybe my cyst had burst. The doctor told me & my husband that I was pregnant. It was an ectopic pregnancy so I had to have emergency surgery because my left tube had burst, I was also internally bleeding. The doctor removed the Mirena while I was in surgery. It was one of the most scariest thing I have ever been through and something I don't ever want to experience again. Mirena users be very careful.
Dana MApril 16, 2013 at 4:41 am
I started with the Morena after my daughter was born in 2009. It was alley breastfeeding at the time and after I had it put in my milk decreased so quickly daughter lost so much weight I had to supplement. They said it wasn't supose to effect milk supply but that was false. It ended up getting pushed into my uterus. It took two trips to the doctor and weeks of constant pain to get it removed. Now [Show More]I started with the Morena after my daughter was born in 2009. It was alley breastfeeding at the time and after I had it put in my milk decreased so quickly daughter lost so much weight I had to supplement. They said it wasn't supose to effect milk supply but that was false. It ended up getting pushed into my uterus. It took two trips to the doctor and weeks of constant pain to get it removed. Now with my son I chose the copper. I figured no hormones would be better and my milk never had any problems. But I have developed long monthly migrains since he was born. Now I'm on multiple drugs to try to prevent and maintains the pain. In addition, I am on my 3rd Jud because the second fell out months after getting it.
DawnApril 16, 2013 at 1:41 am
Hello ladys I guess I ain't alone this prouduct needs to be put to a stop I am ready to share my story .2004 I gave birth to mydaughter 6 weeks later I got Iud inserted talk about hell that thing was uncomfortable I felt it clamping down very hard for about a year . The pain kept getting worse it Wasent only hurting me but my husband as well but the intercourse was not the problem my case happe[Show More]Hello ladys I guess I ain't alone this prouduct needs to be put to a stop I am ready to share my story .2004 I gave birth to mydaughter 6 weeks later I got Iud inserted talk about hell that thing was uncomfortable I felt it clamping down very hard for about a year . The pain kept getting worse it Wasent only hurting me but my husband as well but the intercourse was not the problem my case happens to be way more severe I started spotting with very bad cramps cramps icouldent take I remember crying alot I schelude an appt with obgyn to figure out this pain so we do a sonogram he's convinced it Is not the Iud. A sonogram was given his face turned into shock as he told me that there's something elese besidesthe Iud. That something elese was a baby Um yeah there was a baby and somewhere above the baby was a Iud I heard a heart beat and thought to my self ok I have 9 months to prepare my self for this child but unfortunately that Wasent the case I had to terminate my pregnancy talk about dazed and confused they just took a part of me and with that was a Iud removed it left scars like crazy especially mentally. What a tragic device they created after that I got cyst and then years later I get pregnant my second child third pregnancy after Iud unfortantly my 4 year is diagnosed with epilepsy. What a ugly feeling having ur child go limb in ur arms I as a woman and mother encourage any one with I iud to get it removed it's a bloody murder. That's beyond a mistake bayer but the company Is not the one who suffers . Good day to all
tristanApril 12, 2013 at 1:37 pm
i had the mirena put in about 6 yrs ago it was ok for about 3 yrs then i had very bad lower stomach cramping and went to the dr they done xrays and found that it had migrated and almost attached to my bowel i had to have surgery to get it takin out it was vry painful i talked to the lawyers they said i might not be able to have kids anymore but i dont have the money to go to a fertilaty dr to find[Show More]i had the mirena put in about 6 yrs ago it was ok for about 3 yrs then i had very bad lower stomach cramping and went to the dr they done xrays and found that it had migrated and almost attached to my bowel i had to have surgery to get it takin out it was vry painful i talked to the lawyers they said i might not be able to have kids anymore but i dont have the money to go to a fertilaty dr to find out forsure.... since i couldnt use this kind of birth control i have had the implanon it is the stick that goes in your arm and last for 3 yrs it works great i think it is the best kind of birth control out right now i have had it for about 4 yrs now and have nothing bad to say about it... it is pain free and you cant even tell you have it in your arm!!!
AlyssaApril 11, 2013 at 6:45 pm
A couple of other side-effects that have subsided since getting Mirena removed: the migraines the anemia For those coming onto this site to tell the women here to stop complaining and for us to take responsibility, we DID! That's why our Mirena has been taken out, you nitwits. Why did it take so long? OH, perhaps it's because DR's everywhere are saying that this is a safe device to use! Maybe beca[Show More]A couple of other side-effects that have subsided since getting Mirena removed: the migraines the anemia For those coming onto this site to tell the women here to stop complaining and for us to take responsibility, we DID! That's why our Mirena has been taken out, you nitwits. Why did it take so long? OH, perhaps it's because DR's everywhere are saying that this is a safe device to use! Maybe because we all just had children, so we might attribute these side effects to postpartum depression, fatigue from taking care of our children, on top of having jobs (for those who are working), and if you had read through any of these comments you would realize that most of us HAVE GONE TO THE DR's and guess what we were all told?! That nothing is wrong, by "professionals"! Get off the damn site since you have not place here in the first place. Thank you.
AlyssaApril 11, 2013 at 6:16 pm
Soon after having Mirena inserted I was having the worst depression of my life. Never before had I contemplated suicide, but I was. And with a little one, it made it all the more scary. I felt worthless, and that my son and his father would be better-off without me. I went to see a psychiatrist, but, he thought I might have OCD, anxiety and depression. So, he wrote a referral to my primary care pr[Show More]Soon after having Mirena inserted I was having the worst depression of my life. Never before had I contemplated suicide, but I was. And with a little one, it made it all the more scary. I felt worthless, and that my son and his father would be better-off without me. I went to see a psychiatrist, but, he thought I might have OCD, anxiety and depression. So, he wrote a referral to my primary care provider for a second opinion. My Dr thought I MIGHT have been slightly depressed with anxiety but no OCD. So he wrote out a prescription for an anti-depressant and it made me feel TERRIBLE, even worse, like I was in a daze; day-in and day-out. I didn't feel like myself as if I didn't have any emotions. Later that year, I ended up leaving my son's father and stopped taking my anti-depressants and I felt like I could get by without them (I had attributed my depression to him, which it may have been in part, but, I don't think the suicidal thoughts were coming from my being in the relationship, who knows). Anyway, I started a new relationship with a man who really cared about me, and I WAS STILL feeling worthless, and suicidal. I had bouts of rage, feeling hate towards loved-ones without any apparent reason. I was on such high-highs and low-lows that my own mother thought I was on drugs (well, I was, it's called Mirena! I just didn't put it together). I had Mirena in for almost 4 years. Throughout those four years, I had a constant sharp pain in my pelvic area, where my ovaries are located (only to find out later that I had a cyst on my ovary probably caused by Mirena) and the anxiety was at a constant high level. Throughout my new relationship, I was having a feminine odor as well that was becoming increasingly more and more embarrassing for me at work, at school and in public in general and it was affecting my and my boyfriend's sex drive, to the point where he suggested I go to the doctor. I went to my Gynecologist and told him my concerns with the IUD, and he said that the odor could be cause by Mirena and we decided on a date for it to be removed. Since removal, NO MORE odor! It is a revolution. It did take a week or so for it to be completely gone, but I've had it out for about 4 months now, and the change is AMAZING. Immediately following removal my whole being felt freer, my mind was felt like a fog, no, a weight had been lifted and my pelvic area is no longer in pain! That constant cramping, and sharp pains are completely gone. Now, for the negative emotions that I'm feeling even after removal, what I'm suspecting is the "Mirena Crash": Bouts of anger Mood swings some anxiety and Insomnia (BAD INSOMNIA) My insomnia is the worst part of post Mirena Removal, probably because I've never dealt with insomnia before this. But, I work a full-time job and go to school, making my work days 14 hrs long at times, and I'll be exhausted by the time 10pm rolls around, but I'll go to bed and be WIDE awake! I've laid in bed for hours on end and not be tired. 3 am rolls around, still not tired. 5 am rolls around and I'll finally go to sleep until I have to start my day all over again. I obviously can't function, or don't want to function like this, so I started taking melatonin tablets, which help, but make me groggy the next day. The lack of sleep does not help with my irritability either. So, it is a vicious cycle. I am going to stop taking the melatonin to see if I can wait out the insomnia. NEVER using hormonal birth control, again! EVER! I think we need to revolutionize how America thinks of sex because it isn't right that so many women are having these side-effects from hormonal birth control use just to fulfill this "need" for sexual freedom and America's obsession with it. Is it worth your sanity? Not for me.
M. WagnerApril 11, 2013 at 3:51 am
I go tomorrow to get my Mirena out. I've had it 22 months. The 1st year wasnt bad, I enjoyed not having a period. Then I began cramping. Not regular cramps, but similar to the ones I had when I miscarried years ago. This has been every day for the last 6 weeks. I am just praying it's a cyst (I have PCOS and it turns out Mirena aggravates that) and not because it's embedded. I believe Mirena has me[Show More]I go tomorrow to get my Mirena out. I've had it 22 months. The 1st year wasnt bad, I enjoyed not having a period. Then I began cramping. Not regular cramps, but similar to the ones I had when I miscarried years ago. This has been every day for the last 6 weeks. I am just praying it's a cyst (I have PCOS and it turns out Mirena aggravates that) and not because it's embedded. I believe Mirena has messed up my hormones, I've had acne, mood swings, depression, I've developed high blood pressure, bloating and breast tenderness. I'm only 30 and have a 2 1/2 yr old. Please don't try this drug if you haven't already got it!
DanaApril 7, 2013 at 12:27 am
I had the Mirena inserted September 21st 2012 and since I have gained 10lbs, heart palpitations,severe headaches,no sex drive and on top of everything it makes my lupus act up BAD! I am making an appt to get it removed immediately.
ChalayApril 2, 2013 at 2:12 am
I had the Mirena inserted in 2006 after giving birth to my first child. In 2009 I had my Dr. remove it as my husband and I wanted to try for another child. After irregular cycles and several visits to a fertility specialist, I was diagnosed with premature ovarian failure. I believe it was the Mirena that caused this.
MaureenMarch 14, 2013 at 7:56 pm
I had the Mirena inserted Jan 2009 right after having my first child. I have gained over 30 pounds on top of what I was post partum and I have not had a regular appetite in 3 years. I barely eat anything and yet have not lost a pound. My weight changes with my period - thats it. To say I've had mood swings would really be the understatement of the year! Sex has been painfull, even after having it [Show More]I had the Mirena inserted Jan 2009 right after having my first child. I have gained over 30 pounds on top of what I was post partum and I have not had a regular appetite in 3 years. I barely eat anything and yet have not lost a pound. My weight changes with my period - thats it. To say I've had mood swings would really be the understatement of the year! Sex has been painfull, even after having it out my period has never gone back to normal. Its been 6 months since i've had it out and even getting it taken out was a pain! My doctor swore up and down that Mirena has a very low dose of hormones and it was impossible that the Mirena was causing any of my symptoms, two doctors told me that even after I told them that I had found people online with the same symptoms as me.
lexieMarch 7, 2013 at 9:25 pm
I've had the mirena ...and still do and nothing has went wrong. So everything is great. I honestly believe that this is the best birth control to take and best recommended. For those who are complainers I prefer for u to take something like the pills or shot. If u know u can't take pain, etc then just don't get it.
KristinMarch 5, 2013 at 5:07 am
After having my second child, my doctor insisted that i get mirena put in. I had so many problems, it was ridiculous. I kept going back to my gyn telling him something was wrong. He put me on percacet (dont know spelling) just to put it in because i was so nervous about it. I was told that it would make your period irregular for a while but i never imagined having my period for 6 months straight a[Show More]After having my second child, my doctor insisted that i get mirena put in. I had so many problems, it was ridiculous. I kept going back to my gyn telling him something was wrong. He put me on percacet (dont know spelling) just to put it in because i was so nervous about it. I was told that it would make your period irregular for a while but i never imagined having my period for 6 months straight and it was continuous and heavy. I called several times and he kept saying to call if it doesnt stop in next couple months, finally it stopped. But then i consitently got infections, in which i never had a problem with before. I had painful intercourse and was eventually diagnosed with vaginitis. I kept telling him i wanted it out and he would try to convince me that everything was fine and sent me back home. After going to several different doctors to find out why sexual intercourse was so painful and unsuccessful and always being told they didnt know what was wrong and NO MIRENA WOULD NOT CAUSE THIS, i finally went back and insisted to have it taken out. Needless to say, i never went back to that gyn ever again! I had headaches, depression, painful intercourse, mood swings, etc. After getting through vaginitis and being able to have successful intercourse again, we decided we wanted to try for another child. That was last March and here we are a year later and we still are not pregnant. I have asked several times to even my now gyn if mirena could cause infertility and even she swears it doesnt. I feel like every doctor takes up for mirena and my husband swear they get a cut back from it. I just want to know the truth, but she insists on not researching whats going wrong until June. I became pregnant with my first child after 2 months of trying and now my boys are 16 months apart, so it wasnt hard getting pregnant with my second child. I want to investigate but after hearing womens testimonies of having surgery etc, i feel like possible infertility isnt so important. I feel like if something happened to the mirena while it was in me and the doctor just never told me and i was under medication during insertion/removal, how would i know or even be able to prove it? Ive always known in my heart something went wrong but never had a doctor to speak up and tell me the truth even the doctor whom i have now, whom i trust.
AmyMarch 5, 2013 at 1:20 am
Hi. I had Mirenta inserted in June 2011 about two months after having my daughter. The first like 4 months were fine.... Thereafter, I started experiencing side pain, headaches and infections.. I went to my ObGyn and he prescribed antibiotics and said I had a cyst on my ovary. Since then my headaches are worse (like everyday) my hips hurt and the pain goes all the way down my legs into my feet, [Show More]Hi. I had Mirenta inserted in June 2011 about two months after having my daughter. The first like 4 months were fine.... Thereafter, I started experiencing side pain, headaches and infections.. I went to my ObGyn and he prescribed antibiotics and said I had a cyst on my ovary. Since then my headaches are worse (like everyday) my hips hurt and the pain goes all the way down my legs into my feet, making my feet swell. I have shortness of breath and it feels like panic attacks when I am trying to sleep at night. I also have gained weight... I plan on having my Mirena removed ASAP; it's the only way I will know for sure if it's the culprit of all of this.
TGMarch 4, 2013 at 4:33 pm
After having tha Mirena for 4 years I took it out in hopes to get pregnant ... Now over a year later still cant get pregnant. Dr say nothing is wrong with me :(
CarrieMarch 3, 2013 at 6:53 am
In December of 2009 I had the mirena inserted. I was told it would only be minor discomfort to insert it and the only side effects were light bleeding for the first few days. Little did I know I was in for a nightmare. It hurt a lot when it was being put in. My sex drive went away completely, I had heavy bleeding for two weeks after it was inserted, pain in my ovaries that had me doubled over in p[Show More]In December of 2009 I had the mirena inserted. I was told it would only be minor discomfort to insert it and the only side effects were light bleeding for the first few days. Little did I know I was in for a nightmare. It hurt a lot when it was being put in. My sex drive went away completely, I had heavy bleeding for two weeks after it was inserted, pain in my ovaries that had me doubled over in pain and increased cysts on my ovaries. I gained 40 pounds in 3 months and I was sick to my stomach all the time. I am a suffer of depression and this exacerbated the problem badly. I also experienced horrible mood swings that would leave me exhausted. I went to have it removed after three months and was told by Dr that I should give it six months. I ended up gaining 20 more pounds and all the things I described above got worse. I bleed almost the whole time. I believe that I am still suffering ramifications from the mirena. The bleeding and mood swings stopped, my depression got better but I still get pain and frequent cysts in my ovaries. My cycle has never gone back to normal and I haven't been able to conceive a child. I lost insurance and could no longer pursue medical treatment from the damage. My life has been altered and made harder due to this device.
RachelMarch 1, 2013 at 11:05 pm
For me, the person who was having the seizures, I didn't even think about the two being related until more recently. The neuro had me looking at all kinds of things ie. stress, not getting enough sleep, and even caffeine. If the Mirena is the cause for my seizures I defiantly feel a sense of responsibility in keeping it in for so long. My purpose is more to get that information out there. I do[Show More]For me, the person who was having the seizures, I didn't even think about the two being related until more recently. The neuro had me looking at all kinds of things ie. stress, not getting enough sleep, and even caffeine. If the Mirena is the cause for my seizures I defiantly feel a sense of responsibility in keeping it in for so long. My purpose is more to get that information out there. I don't want other women to suffer the way I have. I just recently had a seizure in front of my work colleges at a work training. That was in front of 150 people. I am so sad that this may be something I have done to myself. Also to update, I had the Mirena out on 2/7, and had 2 grand mal seizures on 2/20 i then had my first period since having the IUD removed on 2/22. I am certain there is a correlation. I am going to figure this out. I also called into the bayer healthcare line and reported seizures as a possible side effect after being urged to by other people that have suffered from seizures and had the IUD placed. Despite other women having made reports, the healthcare line actually told me they have had no reports. Something is going on!!!
Tiffany RFebruary 28, 2013 at 7:26 pm
I was on the Marina for 7 years. First 5 years was great. Once I had the first one removed and second one insterted is when the troubles all started. I had a few infections and lots of pain. After two years I went back to doc thinking I had another infection. Come to find out I had CIN 3 also. Some docs say its from being on IUD and some say its not. I have been off it since June and periods are s[Show More]I was on the Marina for 7 years. First 5 years was great. Once I had the first one removed and second one insterted is when the troubles all started. I had a few infections and lots of pain. After two years I went back to doc thinking I had another infection. Come to find out I had CIN 3 also. Some docs say its from being on IUD and some say its not. I have been off it since June and periods are still not totally normal. I would like to speak to anyone else who came down with CIN 1, 2 or 3. Or Cervical Cancer at all.
sharonFebruary 25, 2013 at 4:16 pm
I don't understand how some of you could leave something in for so long that was causing so many problems you don't feel responsible at all? Not even s little bit?
chaseFebruary 24, 2013 at 9:55 pm
my wife had a mirena IUD for about a year and a half. she took it out 2 months ago, after complaining about some abdominal pain and general weakness. the doctor found that she had a slight anemia. anyway, he said that everything will be ok. a few days ago we heard about the Mirena recall and now we are worried. we set up an appointment with the doctor and now hope everything is fine.
AlyFebruary 21, 2013 at 11:07 pm
i had the mirena put in a few months after i had my third child i had it in for about two years. come to find out im 6 weeks and 3 days pregnant went to get the mirena removed and they could not removed it. i now have to get it surgically removed. my doctors were shocked.
AmyFebruary 19, 2013 at 8:56 am
I have had some of the same issues as everyone: mood swings, a general feeling of not feeling well, melasma, cystic acne, and thyroid issues. I had the Mirena in July 2007. In October 2008, I developed ovarian cysts. I was given birth control pills to shrink the cysts. At that time, I received a breast exam. Nothing was found. In May 2009, I found a lump and it was determined to be Breast Ca[Show More]I have had some of the same issues as everyone: mood swings, a general feeling of not feeling well, melasma, cystic acne, and thyroid issues. I had the Mirena in July 2007. In October 2008, I developed ovarian cysts. I was given birth control pills to shrink the cysts. At that time, I received a breast exam. Nothing was found. In May 2009, I found a lump and it was determined to be Breast Cancer. I was treated with surgery, chemotherapy and radiation. I then had a hysterectomy and my gall bladder was removed that same year. I had about a year and a half of thinking I was cured and was living my life. In March 2012, after feeling off, I was informed that the cancer had spread to my bones. I was considered Stage 4, life expectancy unknown. I wish Bayer could look at my 5 children in the face and tell them why their mom is going to die because of their product. I was just 40 when I was diagnosed with breast cancer. I just hope I can live long enough to sue these people.
RachelFebruary 15, 2013 at 9:43 pm
I had my IUD inserted in December of 07. After accidentally pulling it out in December of 09 I had one reinserted in January 2010. I had my first seizure in March '10 while sleeping. After that I had a series of "black outs" basically i would start talking about things unrelated to the conversation I was having. I ended up on seizure meds in September of 2011. Things seemed to be getting bett[Show More]I had my IUD inserted in December of 07. After accidentally pulling it out in December of 09 I had one reinserted in January 2010. I had my first seizure in March '10 while sleeping. After that I had a series of "black outs" basically i would start talking about things unrelated to the conversation I was having. I ended up on seizure meds in September of 2011. Things seemed to be getting better but then I started having grand mals. I had 1 December 2011 and then some more in May 2012 and finally 1 in June 2012. I stopped taking the seizure medication. The grand mals went away but I began tracking my "black-outs" and noticed that I got them 2x's a month, once at the beginning of my period and then another just after. I finally got the IUD removed February 7, 2013 I had my last "black-out" on February 6, 2013.
StephanieFebruary 15, 2013 at 4:13 pm
I had my mirenia inserted in aug 2009 2 months after my sons birth, no problems noticed at first. Then I got severely depressed and had anxiety that I was medicated for. I also had severe cramping. I've had 2 abnormal paps which I still don't know for sure why, and no sex drive what so ever. I just got my mirenia removed last wk I've had a headache and felt weak, tired, and nauseous since. Pls hel[Show More]I had my mirenia inserted in aug 2009 2 months after my sons birth, no problems noticed at first. Then I got severely depressed and had anxiety that I was medicated for. I also had severe cramping. I've had 2 abnormal paps which I still don't know for sure why, and no sex drive what so ever. I just got my mirenia removed last wk I've had a headache and felt weak, tired, and nauseous since. Pls help.
KarenFebruary 1, 2013 at 6:07 pm
I had my Mirena IUD removed about 31/2 yrs ago,after having it in me for 10 yrs.I never could figure out why I had real heavy bleeding during my menstrual, and why all the sudden I had cramping after it be removed. Because I never had cramp before it was placed. I started have dizzy spells,lower back pain, no energy,racing thoughts,blurred vision, ( I now wear proscription-glassing all the time wh[Show More]I had my Mirena IUD removed about 31/2 yrs ago,after having it in me for 10 yrs.I never could figure out why I had real heavy bleeding during my menstrual, and why all the sudden I had cramping after it be removed. Because I never had cramp before it was placed. I started have dizzy spells,lower back pain, no energy,racing thoughts,blurred vision, ( I now wear proscription-glassing all the time when I only needed them to read), I see spots,. I now have uncontrollable mood swings.I was diagnose of have anxiety. I now have a uncomfortable felling when having sex .
DanaJanuary 29, 2013 at 12:59 am
I had the Mirena IUD put in in 2002, I was told it was the only safe birth control i could use for health reasons. when it was inserted it was so painful that the gyn finally did a nerve block. I had severe pain from the start, severe headaches, night sweats, mood swings,abdominal/back pain, cyst on the ovaries, tachycardia, pcos, fibromyalga. The pain was so severe it caused my blood sugars to go[Show More]I had the Mirena IUD put in in 2002, I was told it was the only safe birth control i could use for health reasons. when it was inserted it was so painful that the gyn finally did a nerve block. I had severe pain from the start, severe headaches, night sweats, mood swings,abdominal/back pain, cyst on the ovaries, tachycardia, pcos, fibromyalga. The pain was so severe it caused my blood sugars to go high, thus causing multiple trips to the ER and hospital admits. I was sent to physician after physician, I was tried on many different meds, had reactions, nerve blocks for the headaches, nobody could say what was wrong, then they said it was all in my head. I had to have eye surgery due to the high sugars and vomiting. this has been a living hell. now i see this on tv and am reading about this? I am a nurse, or was before the surgeries I had to have. I know how it all sounds, but I said all along there was something wrong. I had the IUD taken out in 2006, but still have issues.
LoriJanuary 24, 2013 at 1:14 pm
I have had a Mirena IUD inserted in me since Feb. 2011. I have lower abdominal pain so bad it makes me nauseated, migranes, dizziness, muscle aches, mood swings, depression, etc. I am afraid to have it removed because it was inserted to stop me from hemorraging to death. I need help.
EOJanuary 23, 2013 at 6:50 pm
I had the Mirena inserted on January 2011, to help control my heavy periods. The insertion was very painful to the point that it took me about two days to recover. I had abundant periods for about three months, then it stopped completely for a few months. Now I have my periods sporadicly and not very heavy. A few months back, I started having migraines, severe mood swings, I'm tired all the time, [Show More]I had the Mirena inserted on January 2011, to help control my heavy periods. The insertion was very painful to the point that it took me about two days to recover. I had abundant periods for about three months, then it stopped completely for a few months. Now I have my periods sporadicly and not very heavy. A few months back, I started having migraines, severe mood swings, I'm tired all the time, some nights I can't sleep, although I'm exhausted. When I went to have my Pap smear, the doctor who inserted it could not find the strings and ordered a sonogram, which revealed the Mirena was still in place. However, my health keeps deteriorating, I'm having real bad PMS symptoms, painful cramps almost like having contractions, hot flashes, sweats, balance problems, my lower back hurts, now my upper back started hurting, also my knees, feet, neck arms, shoulders hurt a lot. Libido is none existent and my breast are very tender. Did I mention that I gained about 50 pounds? I just called my doctor for an appointment next week to have it removed.
lorraineJanuary 21, 2013 at 9:37 pm
I had the Mirena fitted in 2003, I had it for 5 years and had it removed in 2008. I didn't have any problems while it was inserted. However in 2008 I started to have slight menstrual problems, in 2011 i was diagnosed with adenomyosis and now its at a severe stage. Could the Mirena be the cause of this problem, as I never had any problems before.
JessicaJanuary 21, 2013 at 1:59 pm
I just had laporascopic surgery to have the Mirena removed from the outside of my uterus. My new OB that did the surgery says it most likely perforated when it was inserted since at 6 weeks pp our uterus is still very soft and healing. My left Fallopian tube was wound around the device. Luckily, the dr was able to just slip it off with no damage but it could have been so much worse. Like others, I[Show More]I just had laporascopic surgery to have the Mirena removed from the outside of my uterus. My new OB that did the surgery says it most likely perforated when it was inserted since at 6 weeks pp our uterus is still very soft and healing. My left Fallopian tube was wound around the device. Luckily, the dr was able to just slip it off with no damage but it could have been so much worse. Like others, I had weight gain (I've lost 8 lbs since the surgery 2 weeks ago), crazy mood swings, I had a period every month, and excruciating pain after exercise and sex. If you found this link doing research for getting the Mirena, please know that these testimonies are very real and some of us have real injuries. Please, just use something else for birth control.
MieshaJanuary 18, 2013 at 11:16 pm
Hey everyone I'm just seeing all of this but I got a iud back in 2009 & it was painful, I had problems every since I actually had an ectopic pregnancy & had to go to the er every week on Sunday to get a shot in my butt for the baby to dissolve it made my period come out heavy thick & looked like dried up boogers sooooo I kept calling the hospital to talk to any nurse about it because I[Show More]Hey everyone I'm just seeing all of this but I got a iud back in 2009 & it was painful, I had problems every since I actually had an ectopic pregnancy & had to go to the er every week on Sunday to get a shot in my butt for the baby to dissolve it made my period come out heavy thick & looked like dried up boogers sooooo I kept calling the hospital to talk to any nurse about it because I was scared but they told me everything was ok so I was on my way out & I got to the front porch of my buiding & collapsed I had a fever of 110 & hot & cold flashes along with migranes everyday. I had to get my left ovary cut off where the baby was & that was june 27th 2009 but turned around & got pregnant on august 8th 2009 & still be having migranes & pains in my pelvic & stomach area.
BeckyJanuary 18, 2013 at 10:10 pm
I've had my Mirena in for almost 7 years. Don't let them kid you, it hurts to have put in, there was quite a bit of spotting the first month but after that no periods (best thing ever!!) and absolutely no problems. I loved it....UNTIL I wanted it removed to have a new one inserted. Two doctors have been unable to get it to come out, without excruciating pain that I just can't handle. The doctor th[Show More]I've had my Mirena in for almost 7 years. Don't let them kid you, it hurts to have put in, there was quite a bit of spotting the first month but after that no periods (best thing ever!!) and absolutely no problems. I loved it....UNTIL I wanted it removed to have a new one inserted. Two doctors have been unable to get it to come out, without excruciating pain that I just can't handle. The doctor thought it might be embedded but wanted to keep yanking on it anyway. Now my doctor wants to do surgery to remove it, my insurance called it a pre-existing condition and would not pay for the surgery, soooo that's why I still have it when it was only good for 5 years. Have no clue if it is still working or not, after 7 years still no periods so I am guessing it is but NOT counting on it. Just hoping to get this thing out soon!!!
maritzaJanuary 14, 2013 at 4:40 pm
i had my marina put on on dec or 2011 for the reason i was having veryheavy piriod and they would last at times 15 day i would get anemic at time so the doc recomended that i put the marina on so that my perior would go away and then it would probaly be menopause time when i took it of but the day it was put was very painful i still kept getting heavy piriod for the first six month then it stated[Show More]i had my marina put on on dec or 2011 for the reason i was having veryheavy piriod and they would last at times 15 day i would get anemic at time so the doc recomended that i put the marina on so that my perior would go away and then it would probaly be menopause time when i took it of but the day it was put was very painful i still kept getting heavy piriod for the first six month then it stated to slow down my problem is i still have it on i was check doc told me it was in place but i have bad migrain, retaining water in my ankle .my back is away hurting me. i hurt all over i am moody all the time i do have anxiety since i was young but its gotton worst my medication have been chance i still feel axiety all the time i have light bleeding still that last abot a week ,feel weel nausuase and have gain alot of wieght.my breat hurt alot and for some reason i am lacktating still. i urinate 8 or 9 time in a day my mouth is always dry when i wake up my voise barely is hard until i got wash my mouth and gargle.i get cramp and i have to bend down until it goes away.i cant sleep and i am also on medications to sleep and at time it woks i am crying all the the time but i suffer from depression but its wort now.get hot flashes like crazy sleep with fan on even in cold nights.
scottJanuary 12, 2013 at 8:41 am
Gang, this can cause severe consequences to a guy. A year ago I had sex with a woman where I got poked by the string on deep thrusts. Woke up the next morning with significant pain. That turned into a serious infection (not STD), and either the trauma of the poking or the infection led to a recurring urethral stricture (scar tissure buildup in the urethra) that has required three surgeries in one [Show More]Gang, this can cause severe consequences to a guy. A year ago I had sex with a woman where I got poked by the string on deep thrusts. Woke up the next morning with significant pain. That turned into a serious infection (not STD), and either the trauma of the poking or the infection led to a recurring urethral stricture (scar tissure buildup in the urethra) that has required three surgeries in one year. Definitely not fun.
CharmaineJanuary 3, 2013 at 10:26 pm
i got the merina iud put in like on oct of 2012 i been haveing really bad expirence off this iud i have had bad headaches bad skin break outs acnes out this wrld feeling tired, depressed, also have bad tenderness of my nipples and breast, cant sleep at night cionstantly going to the bathroom at night, i have picked up a lot of weight past 2 months as well and haveind bad side pain and lower stoma[Show More]i got the merina iud put in like on oct of 2012 i been haveing really bad expirence off this iud i have had bad headaches bad skin break outs acnes out this wrld feeling tired, depressed, also have bad tenderness of my nipples and breast, cant sleep at night cionstantly going to the bathroom at night, i have picked up a lot of weight past 2 months as well and haveind bad side pain and lower stomach pain
briannaJanuary 3, 2013 at 8:56 pm
i had the mirena placed in sep 18th and as soon as i left i gotsick i couldnt sleep and had serious pain throwing up and soon a month later i started having pains in my left side so i went to a different doc and told them and so they did ultrasound and i found out the mirena was emmebedded im my left side so they called my orignal doc and he told me they were lien and its not emmebedded so i we[Show More]i had the mirena placed in sep 18th and as soon as i left i gotsick i couldnt sleep and had serious pain throwing up and soon a month later i started having pains in my left side so i went to a different doc and told them and so they did ultrasound and i found out the mirena was emmebedded im my left side so they called my orignal doc and he told me they were lien and its not emmebedded so i went back to the doctor today and complain about heart burn and weight gain,depression and still pain in my left side he told me it had nthin to do with the mirena and that i had to wait until april 3rd 2013 to have it removed should i be worried
RubyJanuary 3, 2013 at 4:09 pm
I have had the marina since late 2009. I feel like I'm going crazy. The mood swings are driving me insane, blurred vision and the tenderness of my boots. I still have my period and is very painfull. Sometimes it lasts for 2 days and other times it lasts up to 3 weeks. I have called about having to removed but the wait is too long to see doctor and the cost to have it removed is outrageous. I have [Show More]I have had the marina since late 2009. I feel like I'm going crazy. The mood swings are driving me insane, blurred vision and the tenderness of my boots. I still have my period and is very painfull. Sometimes it lasts for 2 days and other times it lasts up to 3 weeks. I have called about having to removed but the wait is too long to see doctor and the cost to have it removed is outrageous. I have decided to remove it myself when my cycle starts again.
GillianDecember 29, 2012 at 4:21 am
I'm 35 and have had the mirena coil in for almost 5 years now. I believe that it has cause me such drastic" mood swings" (a term I'm using lightly) that I ended up in a mental ward on and off for a few months and I received shock treatment! After all of this I had my first period since having the coil and magically I'm all better. I was due to have it removed in a month or so anyway but I'm gettin[Show More]I'm 35 and have had the mirena coil in for almost 5 years now. I believe that it has cause me such drastic" mood swings" (a term I'm using lightly) that I ended up in a mental ward on and off for a few months and I received shock treatment! After all of this I had my first period since having the coil and magically I'm all better. I was due to have it removed in a month or so anyway but I'm getting it out ASAP. I'm so glad I found this site
ChasityDecember 28, 2012 at 9:38 pm
In August 2005 I had my son and at my ob checkup I decided to go with the mirena for my form of birth control. I bled heavily with the mirena for several weeks and had terrible cramping. I informed my doctor of the situation and upon discussing the problem, we decided to give the iud more time to "adjust" to my body. Once the bleeding stopped, but not the cramping, a new problem arose. Whenever[Show More]In August 2005 I had my son and at my ob checkup I decided to go with the mirena for my form of birth control. I bled heavily with the mirena for several weeks and had terrible cramping. I informed my doctor of the situation and upon discussing the problem, we decided to give the iud more time to "adjust" to my body. Once the bleeding stopped, but not the cramping, a new problem arose. Whenever my husband and I tried to have sexual intercorse, it would hurt the both of us. I would have shooting pain and my husband said that it felt like something was poking him. I went back to my doctor over this mattter and he decided to cut the insert cord a little shorter so things would feel better for my husbannd and I. Afew weeks went past and we were still having trouble sexually and I was still having the pain. I also was bleeding very heavily with my monthly cycle and severe cramping. After another visit with my doctor, I decided to have the mirena removed. When my doctor attempted to remove the iud, he was unsuccessful at doing so. He then asked one of his colleuges to attempt to remove the iud. The other doctor was unable to remove the iud as well. My doctor had decided that they couldn't get the Mirena out so he was going to refer my to an specialist for a DNC and had me go to that office as soon as I left him. Once I got to the specialist's office, I was taken back to his personal office and discussed the situation with him. He had one of his nurses direct me to an exam room so he could look for himself. Once he came in, he had me lay back on the exam table as he took a look. He asked the nurse to hand him a certain instrument and the next thing I knew, I was screaming and crying, crawling back on the table with my elbows trying to get away from him. I was yelling at him to stop and he never did. He stood up, handed the nurse the instrument and looked at me and said, "There, I just saved you from having a major surgery." He told me to stay laying down for 15 minutes to try to get the bleeding to stop and the nurse would bring me an ibprophen for pain. Since this horrible incident, I have had severe bleeing with my periods that I not only get once a month but twice a month since then. I have had very large cysts on my ovary that almost became a major faction in my daughter's pregnancy as to where they were considering doing surgery on me while pregnant if the cycts continued to grow. I will never forget the pain and suffering that the mirena has done to me mentally and physically to my body. I do not recommened this form of birth control to anyone and I have let every woman know my story when I hear them metion the word "Mirena".
MelissaDecember 16, 2012 at 4:44 am
I had my Mirena inserted in February 2008, my son was 8 weeks old when I did this. It was painful the entire time I had it. My ob/gyn told me the pain would go away eventually and so would the night sweats and the periods. My periods stopped but the pain and night flashes did not go away. My gyno did an internal ultrasound in June of 08 and found that the Mirena had shifted into the left Fallo[Show More]I had my Mirena inserted in February 2008, my son was 8 weeks old when I did this. It was painful the entire time I had it. My ob/gyn told me the pain would go away eventually and so would the night sweats and the periods. My periods stopped but the pain and night flashes did not go away. My gyno did an internal ultrasound in June of 08 and found that the Mirena had shifted into the left Fallopian tube and there were also cysts. She moved it back to where it was supposed to go and I was still constantly in pain. August of 2008 I had it removed and was still in constant pain. I went to my mom's ob/gyn and he diagnosed me with endometriosis caused by the Mirena. In October of 2008 I had laprascopic surgery to remove the endometriosis and was put on the Lupron injection to shut my reproductive system down temporarily while I healed. I was on the Lupron for 3 months before I had enough of that. I was still in constant pain for the next 2 years. In 2009 I got pregnant with my daughter and that stopped the pain for awhile and we thought that the endometriosis was gone. Come to find out it was not. When I had my tubes tied in October 2010 they found more endometriosis and cleaned it up yet again. I had been dealing with pain for numerous years until I found a doctor that actually took care of the problem. This year in February I had a complete hysterectomy at the age of 32. Over the last several years I had tried more than one birth control and had been on and off Ibuprofen for the pain. I had enough and had the problem taken care of even though it was in the extreme way of major surgery.
CamilleDecember 13, 2012 at 5:05 pm
2 1/2 years ago about 12 weeks after my son was born I had the Mirena IUD put in. I was told the pain was minimal. I still remember how painful the insertion was! I was in tears and shaking. When the doctor was measuring my uterus she made a comment how large my uterus was. After I left for weeks I could "feel" the IUD in me. I hated it right away so for the follow up appointment I decided I [Show More]2 1/2 years ago about 12 weeks after my son was born I had the Mirena IUD put in. I was told the pain was minimal. I still remember how painful the insertion was! I was in tears and shaking. When the doctor was measuring my uterus she made a comment how large my uterus was. After I left for weeks I could "feel" the IUD in me. I hated it right away so for the follow up appointment I decided I wanted it out. When the doctor went in to get the strings she could not locate them. She sent me to get a ultrasound of my uterus to make sure it just moved from the right place. Nothing showed up on the ultrasound so she said my body must have expelled it and I didn't notice that if fell out. The pain of having it had diminished so I figured she must be right. I threw my back out of whack about a week ago. I started seeing a chiropractor. He gave me an xray on Monday to look at my spine. He asked me if I had an IUD or abdominal surgery. I told him no to both since all this time I figured my IUD expelled itself and it hadn't crossed my mind since. He said "this sure does look like an IUD" as he pointed to the xray right beneath my rib cage. Yep, it has been MIA for 2 1/2 years just floating or embedding itself to some organ. I went to my PC doc, the one who put in and she was dumbfounded by it. She said to me that she must have done at least a thousand of these and have never had this happen. Well, lucky me I am the 1 in 1000! So now I am going to have surgery to get it removed. With an xray you cant really tell if its embedded somewhere or exactly where it is. I am waiting to hear from the surgeon. I have mixed feelings about the whole thing. I'm afraid of what damage it has done to my body. Its like a freak accident...surreal. I hope they will just be able to go in and get it out with no complications. I hope it hasn't caused any damage to my organs. 2 1/2 years is pretty long for something to be floating around in my body. I really trust and like my doctor and I know people make mistakes...even doctors. I guess I don't know what to do at this point. My husband doesn't think our insurance or my boss should pay for lost pay and the medical bills.
AudraDecember 13, 2012 at 4:05 am
I have the mirena and have bad back pain constant infections and its painful etc. But I don't have healthcare and can't afford to pay out of pocket. I haven't read to much on this but I'm hoping it doesn't get worse. Anyone know if you can get this removed for free or anywhere you can see if there is a problem.
JenDecember 12, 2012 at 4:09 am
I had the Mirena IUD put in approximately April 2008 at age 35. I already had two boys but having the IUD inserted was VERY uncomfortable and not something I would have ever volunteered for again. I had a follow up with my doctor in October of 2008 and everything was normal. In February of 2009 I went to the emergency room and was advised that I had a prolapsed uterus. I contacted my OBGYN and had[Show More]I had the Mirena IUD put in approximately April 2008 at age 35. I already had two boys but having the IUD inserted was VERY uncomfortable and not something I would have ever volunteered for again. I had a follow up with my doctor in October of 2008 and everything was normal. In February of 2009 I went to the emergency room and was advised that I had a prolapsed uterus. I contacted my OBGYN and had to have a vaginal hystorectomy two weeks later in March 2009. My OBGYN could not figure out why this had happened since my pap in October, just 5 months prior was normal. I had not done anything unusual to cause this to happen. Not only was my uterus basically falling out but my bladder had to be stitched back up because is was falling as well. Luckily my bladder had not fallen too far otherwise I would have had permanent damage done and would not be able to hold my bladder. After reading all the comments above I am beginning to wonder if this happened to me due to the IUD. I am now 40 with two beautiful boys (9 & 11). I had previously been on the depo shot for many years both before and after having my children. With the depo I did not have a period so I did not experience the heavy bleeding others have had with the IUD since I went from depo to the IUD. I did have some cramping but figured it was due to the fact I was not on the depo shot anymore. I suffer from migraines and back pain and have been on many different medicines in an attempt to alieviate some of the pain my body is in. I am not sure if this can be attributed to the IUD or not. I am however not very happy with the fact that I may have had to have a hystorectomy due to having the IUD in my body. I would not recommend the IUD to anyone. I was on and off the depo shot for years and never had any problems the only side effect I experienced was no period, which was a side effect I could live with. I originally decided to get the IUD because it lasted 5 years and I did not have to receive a shot every three months. If I had it to do over I would have stuck with the shots...
EccoDecember 10, 2012 at 2:41 am
I had Mirena for 8 months I had it removed 2 mo. ago and the other night was sitting and felt intense pain in my wrist and my vein was blown up 2-3 times its regular size... I went to ER where they said I had no risk factors for blood clots and was probably just trama, but did an ultrasound of my arm and neck...Sure enough I have blood clots all down my left arm from my neck to wrist and now have [Show More]I had Mirena for 8 months I had it removed 2 mo. ago and the other night was sitting and felt intense pain in my wrist and my vein was blown up 2-3 times its regular size... I went to ER where they said I had no risk factors for blood clots and was probably just trama, but did an ultrasound of my arm and neck...Sure enough I have blood clots all down my left arm from my neck to wrist and now have to be on blood thinners, the Dr said the only factor I had to explain it was my Mirena and the fact that I smoked 1-2 cigarettes a day which my Dr said was safe at my age... I am convinced the mirena caused it and for those of you with numbness and tingling please see a DR to rule out blood clots!!!! I have never been so scared in my life I wish I never had it inserted.....
KariDecember 5, 2012 at 4:03 am
I just found out that I am pregnant with my 3 child I have the mirena in for about a month in a half it turns out that it now can not be taken out because it is stuck somewhere in my cervis or somewhere close to that. So now i am going to be a high risk they still want to try and take it out. I am in pain from them trying to pull it out.
ElizabethDecember 5, 2012 at 12:45 am
Okay, I am 15 and my mom made me get on this birth control on June 7th 2011 and the very next week I started my period and I had it every day until August 20th 2011. I have woken up in cold sweats and I have suffered from bibolar and depression worse since then. I have read all of this stuff and I'm afraid of what might happen if I get it taken out. I haven't had a period since August 20th 2011. T[Show More]Okay, I am 15 and my mom made me get on this birth control on June 7th 2011 and the very next week I started my period and I had it every day until August 20th 2011. I have woken up in cold sweats and I have suffered from bibolar and depression worse since then. I have read all of this stuff and I'm afraid of what might happen if I get it taken out. I haven't had a period since August 20th 2011. The doctors never told me these side effects could be possible.
TyanesaDecember 2, 2012 at 8:15 pm
I had the iud inserted in July 2008 4 months after having my son. After having it put in I bled for 4 weeks straight. Then when that finally stopped I had my period every month for 14 days straight. then after a year I missed a month and then the next month I had the worst cramps and then started my period again and it wasn't normal. I had my period the first and third week of the month every mont[Show More]I had the iud inserted in July 2008 4 months after having my son. After having it put in I bled for 4 weeks straight. Then when that finally stopped I had my period every month for 14 days straight. then after a year I missed a month and then the next month I had the worst cramps and then started my period again and it wasn't normal. I had my period the first and third week of the month every month for about two years or so. Then I missed a period and when I started again It was all over the map, they could be any week of the month and I never knew when it would start. When I would miss my period I would gain a little weight. There was a time where I went to the Dr. because I had a weird smell down stairs and when I was seen the Dr. said I had an infection from having my period while on the iud. While having the iud in I had no sex drive at all really. When my husband and I would have sex it was painful for me. My pelvic area hurt really bad and I would get cramps plus sometimes I would spot afterwards. I had the iud removed in September 2012 and It was the worst pain I have ever felt besides childbirth. I spent about 30 minutes laying on the table in pain because the lining of my uterus had grown over the iud. When they finally got it out i was lightheaded and had this horrible pain in the female area. My first period after having the iud removed I had really bad cramps to the point I didn't want to move. The second period after removal I had a very heavy flow and had huge and a lot of blood clots. I went thru a 18 pack of tampons in about 12 to 14 hours. I hurt so bad that I didn't want my husband to touch me or for my 4 year old son to lay on me or love on me. I have had mood swings since having the iud inserted and worse since having it removed. I can be sitting watching tv and then all of the sudden start crying for no reason. I am just now getting my sex drive back and my husband and I are wanting to have another baby so I had the iud removed. I still have some pain during intercourse and it is making it hard to enjoy intercourse. If I would have known that this is what I would have had to deal with while having and after removal of the iud I would have never got it. When I was pregnant I was told I had HPV and they looked at my cervix and told me the size and location on the hpv positive area. Then I had the Iud put in and a few months after having it put in I switched Doctors. Well my son was about a year old and the found the HPV and come to find out I had had it since I was pregnant they just missed it. I had to have the leap procedure done while having the IUD to remove the HPV. The IUD did nothing but cause issues for me but I was not ready for another baby and so I delt with it. I would never tell anyone to use the IUD.
BreannDecember 2, 2012 at 7:34 am
I had this IUD in for approx.12 month. I had constant bleeding similar to reverse menstruation, breakthrough bleeding spells and blood streaking and pink mucous after sex. I gained weight and developed cystic acne that left visible scarring to my skin. I had developed vaginal odor and multiple vaginal infections including yeast, bacterial vaginosis,and vaginitis. worst of all I found out I was Pr[Show More]I had this IUD in for approx.12 month. I had constant bleeding similar to reverse menstruation, breakthrough bleeding spells and blood streaking and pink mucous after sex. I gained weight and developed cystic acne that left visible scarring to my skin. I had developed vaginal odor and multiple vaginal infections including yeast, bacterial vaginosis,and vaginitis. worst of all I found out I was Pregnant and that my Mirena IUD had dissapeared all while thinking I was protected from pregnancy. My OBGYN said the device expelled. The expulsion had occurred without my knowing and led to my pregnancy which I ended up miscarrying . I want some compensation for my pain and suffering.
starrNovember 30, 2012 at 10:25 pm
I'm 24 yrs old and I've had the miriena for 3 yrs. I got it 7 weeks after I had my son and I've had migrains every day, no sex drive, depression and moodiness and back and hip pain and I've been to the doctors all over the town I live in and my obgyn refused to take it out when I asked 3 months ago. I feel like I'm. Failing as a wife and mother bacause I am in so much pain all of the time and I ca[Show More]I'm 24 yrs old and I've had the miriena for 3 yrs. I got it 7 weeks after I had my son and I've had migrains every day, no sex drive, depression and moodiness and back and hip pain and I've been to the doctors all over the town I live in and my obgyn refused to take it out when I asked 3 months ago. I feel like I'm. Failing as a wife and mother bacause I am in so much pain all of the time and I can't sleep and I'm loosing myself. At this point I'm just trying tofind a dr that will take it out.
NatalieNovember 29, 2012 at 2:19 pm
I had my Mirena IUD inserted in February 2008. As I have never given birth, I was told it may be a bit uncomfortable. Actually, the pain was incredible and drove me to tears, but I managed to get through it. I had irregular periods for months after the insertion along with excruciating cramps which I could only imagine might have been what a contraction would feel like. I had been warned that [Show More]I had my Mirena IUD inserted in February 2008. As I have never given birth, I was told it may be a bit uncomfortable. Actually, the pain was incredible and drove me to tears, but I managed to get through it. I had irregular periods for months after the insertion along with excruciating cramps which I could only imagine might have been what a contraction would feel like. I had been warned that I might have some of these issues for the first few months, but after that things should improve. Eventually, after 14 months, my periods stopped (which is great) and the cramps finally subsided. However, I was concerned with the sudden stop of my period that the IUD may have come out (neither I or my doctor could feel/see the strings during an examination). I was afraid I might have become pregnant. Turned out I wasn't pregnant and an X-ray confirmed that the IUD was in place, but had moved way up in my uterus to the point that my doctor may have to send me to the hospital to have it changed. I'm due for a new IUD in February 2013 and I'm terrified of going through the pain of having them poking around trying to find it AND of possibly going through a year's worth of agony again after a new one is inserted. After weighing the options, I've decided to go ahead with getting a new one, but I think I'll insist on having it done at the women's clinic in the hospital and that a local anesthetic be close by!!!!!
JeanieNovember 27, 2012 at 6:18 pm
I had my I.u.d inserted in 2008,and I haven't been right since.It hurt to get it put in, I was hurting for days after it was inserted.My periods are never regular, they are extremely heavy, I hurt really bad, I bleed after my menstrual cylce is over, and it doesn't smell like menstrual blood, it smells like death.I never feel good, and I'm always moody.Ive been to the hospital one time, but they d[Show More]I had my I.u.d inserted in 2008,and I haven't been right since.It hurt to get it put in, I was hurting for days after it was inserted.My periods are never regular, they are extremely heavy, I hurt really bad, I bleed after my menstrual cylce is over, and it doesn't smell like menstrual blood, it smells like death.I never feel good, and I'm always moody.Ive been to the hospital one time, but they did nothing for me, they just checked to make sure it was in place.It hurts to have intercourse, and the pain gets even more intense after intercourse is done.This has ruined who I am!!!!! I have no insurance to have it removed.What can I do?????? I cant take it anymore!!!!!!!!
brittanyNovember 27, 2012 at 7:12 am
I got Mirena in 2007 after I had my first son and I had it til 2009 when I got it removed, I had no problems at all with it. After my second son in 2010 I got it again and I have had nothing but problems. I now have abnormal paps, I have clots of blood, stomach cramps that hurt so bad I can't move and I can not have sex with my husband because my side hurts so bad I feel as though I'm in labor. I [Show More]I got Mirena in 2007 after I had my first son and I had it til 2009 when I got it removed, I had no problems at all with it. After my second son in 2010 I got it again and I have had nothing but problems. I now have abnormal paps, I have clots of blood, stomach cramps that hurt so bad I can't move and I can not have sex with my husband because my side hurts so bad I feel as though I'm in labor. I have asked my doctor for months to take it out and he saids everything I am going threw is normal and no other doctor can take it out. I do not know what else I can do as I don't have insurance anymore but I can not continue living like this. The pain is so bad I have to lay in bed and ball up in a ball, I fell as though I'm missing out on my kids growing up. Will someone please help me...what can I do?
ReginaNovember 26, 2012 at 1:33 am
A positive experience! I am currently 29. I had my first child at 18, my second at age 20 (while irregularly taking the pill), had Mirena for 5 years (down to the month) with mild cramps at insertion and no known side effects other than little to no regular periods, used the patches for birth control for a few months after that and shortly after stopping with the patches we became pregnant a 3rd t[Show More]A positive experience! I am currently 29. I had my first child at 18, my second at age 20 (while irregularly taking the pill), had Mirena for 5 years (down to the month) with mild cramps at insertion and no known side effects other than little to no regular periods, used the patches for birth control for a few months after that and shortly after stopping with the patches we became pregnant a 3rd time in 2009 at age 26. Did have to have an emergency appendectomy in the 3rd trimester, but don't believe that was anyway connected to Mirena. We are currently trying to have our 4th child, would like to add a daughter to our family of all boys, I have suggested Mirena to family and friends that are viable candidates for Mirena. Mirena is suggested ONLY for younger women who have had children and only one sexual partner with low risk of getting an STD.
HelenNovember 25, 2012 at 11:40 am
I have to be honest and say that I have not had any of the horrible experiences that many of you have had. I am 52 and had the mirena fitted 3 years ago due to very heavy periods. Yes it was a little uncomfortable when it was placed, and I did bleed irregular with stomach cramps for a couple of months. But It then settled and I found my periods were very light and no other problems at the time. I [Show More]I have to be honest and say that I have not had any of the horrible experiences that many of you have had. I am 52 and had the mirena fitted 3 years ago due to very heavy periods. Yes it was a little uncomfortable when it was placed, and I did bleed irregular with stomach cramps for a couple of months. But It then settled and I found my periods were very light and no other problems at the time. I am however thinking of removing it as just recently I have experienced soreness internally and I feel it might be where the string is, I also am experiencing irritation with my cervix like its trying to expell something. I am hoping to have it removed soon and see if this alleviates my issues. Otherwise I feel that I have been one of the fortunate, for the Mirena has done what it was supposed to have done for me.
DorothyNovember 24, 2012 at 6:21 pm
I got the Mirena in 2010 after I had my son. I have had all these symptoms at on time or anther. Mostly I deal with the dizzy spells, headaches everyday, not bleeding for months and then spotting twice a month, lower abdominal pain and cramping, and a extremely lowered sex drive. I would love more then anything to get it removed but I dont know where i can go without health insurance.
NicNovember 22, 2012 at 3:16 am
OB Doc how can a device that by its very nature requires the physical insertion of an object be THE safest brith control method around? Surely performing a procedure, by its nature entails more risk than swallowing a pill? The risks could include getting an infection, a perforation as you stated and migrating to other parts of the body. The pill is available OTC in Europe. It is a myth that it is[Show More]OB Doc how can a device that by its very nature requires the physical insertion of an object be THE safest brith control method around? Surely performing a procedure, by its nature entails more risk than swallowing a pill? The risks could include getting an infection, a perforation as you stated and migrating to other parts of the body. The pill is available OTC in Europe. It is a myth that it is dangerous. And futher more, the mini pill is just progesterone like the Mirena but it comes without the risks outlined above. Your dismissive attittude to the women's complaints here are offensive. Any goood doctor would take the time to listen to complaints, not reject them and imply that they are not real or related.I suggest you familiarise yourslf with the info sheet that comes with the Mirena because it lists many of these women's complaints as side effects.
andreaNovember 20, 2012 at 10:41 pm
has anyone been called or emailed about a case yet? i see people talking from 2011 till today and not one has said anything about being contacted? im tired of the pain and suffering over this stupid thing!!!!! the headaches. the low sex drive, the cramps and dizzyness and the list like all of yours just goes on and on!!!!!
TiffanyNovember 20, 2012 at 9:22 pm
I had my second IUD placed in April of 2012. After having it the first time in 2007, I thought it was going to work great! This time has been a night mare. My stomach stays bloated and I get nauseated when eating. It almost seems like I have more "fat" around my stomach area (and I see I'm not the first to say that). My doctor had a problem getting it in to begin with and said my uterus was high. [Show More]I had my second IUD placed in April of 2012. After having it the first time in 2007, I thought it was going to work great! This time has been a night mare. My stomach stays bloated and I get nauseated when eating. It almost seems like I have more "fat" around my stomach area (and I see I'm not the first to say that). My doctor had a problem getting it in to begin with and said my uterus was high. After irregular bleeding and pain in the bottom of my stomach all the time my OB doctor decided to do a vaginal ultrasound and found that the IUD had dropped and was at the bottom of my uterus! They then decided to remove it and ever since they removed it I have been bleeding for 7 weeks now! So I have pretty much been bleeding since April. Now my blood count is low, I am anemic, and I'm constantly tired! This is extremely frustrating to have 2 kids and not enough energy to get out of the bed during the day. I will now be having another ultrasound to see where to blood is coming from and why after being prescribed something to stop the bleeding, it won't stop. Has anybody else experienced the same symptoms?
micheleNovember 20, 2012 at 1:13 pm
I am on my second IUD i have had no problems decreased my bleeding amount threefold, currently I am now in full menopause so I will be having it removed because i have to take progesterone with HRT. I really cant understand all these problems it is a low dose of progesterone if I am not mistaken, and that is in almost every birth control pill ??? And as for all the side affects I think its listed [Show More]I am on my second IUD i have had no problems decreased my bleeding amount threefold, currently I am now in full menopause so I will be having it removed because i have to take progesterone with HRT. I really cant understand all these problems it is a low dose of progesterone if I am not mistaken, and that is in almost every birth control pill ??? And as for all the side affects I think its listed on most meds before you take them???clots, have been around forever with the use of birth control no??
ob docNovember 20, 2012 at 12:04 am
mirena is the safest most effective method of birth control in the world. It doesn't migrate if you have a perforation it happened at the time of insertion it does'nt mean the doc was incompetent it happens. like strokes and blood clots from pill. side effects are rare but acne and depression and weight gain may be reported 1% of the time doesn't mean it is caused by mirena
WendyNovember 19, 2012 at 9:12 pm
I had the mirena inserted in February 2012, right after I had my son. I had severe abdominal pains when it was inserted. I also suffered headaches, depression, anxiety, loss of libido, night sweats. I finally had it removed in March 2012, my husband and I decided it was time for another child. Since then I have had no luck getting pregnant, having abdominal pains, and I went to my OB in September [Show More]I had the mirena inserted in February 2012, right after I had my son. I had severe abdominal pains when it was inserted. I also suffered headaches, depression, anxiety, loss of libido, night sweats. I finally had it removed in March 2012, my husband and I decided it was time for another child. Since then I have had no luck getting pregnant, having abdominal pains, and I went to my OB in September only to find out I am not ovulating. I'm hoping that I will be able to have more children as I am only 22, but it is not looking good. I am very upset that there was no concerns from my doctor, or even any information about how bad the aide effects of this BC are.
KristinNovember 18, 2012 at 4:58 pm
hello i got mirena inserted in 2005 pretty soon after having my first and only child i am 25 now and i heard bout this bull about the birth control i use i am concerned i have had alot of stomach pains in the past year or so and have got kidney infections is any of my side effects due to this?
YvetteNovember 17, 2012 at 7:35 pm
The device perforated my uterus. I had to go to a surgery so it could be removed, I was luckly because it was near my intestines and it could kill me.
GigiNovember 16, 2012 at 2:49 pm
To: Comment by teresa on 18 September 2012: Yes, UTIs and IUDs are related. See, and read “discussion”: http://www.urologyjournal.org/index.php/uj/article/viewFile/156/156 I had a copper IUD placed 5 years ago and since then I've had a UTI. I asked years ago if there could be a relationship and was told none at all. It's time to replace my IUD and the gynecologist recommends Mirena. My husband s[Show More]To: Comment by teresa on 18 September 2012: Yes, UTIs and IUDs are related. See, and read “discussion”: http://www.urologyjournal.org/index.php/uj/article/viewFile/156/156 I had a copper IUD placed 5 years ago and since then I've had a UTI. I asked years ago if there could be a relationship and was told none at all. It's time to replace my IUD and the gynecologist recommends Mirena. My husband said it’s too much a coincidence that my UTI’s started 5 years ago, so I looked up IUD and UTI and bumped into this page of Mirena horrors. I have been to two urologists in two different continents, have had a urinary tract endoscopy performed, I take a low dose of antibiotics daily, have received horse doses of pills and things only work for a while. At the moment I’m having a UTI once or twice a week and I’m trying to make it with the AZO pills to next week when I’m having the IUD removed. I will never start on the Mirena or any other IUD ever again. I think and hope my UTI will go away (like I’ve read has happened to most here). I feel I have a problem, but when I read these Mirena horrors, I think mine is small. I feel for you and your daughter. Maybe an option for your daughter are the contraceptive injections, they too stop the period.
MackNovember 16, 2012 at 1:23 pm
I had the mirena inserted in April of 2010 after 6 weeks of birth. My doctor explained the side effects because that his job. I understood the side effects but still decided mirena was the best way to go. It took a little over a year but my cycle is now just spotting but it is irregular. I check to make sure my strings are still in every now and then. Everything has side effects especially somethi[Show More]I had the mirena inserted in April of 2010 after 6 weeks of birth. My doctor explained the side effects because that his job. I understood the side effects but still decided mirena was the best way to go. It took a little over a year but my cycle is now just spotting but it is irregular. I check to make sure my strings are still in every now and then. Everything has side effects especially something that's going into your body. It also contains hormones so yes it's possible to have other things happen. If your doctor didn't explain all the risks that would come then that's who should be mad at right now, not the makers of mirena.
HeatherNovember 15, 2012 at 3:41 pm
I have had the mirena in since Dec 2007, this has been the worse 5 years of my life. I am getting this removed 12/11/12. My Dr. told me they would have to sedate me to remove it. i have heard this is a very painful procedure . When i had the IUD inserted I was 215 lbs. I am weigh 295 lbs. I had planned on getting another Mirena put in after i get this one removed , but after reading all these conc[Show More]I have had the mirena in since Dec 2007, this has been the worse 5 years of my life. I am getting this removed 12/11/12. My Dr. told me they would have to sedate me to remove it. i have heard this is a very painful procedure . When i had the IUD inserted I was 215 lbs. I am weigh 295 lbs. I had planned on getting another Mirena put in after i get this one removed , but after reading all these concerns i am definetly not going to chose this IUD ever again. I have had heart palpatations,which i saw a cardiologist for and had a heart cath done because they thought my artieries were clogged and when they went in there nothing was wrong.. stomach issues, depression, Anxiety, Tiredness, Low sex drive, some pain with sex, weight gain, headaches, blurred vision, dizzy spells, high blood pressure, acne, and other emotional problems. I do not recommend this IUD at all. I am 33 years old and feel like i am 65. I dont want to do anything but sleep.
DevanyNovember 15, 2012 at 5:33 am
I received the Mirena IUD in May 2009 and had no problem with it for 3 years, until October 2012. Out of nowhere, a sudden horrible pain came over me and after several hours of severe pain I went to the hospital. My IUD caused an absess to form in my fallopean tube, and I had a hardcore Pelvic Inflamatory Disease. I was in the hospital for 8 days, and I have been on IV antibiotics for over a mo[Show More]I received the Mirena IUD in May 2009 and had no problem with it for 3 years, until October 2012. Out of nowhere, a sudden horrible pain came over me and after several hours of severe pain I went to the hospital. My IUD caused an absess to form in my fallopean tube, and I had a hardcore Pelvic Inflamatory Disease. I was in the hospital for 8 days, and I have been on IV antibiotics for over a month now.
ShaquitaNovember 15, 2012 at 12:51 am
I had the IUD for 4 years I had constant lower abdoman pain along with back pains and awful migranes. I also had overian cysts which was the worst pain ever. My IUD came out on its on after I reached my 4 year mark since then my periods have been very irregular and most times I bleed for more than a week and alot of times have periods every 3 to 4 weeks when before i only had them once every 30 da[Show More]I had the IUD for 4 years I had constant lower abdoman pain along with back pains and awful migranes. I also had overian cysts which was the worst pain ever. My IUD came out on its on after I reached my 4 year mark since then my periods have been very irregular and most times I bleed for more than a week and alot of times have periods every 3 to 4 weeks when before i only had them once every 30 days r so. For the past 2 years me and my husband have tried to have another job and its just constant disappointment. I am 23 years old if I would have known that this form of birth control would have made me infertale I would not have chosen it because now im afraid my child will never have siblings and I will never be able to have another child for my husband
AScott RNNovember 14, 2012 at 10:35 pm
WOW!!!! I just seen MIRENA RECALL commercial on TV. So here i am looking at this website. I had my MIRENA placed in Nov 2010 after having my second son in Aug 2010, in which i got pregnant for while on birthcontrol pills. All of these symptoms listed above by others I have expereinced in the last 2 years. Abdonminal pain, crazy bleeding schedule, no sex drive, severe mood swings, (alomost caus[Show More]WOW!!!! I just seen MIRENA RECALL commercial on TV. So here i am looking at this website. I had my MIRENA placed in Nov 2010 after having my second son in Aug 2010, in which i got pregnant for while on birthcontrol pills. All of these symptoms listed above by others I have expereinced in the last 2 years. Abdonminal pain, crazy bleeding schedule, no sex drive, severe mood swings, (alomost caused divorce) in which I went to OBGYN and got put on high dose of Zoloft for post part depression, PCP has tried to wean off the Zoloft but not successful continue to have the swings. I am continuously drained of energy, and not even going to get started on my weight gian (unexplained) All i can say is OMG. I guess Im calling my OB in the morning to get this thing out!!! Hopefully my health will return, and dont have any complications afterwards.
MichelleNovember 13, 2012 at 2:38 am
I had mirena put in April 2009 after the birth of my daughter in january 2009 at age 16 (judge all you want). My doctor told me that I needed to get mirena and didn't even discuss alternatives or side affects with me so being young and just having a child I trusted her and got it. After a year and a half of severe abdominal pain, ultrasounds and X-rays, my doctor told me it was "settling in". I al[Show More]I had mirena put in April 2009 after the birth of my daughter in january 2009 at age 16 (judge all you want). My doctor told me that I needed to get mirena and didn't even discuss alternatives or side affects with me so being young and just having a child I trusted her and got it. After a year and a half of severe abdominal pain, ultrasounds and X-rays, my doctor told me it was "settling in". I also experienced a loss of sex drive and pain during sex which at age 17,18,19, and 20 I should be pretty much ready for sex anytime. No drive. So a couple years have passed now with irregular bleeding and still severe cramping and abdominal pain my doctor hasn't mentioned anything about the lawsuits or even suggested that my problems could be caused by this miracle drug. Now, I've been bleeding for 3 weeks and am scared that something is really going wrong and after finding this information on my own I dont know what to do. I'm only 20 now and terrified I may not be able to have more kids :'( any advice?
KatrinaNovember 12, 2012 at 6:45 pm
Omgoodness now I am so worried. My symptoms have been on and off for quite awhile: Untreatable hormonal ance Breast swelling Cramping Mood swings Weird hair growth infacial area :( Severe random headaches Dizzy spells Racing heart And when I go into the doctors, they look at me like I am crazy and looking for attention. Makes me feel like they do not care. I have talked about getting it removed o[Show More]Omgoodness now I am so worried. My symptoms have been on and off for quite awhile: Untreatable hormonal ance Breast swelling Cramping Mood swings Weird hair growth infacial area :( Severe random headaches Dizzy spells Racing heart And when I go into the doctors, they look at me like I am crazy and looking for attention. Makes me feel like they do not care. I have talked about getting it removed or checked or my hormone levels checked and they always turn me down. Well this Thursday its gone I skipped the doctor and am going straight to my ogyn. I'm glad I'm not the only one. Just hopeand pray my body will go back to normal and I can have kidds still. <3 praying for all of us.
AngelNovember 9, 2012 at 4:10 pm
ON A GOOD NOTE: I have had a Mirena IUD in since 2004, had one removed and another put in after 5 years!! No problems what so ever. I understand that a bunch of people may have had problems and everyones bodies and activities are different which will make the different stories you have heard come true. But they should not scare everyone off, I love my birth control and never having to worry about [Show More]ON A GOOD NOTE: I have had a Mirena IUD in since 2004, had one removed and another put in after 5 years!! No problems what so ever. I understand that a bunch of people may have had problems and everyones bodies and activities are different which will make the different stories you have heard come true. But they should not scare everyone off, I love my birth control and never having to worry about protection or a monthly cycle. I did gain a little weight but through excercise and diet I lost it too, so its not just the IUD causing it. I feel like a a women we are hormonal and moody anyways and that with or without the IUD we are not going to always be happy. I am sorry to hear that some bad things have happend to people but I am hear to say that some good things have come out of Mirena too!!
AmandaNovember 7, 2012 at 9:50 pm
I had my son May 24th of this year and had the mirena put in 4 weeks after. It was very painful and I had bad cramps. I always got sharp pains on my left side and I ignored them thinking it was from the mirena just getting used to my body. I went back 2 weeks later and my nurse couldn't find the strings with her fingers or through an ultrasound. I had to have an xray done and it had went through m[Show More]I had my son May 24th of this year and had the mirena put in 4 weeks after. It was very painful and I had bad cramps. I always got sharp pains on my left side and I ignored them thinking it was from the mirena just getting used to my body. I went back 2 weeks later and my nurse couldn't find the strings with her fingers or through an ultrasound. I had to have an xray done and it had went through my uterine wall and was sitting in my abdomen. I had never had a surgery so I was not happy to hear I was having a laparoscopy to remove it. I was only supposed to have two incisions but they made a 3rd because it had embedded itself in some tissue so they had to cut that out with it. I had no stretch marks from either of my children and now I have 3 ugly scars all from this stupid birth control!!!
JenniferNovember 7, 2012 at 1:13 am
I have a story for you! About ahead two years ago I had the mirena in placed and let me tell you it was the absolute worse thing I have ever experienced. I went to my ob doctor and while he placed it it hurt so bad a almost jumped off the table. I never bother to complain however because I thought that was to be expected. A few days later I still felt the same plan and like a knive stabbing me in [Show More]I have a story for you! About ahead two years ago I had the mirena in placed and let me tell you it was the absolute worse thing I have ever experienced. I went to my ob doctor and while he placed it it hurt so bad a almost jumped off the table. I never bother to complain however because I thought that was to be expected. A few days later I still felt the same plan and like a knive stabbing me in my lower back. I called my ob doctor and they just told me it was going to take a while for my body to get use to it. So I just blew it off! On top of it all my relationship went rocky cause my sex drive went right out ghe window. Little did I know a year and some change goes by and I still have this annoying pain in my abdominal area and my back, I have a very high pain tolerance so I once agian put it off. Until my check up for the mirena it was late cause I didn't have insurance once I was able to get into the doctor and explained to them my pain problem I thought it was the stings so I had them cut down. Nontheless that didn't work either a few months later the pain increased. I started having sharp pains travel from my vagina to my inner leg and make my privates feel like it was asleep. Now I know damn well that wasn't normal so I called and thankfully they removed it. It has been about two months now and unfortantly I'm still having the same pain . It never went away! So within the next week I'm going to the doctor to see why. To top it all I get told I have to much scar tissue around my uterus and can not have anymore children. I have to get a hesterectomy because they are unable to tie my tubes due to all the scar tissue :(
DennyielNovember 6, 2012 at 8:58 pm
I had the Mirena IUD put in after my daughter was born in 2007. after having it for two years I had it removed, It was causing pain in my abdomen. After I decided not to use any form of birth control, I have been unable to conceive, which would be 3 years now. I am also currently having serious problems with cysts on my ovaries which I am still dealing with today, I am getting one checked in the n[Show More]I had the Mirena IUD put in after my daughter was born in 2007. after having it for two years I had it removed, It was causing pain in my abdomen. After I decided not to use any form of birth control, I have been unable to conceive, which would be 3 years now. I am also currently having serious problems with cysts on my ovaries which I am still dealing with today, I am getting one checked in the next few days to make sure I don't need surgery to have it removed or that it's not cancerous. I have never had so many female problems until I got that stupid IUD. And The radiologists that have done my internal ultrasounds have asked me if I had the Mirena IUD because it changed the way my cervix looks? I was never informed about that. and I never told them I had it they just knew. I wish I could go back and never have gotten it, It's not worth all this pain. I would never tell anyone to get this device.
ShalainaNovember 5, 2012 at 9:46 am
Omg tears in my eyes reading thru these comments, just heard about this lawsuit and are realizing that Iam not the only one!!!! I have every side affect that I have read and it just blows my mind that I know what's causing it now. Doctors won't do nothing but put me on anti depressants and send me home. I now see a ciropractor once a week and just so tired of being in massive pain allday everyday![Show More]Omg tears in my eyes reading thru these comments, just heard about this lawsuit and are realizing that Iam not the only one!!!! I have every side affect that I have read and it just blows my mind that I know what's causing it now. Doctors won't do nothing but put me on anti depressants and send me home. I now see a ciropractor once a week and just so tired of being in massive pain allday everyday! Iam calling my doctor tomorrow and getting this out of me....I too just want my life back!
LyndsayNovember 5, 2012 at 5:06 am
I had my Mirena inserted in October 2007 right after the birth of our third child. Up until this moment my experiences with this BC has been tolerable. I have had mild weight gain, acne and no cramping or sickness. So I thought that is what I wanted for the next five years. Well two weeks ago I went in to my OB and had my annual PAP done. She told me she could see my strings. Ok so good right[Show More]I had my Mirena inserted in October 2007 right after the birth of our third child. Up until this moment my experiences with this BC has been tolerable. I have had mild weight gain, acne and no cramping or sickness. So I thought that is what I wanted for the next five years. Well two weeks ago I went in to my OB and had my annual PAP done. She told me she could see my strings. Ok so good right? Well with in a week, and obviously after intercourse, I have been sick, headaches, stomach tenderness, pinching all the obvious preggo signs. So last night I checked my strings. They were so low they were almost on the outside. The next day. It fell completley out. Could my PAP have dislodged it in any way. Nothing else negative has happened since I got the IUD. OH and could I be preggo?
AnnaNovember 5, 2012 at 3:59 am
I had the mirena iud put in 8 weeks after i had my baby girl in august of 2010. I had mild pain when it first got put in but it went away within a week so i never really worried about it. my periods were always regular which i liked because my periods were never regular. I went from being the happiest mother to a mother that didnt want to do anything but go wild or cry. well december of 2010 i tri[Show More]I had the mirena iud put in 8 weeks after i had my baby girl in august of 2010. I had mild pain when it first got put in but it went away within a week so i never really worried about it. my periods were always regular which i liked because my periods were never regular. I went from being the happiest mother to a mother that didnt want to do anything but go wild or cry. well december of 2010 i tried to commit suicide by od. i went to rehab and tried to get over the depression. well june of 2011 i did the same again. i stayed depressed for months but i managed. I never felt myself. November of 2011 I started bleeding really heavy. Heavier then I had ever bleed. after bleeding for a month straight and having pains that i compare to labor pains it hurt so bad i decided to go to the obgyn to see what was wrong. They said that the mirena had dislodged. they took it out a week later. Since then my depression has got so much better. but intersoursse hurts. for the longest time i didnt want him to touch me cuz it hurt so bad. my menstrual cycle has become painful and im always exhausted from the time i wake up to the time i go to bed. that week is the terrrible. my fiance is so worried that I will never be the same sexual wise and that we wont be able to have any kids. I had my daughter before I met him and I really want to be able to give him a son one day. I do know if any thing can be proven to be from the mirena but I feel it did.
DianaNovember 4, 2012 at 3:34 am
after having the mirena inserted in 2007 after my daughter was born had gained nearly 40 lbs and had it removed in 2010 ever since been unable to conceive another child ....more than a nightmare now I'm left with a nightmare of never giving my daughter a sybling
IrmaNovember 2, 2012 at 3:21 pm
I have had Mirena going on 9 years now, With my first round, I ended up in hospital with excruciating pain in my abdomen, could not walk. When I got to hospital, i was told that I had an ovarian cyst that ruptured while at the hospital. I was told that it was a normal side effect and should not be concerned. Got my 2nd round five years later and have had excrutiating Cluster/Migraine headaches, ha[Show More]I have had Mirena going on 9 years now, With my first round, I ended up in hospital with excruciating pain in my abdomen, could not walk. When I got to hospital, i was told that I had an ovarian cyst that ruptured while at the hospital. I was told that it was a normal side effect and should not be concerned. Got my 2nd round five years later and have had excrutiating Cluster/Migraine headaches, have gained 30 lbs that i can not seem to lose even though I excercise. Have had several fainting spells, and lord help me during my menstral cycle. I am incapcitated. I had no idea that all of my symptoms could be a result of Mirena until today. I will be making a call to my OBGYN to have this IUD removed asap!
queenaNovember 2, 2012 at 12:09 am
I had my first one inserted Sept. 2007, it worked pretty good. Had it taken out, it took me 6 months to get pregnant. I decided to get it put back in, but now I've seen the commercial I think I'm going to have it taken out. I can't risk going through the things that I'm hearing that's going on with these other ladies. I'm also going to go get myself checked out, cause I've also been having migrain[Show More]I had my first one inserted Sept. 2007, it worked pretty good. Had it taken out, it took me 6 months to get pregnant. I decided to get it put back in, but now I've seen the commercial I think I'm going to have it taken out. I can't risk going through the things that I'm hearing that's going on with these other ladies. I'm also going to go get myself checked out, cause I've also been having migraines. If there seems to be something else serious happening, I'll be pursuing a lawsuit.
jessicaNovember 1, 2012 at 7:23 am
I suffered from a uterus perforation. At the time I didnt really know what that met. Then one of my friends who knew what i went through told me about this page and I just couldnt believe it. On this page I have found women on here who sound a lot like me.1st NO sex drive , doesnt help my relationship, strange movement, moodiness.,ex. I would love to know about uterus perforation and how serious [Show More]I suffered from a uterus perforation. At the time I didnt really know what that met. Then one of my friends who knew what i went through told me about this page and I just couldnt believe it. On this page I have found women on here who sound a lot like me.1st NO sex drive , doesnt help my relationship, strange movement, moodiness.,ex. I would love to know about uterus perforation and how serious is it?Stories or advice, anything will help.:) Thanks
ShannonOctober 29, 2012 at 10:44 pm
I had mirena placed in July 2011. I still bleed about every 2 weeks, but the worst is left lower pelvic pain that I experience on a regular basis. After reading all of these posts I know I need to make an appt and have it removed and have my left ovary checked because I have a feeling its a cyst. I also have experienced mood swings and decreased libido since I've had this, which I never had a prob[Show More]I had mirena placed in July 2011. I still bleed about every 2 weeks, but the worst is left lower pelvic pain that I experience on a regular basis. After reading all of these posts I know I need to make an appt and have it removed and have my left ovary checked because I have a feeling its a cyst. I also have experienced mood swings and decreased libido since I've had this, which I never had a problem with before. If I end up having a cyst I will have no problem pursuing a lawsuit with Bayer.
CharleneOctober 28, 2012 at 10:06 pm
I had Mirena placed in March 2012 for "heavy bleeding" not for contraceptive reasons. I am 40 and have never been able to conceive a child. The procedure was very painful. The OBGYN said he was only able to insert the device 5 cm which he said was odd, but he did it anyway. In May 2012 I started gaining weight, started having migraine headaches and still was bleeding everyday since the Mirena was [Show More]I had Mirena placed in March 2012 for "heavy bleeding" not for contraceptive reasons. I am 40 and have never been able to conceive a child. The procedure was very painful. The OBGYN said he was only able to insert the device 5 cm which he said was odd, but he did it anyway. In May 2012 I started gaining weight, started having migraine headaches and still was bleeding everyday since the Mirena was placed. I had several tests to find out why migraines. I have passed out on the floor many times in my house. I was hospitalized for 4 days due to debilitating migraines and fatigue. Now its October... Have had a period everyday since Mirena was placed, I have gained 18 pounds, on all these meds now to treat migraines, I had Mirena removed on October 23, it was very painful as well, also had a endometrial biopsy that same day / visit. I'm still bleeding and still waiting on the results. A friend told me today about a commercial she saw for Mirena Lawsuits, I told her I would check it out and found this page and read others comments. Now I know it's not just me. Most of all I am experiencing and have been since March is all caused by Mirena. I can't believe what I am finding out now. I would not recommend Mirena to anyone.
Victoria W.October 26, 2012 at 9:20 pm
I had Mirena put in when I was 18, Im 21 now and I seen the commercial for mirena lawsuit and ever since then I have been super scared about it. It was the most painful experience having it put in and I bled for a year straight afterwards, now I have irregular spotting and Im terrified to have it removed but I want it out! and I dont have insurance so im really concerned as to how much it will cos[Show More]I had Mirena put in when I was 18, Im 21 now and I seen the commercial for mirena lawsuit and ever since then I have been super scared about it. It was the most painful experience having it put in and I bled for a year straight afterwards, now I have irregular spotting and Im terrified to have it removed but I want it out! and I dont have insurance so im really concerned as to how much it will cost.
ElisabethOctober 24, 2012 at 4:16 pm
I am 28 years old. I have had the mirena for 5 + years now. They inserted it after a month after the birth of my second child. I went to get it removed considering they told me that I should only have in place for 5 years. They could not get it out! It is stuck!!! Talk about pain. I thought I was giving birth to a third child. I have also had a decreased sex drived, high level of moodiness, and la[Show More]I am 28 years old. I have had the mirena for 5 + years now. They inserted it after a month after the birth of my second child. I went to get it removed considering they told me that I should only have in place for 5 years. They could not get it out! It is stuck!!! Talk about pain. I thought I was giving birth to a third child. I have also had a decreased sex drived, high level of moodiness, and lack of energy, sore muscles, back pain, lumps in my breast, and breast tenderness! I am sooooo scared to get this mirena taken out. It is still in me!!!! After reading all these post I am scared to get it out and expiernence withdrawl effects as well. I don't know what to do, this is not right!!!! I can not believe this many women have experienced these same issues as well!!!!! This is a nightmare!!!!
christineOctober 22, 2012 at 9:57 am
31 years ago I had a laparotomy for a removal of an iud that got stuck in my colon. The following year I had a still born baby because of a retro placental blood clot. I believe this was caused by the damage to my uterus that this had caused At the time I felt the baby could have been saved had the medical profession not messed up my care but could not face litigation because of all the grief I su[Show More]31 years ago I had a laparotomy for a removal of an iud that got stuck in my colon. The following year I had a still born baby because of a retro placental blood clot. I believe this was caused by the damage to my uterus that this had caused At the time I felt the baby could have been saved had the medical profession not messed up my care but could not face litigation because of all the grief I suffered. I gave birth to a healthy baby the following year even at the age of 38 years of age. But I have recently had to have my gall bladder removed by keyhole surgery as I have diverticulosis and have suffered a lot of pain in my life from depression because of the loss of my baby, overweight because of side effects from anti depressants for many years, loss of self esteem and it has ruined my life as I am now 67 and have a lot of pain one way and another
KelliOctober 22, 2012 at 6:10 am
I've always been weary about my mirena. I got it in sept 2009, 1 month after I had my son. The first month was terrible. I bled the whole month, cramped, barely wanted to get out of bed. I bled so much I thought I was dying or something because I'd go thru like 10 pads a day. Not to mention I was so moody, my boyfriend would do the slightest thing and I would go over the deep end and yell and end [Show More]I've always been weary about my mirena. I got it in sept 2009, 1 month after I had my son. The first month was terrible. I bled the whole month, cramped, barely wanted to get out of bed. I bled so much I thought I was dying or something because I'd go thru like 10 pads a day. Not to mention I was so moody, my boyfriend would do the slightest thing and I would go over the deep end and yell and end up crying. And now as time goes on it seems like the symptoms get WORSE AND WORSE. Still moody, random ass periods, I never know when they're coming. Cramps. Back pain. And now extreme dizziness. And I always feel like I'm pregnant every month. It's faking me out and I'm getting so sick of it. I went to the ER a few months ago because since I've had mirena I've had about 10 UTIs and 2 kidney infections and they did an xray on my pelvic area because they couldnt find the strings. They said it LOOKS like my mirena is still in the right place but I'm really worried that it's stuck on the wall or something. My boyfriend and I want to have another baby soon, and after reading all these comments I'm terrified I either wont be able to, or that I'm going to get pregnant with it still inside me. Which I actually know someone who got mirena in 2010 the same as me and she's had two miscarriages in the past 7 months with mirena in. 1/1000 chance my ass.
JessicaOctober 21, 2012 at 9:48 pm
I got the mirena iud in April 2012 had it for 5 months then my husband and I decided that since our daughter will be 5 soon we wanted to try for baby #2.So I went September 25th,2012 and had it removed.My period started back the 27th,it was normal lasted 5 days.That weekend my husband came home and we had bedroom time.That Monday I started feeling horrible,awful craps,stomachs pain,lower back pain[Show More]I got the mirena iud in April 2012 had it for 5 months then my husband and I decided that since our daughter will be 5 soon we wanted to try for baby #2.So I went September 25th,2012 and had it removed.My period started back the 27th,it was normal lasted 5 days.That weekend my husband came home and we had bedroom time.That Monday I started feeling horrible,awful craps,stomachs pain,lower back pain,nausea,migraines (which ivebhad now for 2 weeks),horrible mood swings,it was almost comparable to pregnancy symptoms,so I took 4 pregnancy test and all negatives.Then last Thursday I ended up goin tothe ER around 10:30pm and not leaving there til 1:30 or 2 am.I've been having pain when I pee and it feels like my insides are coming out,I explained all this to the er Dr and his nurse.They did a urine sample,negative on uti ,bladder infection and kidney infection,so the proceeded to do a CTSCAN and that showed no problems and all my blood work was great,so they did a pelvic exam and that was fine.The nurse told me that it could be from the iud.My OB wasn't in her office Friday so I'm goin to make an appt with her one day this week and try to figure out what's goin on.And some days this pain is unbearable!
AlejandraOctober 18, 2012 at 2:33 pm
I had mirena inserted after the birht of my daughter,in Nov 2006, when my uterus was in normal shape. After the 5 year period I went and go it removed only to find out that I have uterine fibroids, and might no be able to have babies anymore. I do not know if there is a correlation with the contraceptive method of myrena, I know they are caused by hormonal imbalance and the mirena was full of horm[Show More]I had mirena inserted after the birht of my daughter,in Nov 2006, when my uterus was in normal shape. After the 5 year period I went and go it removed only to find out that I have uterine fibroids, and might no be able to have babies anymore. I do not know if there is a correlation with the contraceptive method of myrena, I know they are caused by hormonal imbalance and the mirena was full of hormones. It would be intresting to know how many women are experiencing this with no previous medical history of it. Im worried, I will have a uterine ultrasound to better diagnose and treat. Definately a method that should not be used by young women that expect to concive a chiled at some point in their lives.
AlesiaOctober 16, 2012 at 4:12 pm
I got the mirena the first time in 20009 ihad it in for about 5-6 months I had non stop heavy bleeding, horrible mood swing and crapping and loss of sex drive. My husband could also feel it. Not to mention horrible migraines. I got pregnant shortly after and now have a beautiful 2 year old after 1 month of bedrest and losing my job. She was born three weeks early my water broke prematurely and she[Show More]I got the mirena the first time in 20009 ihad it in for about 5-6 months I had non stop heavy bleeding, horrible mood swing and crapping and loss of sex drive. My husband could also feel it. Not to mention horrible migraines. I got pregnant shortly after and now have a beautiful 2 year old after 1 month of bedrest and losing my job. She was born three weeks early my water broke prematurely and she was in the NICU for a week this. :-/ since I have two children and were unsure if we wanted another I desided to again try the IUD thinking it was the best option being that I'm a migrane suffer and there really are no other options. I now have had it for a 1 1/2 years almost 2 actually. I have have had horrible acne which I have never had I switched shampoos even think everything but the dang IUD, swallon and tender Brest, palpitations fainting spells the doctors though may be seizure activity but test were inconclusive what feel like the occasional galbladder attack but ultrasound show nothing. Tingling in my limbs and blurred Vision serve lower pain and burst of anger. And bleeding after intercourse everytime The newest thing is a feel like am pregnant all the same symptoms. Done to frequent urination and sometimes even incontinent :-( horribly embarrassing might I add hot flashes. Isomia etc the list goes on I though I was crazy doctors couldn't find ANYTHING except a couple of disk hernias that they said were smaller and maybe could cause the pain. I since have had treatment injection nothing isn't even remotely better. I even mentioned to my family the other day about it possibly being the IUD the is how I found this website. This reidderates my feeling and I will being getting this removed right away. I can feel the strings so hopefully no complication occur and it is removed with ease I'm slightly stressed about it now.
A.venegasOctober 16, 2012 at 3:54 pm
I have gotten the iud in the past after my second child I thought it was normal not to have a period .I had it in for 5 years in that time I gained alot of weight I was depressed I would constantly get infections thought something was wrong with me it would hurt to have intercourse but I would ignore it . I had it removed after the give years past it was easy to get pg 6 Weeks after I gave birth I[Show More]I have gotten the iud in the past after my second child I thought it was normal not to have a period .I had it in for 5 years in that time I gained alot of weight I was depressed I would constantly get infections thought something was wrong with me it would hurt to have intercourse but I would ignore it . I had it removed after the give years past it was easy to get pg 6 Weeks after I gave birth I got the iud again I had a period for about 2 months thought it was normal started to get very Moody started to very bad pain in my pelvic area but I would ignore it I would get sharp pains in my butt that I would jump up pain in my back . I started to get a white discharge then it turned light green then a funcky smell started happening I went to my dr she said I had a bacteria infection gave me antibiotic it worked for a couple days it would hurt so bad to have sex my husband was scarred to touch me I would bleed everyone after sex finally I couldn't take the pain I got rushed to the ER dr told me I had pelvic inflammatory disease he had to surgically remove my iud and since then i'm on medicine and nothing seems to be working . I just want my life back i'm only 24 with no sex life .
melanieOctober 12, 2012 at 2:30 am
I even asked my object/gyn if I had to check the threads...she said no! I'm curious if these other doctors advised their patients to check?
JudyOctober 11, 2012 at 9:27 pm
I had the Mirena inserted in July of 2011. I had a period for 4 months straight and called to see if that was normal. I was put on hormones to help regulate my cycle. I thought that was the point of this thing outside of BC. I had horrible cramps, particularly on the left side. I gained 15 pounds, no sex drive, HORRIBLE discharge and odor, my husband did not like it during sex, it poked him. I was[Show More]I had the Mirena inserted in July of 2011. I had a period for 4 months straight and called to see if that was normal. I was put on hormones to help regulate my cycle. I thought that was the point of this thing outside of BC. I had horrible cramps, particularly on the left side. I gained 15 pounds, no sex drive, HORRIBLE discharge and odor, my husband did not like it during sex, it poked him. I was lethargic most of the time. I have migraines normally, but this made it so much worse. I felt like I was PMS-ing or on my period 24/7. The answer I got was- every woman's body is different. I just had it removed 2 days ago, and I feel better. I know it will take some time to get back to how I was, but I am better than I was then. My sister had it also and she said she was fine and no symptoms, other than the weight gain she refuses to admit. She had it out to get pregnant, and she waited the time needed and had a miscarriage. I see a trend. To advertise you can take it out and get pregnant soon seems to also be another LIE!!!!! This thing needs to be taken off the market. Makes a lady go crazy!
AndreaOctober 11, 2012 at 5:26 pm
I too had a mirena for the last 4 years! This first 1 was placed after I had my son in 2008 and in 2009 I had to get another one placed because it had came out. Over the past years I started to get a rash on my feet & legs but did nothing but get worse over time & now I am two-toned on the bottom of my shins & feet. I like most post I read asked my doctor & dermatologist could the[Show More]I too had a mirena for the last 4 years! This first 1 was placed after I had my son in 2008 and in 2009 I had to get another one placed because it had came out. Over the past years I started to get a rash on my feet & legs but did nothing but get worse over time & now I am two-toned on the bottom of my shins & feet. I like most post I read asked my doctor & dermatologist could the mirena be the cause & like all other doctors there answer was, No! I decided to remove the Mirena yesterday and hope my legs will get back to normal and the other issues of daily headaches, libido loss, and weight gain go back to normal. I was given a new birth control method Necon 1/35 pills but I rather wait awhile before starting something right away.
TracyOctober 11, 2012 at 4:41 am
I had the marina inserted in feb 2009.. almost four yrs ago. I have gained about 15lbs....I have severe anxiaty, mood swings and recently, I have been exper. really bad pain in my lower back, neck, spine and a lot of pressure in my head! my breasts are sore and I feel sick to my stomache everyday....I made a doc. apt for tom to get this thing out of me!!!! I will let u all knw how it goes. I just[Show More]I had the marina inserted in feb 2009.. almost four yrs ago. I have gained about 15lbs....I have severe anxiaty, mood swings and recently, I have been exper. really bad pain in my lower back, neck, spine and a lot of pressure in my head! my breasts are sore and I feel sick to my stomache everyday....I made a doc. apt for tom to get this thing out of me!!!! I will let u all knw how it goes. I just want to get back to feeling like me again.
melanieOctober 11, 2012 at 1:48 am
Gotta love the hair loss and shortness of breath. I also get the tingling in my hands and arms. I pray that once this is out i will go back to normal. Im 33 and used to work out 4x a week and my job i do alot of physical activity. My BMI was 21 now its a 30 . I want to be around and watch my child grow and enjoy him. Sorry for all these comments but i dont know what else to do right now. Ive been [Show More]Gotta love the hair loss and shortness of breath. I also get the tingling in my hands and arms. I pray that once this is out i will go back to normal. Im 33 and used to work out 4x a week and my job i do alot of physical activity. My BMI was 21 now its a 30 . I want to be around and watch my child grow and enjoy him. Sorry for all these comments but i dont know what else to do right now. Ive been telling everyone i know to get checked. Nobody knows our bodies like we do. If something isnt right stand your ground until you get answers
melanieOctober 11, 2012 at 1:31 am
Oh wow! Just reading these comments makes me feel i dont know, but not crazy i guess? More and more of these symptoms that i have are related to this IUD! I was a ring size 7 before i got pregnant. My baby is almost 10 months old and i wear a size 9 now. My ankles and joint ache if i stand or sit too long. My feet have jumped up 1 size and have gotten wider...feet can get larger i know that but a [Show More]Oh wow! Just reading these comments makes me feel i dont know, but not crazy i guess? More and more of these symptoms that i have are related to this IUD! I was a ring size 7 before i got pregnant. My baby is almost 10 months old and i wear a size 9 now. My ankles and joint ache if i stand or sit too long. My feet have jumped up 1 size and have gotten wider...feet can get larger i know that but a full shoe size..AND width? I am so scared about this surgery next week! I have mitral valve prolapse w regurgitation and my mom was a little but older than me she almost died from endocarditis which is an infection in the inner lining of the heart caused by...dental procedures, piercings, surgical procedures etc anything that can break the skin causes this. This company tricked us into buying their product. The doctors and manufactorers sold us on this product but neglected to warn us about these side effects. Really? What do they say? What are the statistics? Less than 1% or something. Well i dont think that is an accurate account
melanieOctober 10, 2012 at 9:37 pm
Hi, i had my mirena placed march 1,2012 ...3.5 months after i had my son. Since then and the only way to describe it is like i had mono again. Constant fatigue, moodiness, weight gain, no libido, painful intercourse, , weird discharge, migraines just about every day, this past monday october 8, 2012 i started having unbearable pelvic pain, pressure on my bladder, my husband touched me on my left s[Show More]Hi, i had my mirena placed march 1,2012 ...3.5 months after i had my son. Since then and the only way to describe it is like i had mono again. Constant fatigue, moodiness, weight gain, no libido, painful intercourse, , weird discharge, migraines just about every day, this past monday october 8, 2012 i started having unbearable pelvic pain, pressure on my bladder, my husband touched me on my left side of my stomach and i screamed....so the next day i call my dr and it took them 3 hours to call me back. The nurse on the phone tried telling me that i need to go to a lab and do a urinalysis because it sounded like i had a UTI. We almost argued on the phone until she ok'd my demands for an ultrasound. So i show up in my nursing scrubs and they immediatly treated me different. So she performed a very painful transvaginal ultrasound and low and behold it revealed an ovarian cyst (from the mirena) AND it migrated down into my cervix, the strings are totally wrapped around it, and its sideways. She was unable to get it out so next thursday i have to have a hysterscopy and a laparoscopy. I am just praying that they do not have to cut me open and i wont have permanent damage. I also hope i dont lose my new job that i started a month ago
ShanteOctober 9, 2012 at 12:39 am
I am a 34 years old. I had the IUD put in back in 2005 after having my second child. I choose to have it for 5 years. I started having abn pap smears seems like ever since then and have had a few biopsy and I had to surgery on my cervix which my Doctor had to cut a piece because there were "pre cancerous cells." Currently I'm still having abn pap smears coming back and so now my doctors is say[Show More]I am a 34 years old. I had the IUD put in back in 2005 after having my second child. I choose to have it for 5 years. I started having abn pap smears seems like ever since then and have had a few biopsy and I had to surgery on my cervix which my Doctor had to cut a piece because there were "pre cancerous cells." Currently I'm still having abn pap smears coming back and so now my doctors is saying that there is a great possibililty that I WILL have to have a partial hysterectomy in my near future.
kenOctober 8, 2012 at 8:54 pm
yes i am a man and this iud stuff is painful to me when i have intiment relations with my significant other it causes pain in the head of my penis after sex and the pain stays for two or more days depending on the duration of sex time so if it hurts me i can only imagine what it does to the women!!!!!!
tara-raeOctober 8, 2012 at 8:47 pm
i have had the iud in since a few months after the birth of my youngest child in 2009 i did not realize that the mood swings and the angry outburts i started to have after the iud was put in were because of the iud but now that i look back i did not have many of them before it was put in. i also have been very tired and weak since then. i have not had a regular menstrual cycle at all i went the fi[Show More]i have had the iud in since a few months after the birth of my youngest child in 2009 i did not realize that the mood swings and the angry outburts i started to have after the iud was put in were because of the iud but now that i look back i did not have many of them before it was put in. i also have been very tired and weak since then. i have not had a regular menstrual cycle at all i went the first two years with nothing at all then within the last year i have spotted off and on and a few times i have gone an entire month with my period. I have had a big outbreak of blemishes in the last two years and they dont go away no matter what i do. i have abdominal pain and they told me they did not know what was wrong with me. I am going to go get the iud took out and hope that my problems go away.
jasmineOctober 7, 2012 at 1:22 pm
i had the mirena removed march 2012 . i had it in for 3-4 years. sice removed.i have belly button pain. breast pain my periods have been abnormal. 2-3 days. i bleed dark clots. i wipe and it seems like im bleeding internally 2days after my period. imy body doesnt feel right. and im worried.. i had a breast exam just to be sure. my breast are very heavy and sore daily.and imalways tired. i have be[Show More]i had the mirena removed march 2012 . i had it in for 3-4 years. sice removed.i have belly button pain. breast pain my periods have been abnormal. 2-3 days. i bleed dark clots. i wipe and it seems like im bleeding internally 2days after my period. imy body doesnt feel right. and im worried.. i had a breast exam just to be sure. my breast are very heavy and sore daily.and imalways tired. i have been trying to get pregnant. i have taken multiple pregnancy tests. i feel something is wrong. im nervous
HeatherOctober 7, 2012 at 2:31 am
I had the mirena put in in October 2011 after my son was born. I had no trouble with it at first. No cramps or side effects. I thought it was perfect. I have always been relatively healthy. Then out of no where in July 2012 I started suffering from tremors, severe joint pain, back pain, swelling in hands and feet, weakness in my legs and other muscles. I went to the doctor to find out that I have [Show More]I had the mirena put in in October 2011 after my son was born. I had no trouble with it at first. No cramps or side effects. I thought it was perfect. I have always been relatively healthy. Then out of no where in July 2012 I started suffering from tremors, severe joint pain, back pain, swelling in hands and feet, weakness in my legs and other muscles. I went to the doctor to find out that I have hyperthyroism (graves disease). I have done some research and found tons of other women complaining of developing hyperthyroidism from the mirena. I am convince the mirena caused my disease too. I had it removed shortly after but the disease is permanent and something I will have to live with the rest of my life. I know have to visit the doctove ever 3 weeks to check my blood and have to pay close to 100 dollars a month on medication to keep my symptoms under control. I never thought bith control could do this to me.
ashleyOctober 6, 2012 at 3:20 am
i had the mirena inserted after my 2nd child was born in may 2011. both my children were c sections so my cervix had never even been dilated. when it was inserted it was the most painful thing i had ever felt. for 2 weeks i was in excruciating pain. cramps, bleeding. all that. i bled for like 2 months straight. the doctor said it was normal. Then i quit bleeding. its been over a year and some odd[Show More]i had the mirena inserted after my 2nd child was born in may 2011. both my children were c sections so my cervix had never even been dilated. when it was inserted it was the most painful thing i had ever felt. for 2 weeks i was in excruciating pain. cramps, bleeding. all that. i bled for like 2 months straight. the doctor said it was normal. Then i quit bleeding. its been over a year and some odd months and i havent had a period. im only 22 doc said that was normal too. then i noticed the depression, crying feeling worthless. cant lay on my left side whatsoever or it seriously hurts me down there. like something is piercing my insides. tried to go to the health dept to have it removed and with me not having any insurance or proof of income they said it would've costed me 400 dollars to even see the doctor. i have gained over 50 pounds with no change in diet im the biggest i've ever been.i just wanna know if anyone else has had this problem.
NicoleOctober 5, 2012 at 12:46 pm
I had the Mirena from Jan 2010 until today! My health had always been great, I have 3 beautiful kids.. In the first few months of having it I got awful dizzy spells to the point I was afraid to drive, I would get such an ill feeling from it that I actually was sick quite a few times.. The Doc said I had VERTIGO. Then from there my health has spiralled Ive had thinning hair, numbness and tingling[Show More]I had the Mirena from Jan 2010 until today! My health had always been great, I have 3 beautiful kids.. In the first few months of having it I got awful dizzy spells to the point I was afraid to drive, I would get such an ill feeling from it that I actually was sick quite a few times.. The Doc said I had VERTIGO. Then from there my health has spiralled Ive had thinning hair, numbness and tingling in my hands and feet, feel so tired and exhausted all the time, have been severely depressed even to the point of suicide thoughts at times, have fluid retention, have ear aches (but when I go to the docs they say inside my ears look fine), have had this lump like feeling in my throat that keeps me awake at night its that bad.. Ive been to the docs numerous times and was told it was anxiety.. NOW I HAVE DISCOVERED I HAVE MASSIVE NODULES ON MY THYROID AND ARE WAITING ON BIOPSY RESULTS AS IM TOLD THEY ARE CALCIFIED AND COULD BE CANCER.. Ive NEVER had a thyroid issue.. Not to mention the breast pain it was like lactating all over again, ohh did I mention the lumps in my breasts also (ultrasound confirmed I now have FIBRE-CYSTIC BREATS DISEASE.. Mood swings, a copper like taste in my mouth constantly, constant colds and Infections, chest pain, difficulty breathing and swallowing , constant headaches, blurred vision and the list goes on. I was told the Mirena was completely safe and the ideal birth control.. I couldn't wait to get it removed after reading up on other women experiencing similar side effects, now Im going to make an effort to warn every woman I know about the side effects Ive had over the past 2 and a half years in the hope that No-one has to endure the health complications Im having... Im DISGUSTED THAT THIS PRODUCT HAS BEEN SO DEVASTATING TO SO MANY WOMEN. I hope the side effects Ive had will disappear now that the Mirena is out so I can get on with being the Mum my kids deserve instead of a crazed woman thwarted with illness!
susanOctober 5, 2012 at 4:32 am
I had the mirena iud inserted in 2007 since then I have had sevre abdominal pain, cysts after cysts lanced, in pelvic area to my face. I never experienced the discomfort during sex till iud ,my husband could feel it poking him during sex, migranes, mood swings,uncontrolable crying ,feeling angry for no reason, constant bleeding to spotting, no kind of regulation to a menstarl cycle. I think it wa[Show More]I had the mirena iud inserted in 2007 since then I have had sevre abdominal pain, cysts after cysts lanced, in pelvic area to my face. I never experienced the discomfort during sex till iud ,my husband could feel it poking him during sex, migranes, mood swings,uncontrolable crying ,feeling angry for no reason, constant bleeding to spotting, no kind of regulation to a menstarl cycle. I think it was very unfair for these doctors to tell us married women this is the best solution of birth control so we can have more children one day and have to suffer and deal with all the pain.
shawnnagcOctober 5, 2012 at 4:30 am
i had the mirena in for four years now and im 20 years old i havent had all of these problems but dizzyness,abdominal pain,migraines and increase of sex drive i hope it aint bcuz of this but i seems like it would be more serious symptoms i hope nothing else comes up withh me n this thsnks for posting the comments
ChristinaOctober 4, 2012 at 1:04 am
This coming march I will have had my mirena for five years. I have had no discomfort or other issues. I have gained some weight and have noticed a increase in blemishes. I go to my regularly scheduled appointment and they have not found any problems. This is the most convenient form of birth control for me. I was also told of the risks by my doctor. I would recommend this product based on my exper[Show More]This coming march I will have had my mirena for five years. I have had no discomfort or other issues. I have gained some weight and have noticed a increase in blemishes. I go to my regularly scheduled appointment and they have not found any problems. This is the most convenient form of birth control for me. I was also told of the risks by my doctor. I would recommend this product based on my experience.
SarahOctober 3, 2012 at 12:04 am
In the Spring of 2011, I had the Mirena IUD inserted. During the insertion I suffered extreme pain and for weeks after. The pain did gradually go away but the side effects never disappeared. While I had the mirena, I suffered severe depression, fatigue, heart palpulations, dropped/increased blood pressure, severe mood swings, night sweats, suicidal thoughts, insomnia, blurred vision, stutter on so[Show More]In the Spring of 2011, I had the Mirena IUD inserted. During the insertion I suffered extreme pain and for weeks after. The pain did gradually go away but the side effects never disappeared. While I had the mirena, I suffered severe depression, fatigue, heart palpulations, dropped/increased blood pressure, severe mood swings, night sweats, suicidal thoughts, insomnia, blurred vision, stutter on some ocassions, loss of focus, decrease in mental concentration (forgetfulness), muscle weakness, sexual decrease, blood in urine, anxiety, severe migraines, miscarriages, breast tenderness, crying episodes for no reason, weight gain, cyst like acne, painful episodes in my lower back and unbelievable sharp pains in my pelvic area. I also at one point, felt a popping sensation when I walked while having Mirena. I had the IUD removed a few months later (no more than a year) due to the severe side effects I was suffering from. I almost lost my fiance and my son due to the violent mood swings I would burst into randomly. I thought shortly after having the IUD removed, that these side effects would shortly disappear; instead, they've grown so rapidly over the next few months, I have a had time functioning in public situations which affects my career in the medical field and my relationship with my family. My own son and fiance wouldnt come around me because I would burst into this violent mood! No side effect has yet to go away...I was a healthy individual before Mirena and I feel like Ive lost my life within a blink of an eye. I have felt crazy since and I would give anything to get my old self back for my family and myself.
ChastityOctober 2, 2012 at 4:06 pm
I had the Mirena in 2011 and I started having a cycle every other week. I also started having really bad pelvic pain so I went to get it checked out. The OBGYN would just keep telling me the cycles would stop but they lasted for almost a year that way. One night I had bad stomach pain and went to the ER where it was discovered I had fluid in my fallopian tubes and fibroid cyst and I didn't have an[Show More]I had the Mirena in 2011 and I started having a cycle every other week. I also started having really bad pelvic pain so I went to get it checked out. The OBGYN would just keep telling me the cycles would stop but they lasted for almost a year that way. One night I had bad stomach pain and went to the ER where it was discovered I had fluid in my fallopian tubes and fibroid cyst and I didn't have any of that before I got the mirena inserted. I finally got my doctor to remove the mirena but now due to water being in my tubes I can't get pregnant and have to have surgy to have it removed. Please read all the side effects before you get the mirena I wish I had.
cristinaOctober 2, 2012 at 2:17 am
hey everyone.. i keep reading all these negative comments about the IUD.. i had mine in since 2010.. i ewas 21 and hadu my onecharacter and onlyu daughter at 19.it was painful ti get in touch but overall i have no complaints.. i do get sone cramping andonce i do a while spoting.. but nothing seriouse but i am going to make sure its still therr. Reading these comments are getting my paranoid :)i h[Show More]hey everyone.. i keep reading all these negative comments about the IUD.. i had mine in since 2010.. i ewas 21 and hadu my onecharacter and onlyu daughter at 19.it was painful ti get in touch but overall i have no complaints.. i do get sone cramping andonce i do a while spoting.. but nothing seriouse but i am going to make sure its still therr. Reading these comments are getting my paranoid :)i hope everyone is doing ok. God bless.
BrittneyOctober 2, 2012 at 12:08 am
I had the mirena inserted during my 6 week postpartum appt after having my son, this was Nov 08. I had no period, slight weight gain but no cramping significantly, no bleeding, nothing out of the ordinary. Until March 12, I had severe cramping, severe bleeding after intercourse that lasted for days, I could barely sit down. So May 12, I decided to go to my local health department to get it removed[Show More]I had the mirena inserted during my 6 week postpartum appt after having my son, this was Nov 08. I had no period, slight weight gain but no cramping significantly, no bleeding, nothing out of the ordinary. Until March 12, I had severe cramping, severe bleeding after intercourse that lasted for days, I could barely sit down. So May 12, I decided to go to my local health department to get it removed, I didn't know what was wrong but my body kept telling me something was wrong and I needed it out asap. The doctor tried to talk me into keeping it, and I said no, its too much, its done somehting to me, when she went in, she came up and said this will be harder than I expected, It had went up and over to lodge into my uterine wall. It took her 15 minutes to get it out, she said she almost stopped and told me to go to the hospital to get my service dilated and have surgery to get it out. Thankfully, that was not the case. After, having it out, everything went back to normal until I missed a period in June, took a pregnancy test and it was positive, only 68 days after removing the mirena. That ended in miscarriage (A blighted Ovum) at 6 weeks. I'm now pregnant again, I'm 6 weeks now. Everything so far is ok, but I am not getting my hopes up this time. I didn't have a period between the miscarriage and a new pregnancy.
RochelleOctober 1, 2012 at 7:51 pm
Thank You to everyone. I have read all of the posts. I have been going through majority of these symptoms as well. NEVER thought they could be from Mirena. In fact I was told I had a thyroid problem, even when the tests were normal. Test after test. Biopsies, blood work, nuclear med,etc. Goes to show you really can't trust DR's. I didn't even know my symptoms were side affects. I had my first I[Show More]Thank You to everyone. I have read all of the posts. I have been going through majority of these symptoms as well. NEVER thought they could be from Mirena. In fact I was told I had a thyroid problem, even when the tests were normal. Test after test. Biopsies, blood work, nuclear med,etc. Goes to show you really can't trust DR's. I didn't even know my symptoms were side affects. I had my first IUD in 2004 and a second in 2009. All these years with major mood /anger issues, extreme fatigue/back pain, NO sex drive, and the list goes on... I hope I / WE can feel normal again.
ednaSeptember 30, 2012 at 5:48 pm
Stay away from marina it will kill u I got that stupid marina about 1year ago and it has been like hell on earth my life has change completely from no pain to severe back pain and lower abdominal pain! I enjoy sex no more because I don't feel like it and when I do is bleed a lot! I have very low enjoy I use to work out for three hours aday before the stupid marina was insert into my uterus but now[Show More]Stay away from marina it will kill u I got that stupid marina about 1year ago and it has been like hell on earth my life has change completely from no pain to severe back pain and lower abdominal pain! I enjoy sex no more because I don't feel like it and when I do is bleed a lot! I have very low enjoy I use to work out for three hours aday before the stupid marina was insert into my uterus but now I can't even walk a mile without feeling that I'm gonna pass out due to extreme dizziness and fatigue! I has a cycle every 14 day and bleed three_four times a week! Tell me is this a life I would not dear wish that on any woman! I'm depress I have put un twenty pounds in the past year constantly bloated! I can lay on my stomach backaches my back hurt like hell I have pass out three times from severe cramping in my lower abnormal I'm just mess up by all of these sides effect that no one cared to mentioned they said I would go back to my life in three months! But its just a bunch of BULL!!! Its been one year still no change I'm gonna have this shit remove from me ASAP!
ednaSeptember 30, 2012 at 3:15 pm
OMG! That marina is the worst thing that a woman could have insert in her uterus! Ladies if I tell u that I'm suffering the consequences of not knowing u can believe me!!! I got the marina put in me 1year ago and for me my children, my husband its been like hell on earth! I have been. Having Constant lower back pain and sever stomach pain. I can't have no sex because I bleed a lot after sex! I'm [Show More]OMG! That marina is the worst thing that a woman could have insert in her uterus! Ladies if I tell u that I'm suffering the consequences of not knowing u can believe me!!! I got the marina put in me 1year ago and for me my children, my husband its been like hell on earth! I have been. Having Constant lower back pain and sever stomach pain. I can't have no sex because I bleed a lot after sex! I'm always tired and have mood swings! Before the stupid marina I had a good life I use to workout three hour a day, now I can't even walk a mile without feeling like am about to pass out ! I'm a singer but I don't even enjoy sinning anymore ! My 9year daughter wants to do exciting things and so do I ,but ever since I got that marina I can't enjoy anything I'm in bed most of the time I'm also afraid of loosing my husband to this shit!!! Before I got the marina I was told by the nurse and the doctors that my life would go back to Normal in 60days its been 1year and no change! I'm so tired of being lied to! I have a cycle every 14days with sever bleeding and sever cramping must of the time I passed out , there has been incidence where I was coming on my cycle and the pain was so extreme I thought I was about to have a baby that how horrible the pain is whenever I come on my cycle oh my GOD! I would wish this life on anyone! I'm constantly having infections upon infection I have one as we speak because I still have that marina due to lack of medical insurance! But I prom p promise u ladies I'm gonna find some way to have this shit remove from my life and inside of me! Please for those of u who has not have the marina my advise to u is RUN , RUN, RUN for ur life! My heart goes out to all of u who has been affected by this horrible birth control! I pray that u will be able to get ur lives together soon! Peace be wit u all!!!
JMSeptember 29, 2012 at 8:00 am
Seeing all there post makes me feel so lucky i haven't had any issues with my iud. I've had it for 3 years now and it hasn't affected anything but seeing all these post makes me wonder. Im definitely going to go get checked out though.
AlissaSeptember 29, 2012 at 4:28 am
I got the Mirena when I was 17, February 1st 2012, I havent had kids. The doctors said it was now acceptable for younger kids and made it seem like the best choice, for the first two months I was in agonizing pain, it was so bad I would litterally drop to the floor crying in a ball. I have the back pain and also a pelvic pain which seems to stem into my legs, sometimes I can't move. I have to take[Show More]I got the Mirena when I was 17, February 1st 2012, I havent had kids. The doctors said it was now acceptable for younger kids and made it seem like the best choice, for the first two months I was in agonizing pain, it was so bad I would litterally drop to the floor crying in a ball. I have the back pain and also a pelvic pain which seems to stem into my legs, sometimes I can't move. I have to take RX pills just to ease the pain that comes daily at seemingly the same time. I feel the depression as well and have expirienced random outburts of tears for no reason. Also I have consistant bleeding for about 2 weeks out of the month, sometimes its spotting and sometimes its full on heavy flow. I expressed some concern to my doctor but he said it wasn't from my birth control. I have high blood pressure and a "hardened ovary" says a different doctor. It hurts during intercourse and my partner says it hurts him too, it feels like he is being prodded or poked. I decided to do some research and that led me to this page. I have had the Mirena for almost 8 months and it has been the worst 8 months ever, I'd almost rather be pregnant. And I wasn't sure if it was the birth control but I did expirience an increase in weight and acne. Frankly, I am a little scared at what I've seen on this page.
llouSeptember 27, 2012 at 10:23 pm
i have just been told by the doctor dont worry its not cancer but we need to know what coil you have fitted i couldnt remember also asked me did i have any symtons as in what was my immediate thought and yeah i do i bleed every fourteen to twenty one days i have put on weight in my middle i feel like im four months pregnant did have hair loss stopped washig it and brushing it bit better now back a[Show More]i have just been told by the doctor dont worry its not cancer but we need to know what coil you have fitted i couldnt remember also asked me did i have any symtons as in what was my immediate thought and yeah i do i bleed every fourteen to twenty one days i have put on weight in my middle i feel like im four months pregnant did have hair loss stopped washig it and brushing it bit better now back aches numbness in my left hand cant grip things some days dizziness dont know if its causing panic attacks as i suffer with them but the worst thing for me is the feeling of being constantly bloated like being pregnant but im not as i have even done pregnancy tests im fourty four had a new coil in 2010 having had one since my first daughter had it many years bleeding got unbearable so had it removed had a month of no coil felt good my body felt better like it was mine had another coil fitted didnt really think to much of if was glad to have contraception but now i think i would gladly go without sex then feel like this i only came on this website as i wanted to know more about what the doctor had told me and ended up reading all these very simlar feelings sensations i will be getting this coil removed asap
AshleySeptember 27, 2012 at 8:01 pm
I got the Mirena back in September of 2007, about 4-5 weeks after the birth of my daughter. About 3 weeks later, I decided to have it removed because I was expereincing excruitaing headaches, my OB/GYN could not find it, so she sent me to get an X-ray and it was found in my left abdominal wall, had a laprascopy done, and it was removed. Now hear I am at age 24, diagnosed with unexplained infertili[Show More]I got the Mirena back in September of 2007, about 4-5 weeks after the birth of my daughter. About 3 weeks later, I decided to have it removed because I was expereincing excruitaing headaches, my OB/GYN could not find it, so she sent me to get an X-ray and it was found in my left abdominal wall, had a laprascopy done, and it was removed. Now hear I am at age 24, diagnosed with unexplained infertility and IBS, my doctor did not inform that you could suffer infertility after Mirena, We have tried for 3 years straight, I've even taken Clomid and has an HSG done, I am beyond devastated. I didnt ask for this to happen. I wasnt fairly warned about all the side effects. I think that the IBS was intiated because of the location of the IUD being so close to my colon and large intestine, it just isn't fair at all. No one should suffer not being able to have children
candiceSeptember 27, 2012 at 1:37 pm
I got my mirena put in 2009 after having my daughter.I was told it was not a painful procedure.Which was a lie!Mirena stoped my period all together.Until about 2 months ago.I bleed 3-4 day moderately twice a month.Which scares me since I haven't bleed since june 2009 and its now september 2012!But every since I've had mirena in place i have moderate abdominal and pelvic pain.Also I've been severe [Show More]I got my mirena put in 2009 after having my daughter.I was told it was not a painful procedure.Which was a lie!Mirena stoped my period all together.Until about 2 months ago.I bleed 3-4 day moderately twice a month.Which scares me since I haven't bleed since june 2009 and its now september 2012!But every since I've had mirena in place i have moderate abdominal and pelvic pain.Also I've been severe lower back pains, to the point that I can't bend or lay on my back.I would not recommend this to anyone!!If you have it get it removed there are numerous of side effects that your not told about.I suffer from chronic pain shooting down both my legs, feeling numbness.I wake up fatigue every morning since I've had the mirena in place.I've became very depressed I can't enjoy family events like i use too,I have these frequent mood swings.It's something I will NEVER get again in my life,once I have it removed!
AlexisSeptember 27, 2012 at 12:37 am
I had mine for 3 years when I first had issues. Like everyone else, pain, discomfort, facial hair, acne, moody, depressed. I also had inflammation. Eventually my cylces stopped and I got worreid. I went to a different doctor who tried to remove it, but couldn't. I had to have surgery in 2009 to remove it, and a D and C. which was painful. I still have pain because of it. a month later I had lumps [Show More]I had mine for 3 years when I first had issues. Like everyone else, pain, discomfort, facial hair, acne, moody, depressed. I also had inflammation. Eventually my cylces stopped and I got worreid. I went to a different doctor who tried to remove it, but couldn't. I had to have surgery in 2009 to remove it, and a D and C. which was painful. I still have pain because of it. a month later I had lumps show up on my neck. 7 months later, while pregnant, I was diagnosed with Hodgkin's Lymphoma. I was just wondering. If anyone else was diagnosed with a caner while using or just after the removal of there IUD?
AprilSeptember 26, 2012 at 7:50 pm
I am 34 yo and started out on the depo shot after having my 3rd child in October of 2007. After about 6 months of the shots My dr and I decided to change from the shot to the pills due to some complcations from the shot, well in 2010 I started to have increased bllod pressure and major weight gain so my obgyn recommended that I have the Mirena inserted. Well I agreed in September of 2010. I had[Show More]I am 34 yo and started out on the depo shot after having my 3rd child in October of 2007. After about 6 months of the shots My dr and I decided to change from the shot to the pills due to some complcations from the shot, well in 2010 I started to have increased bllod pressure and major weight gain so my obgyn recommended that I have the Mirena inserted. Well I agreed in September of 2010. I had severe abdominal cramps for about the first week and my cycle lasted as normal for about the first couple of months. My cycle then stopped, but I continue to have severe cramps ( they feel like they should be labor pains or something due to severeity of them). I then noticed that my sex drive started falling off, and my headaches has increased. I now am having periods of moodiness, fatigue, restlessness, symptoms of depression, and am still having severe cramps about 1-2 times throughout the month,SEVERE lower back pain( to the point it hurts for me to stand up and wash dishes, and do things with my 3 wonderful children who are very active), acne on the face, vaginal odor, periods of vaginal dryness, and periods of over moisture in the same area, NO CYCLES, spotting after sex, headaches 4-5 times weekly, new onset is breast tenderness. I am going to find me a new obgyn beings my dr that inserted it is now no longer in the area. I hope that I don't continue to have the side effects from this horried thing because I want to be able to enjoy my children while they are still young!
HarmonySeptember 26, 2012 at 4:53 am
Never will I recommend this IUD to anyone. Had mine in for 3 years had a lot of the same problems as everyone else. But two weeks ago I found out I was pregnant they removed it on the spot and than the worst thing that could ever happen to a mother happened. I miscarried I am devastated! Planed or not planed I love all my children. I am now a grieving mother!!! RIP sweet angel I'm sorry I never[Show More]Never will I recommend this IUD to anyone. Had mine in for 3 years had a lot of the same problems as everyone else. But two weeks ago I found out I was pregnant they removed it on the spot and than the worst thing that could ever happen to a mother happened. I miscarried I am devastated! Planed or not planed I love all my children. I am now a grieving mother!!! RIP sweet angel I'm sorry I never got to meet you !!!!!!!
lynnSeptember 25, 2012 at 11:09 pm
I got the mirena in 2009 after having my twins I have not had a period since then I have not really had any problems with it except for these last weeks I have been crapping a lot lately with pelvic pains and lower back pains also in the last week my bf has been complaining of being able to feel it and acually caused sores on the tip of his private area what should u do I am scared
ShannonSeptember 25, 2012 at 4:10 am
I had my son Jan 25,2008 and got the mirena either March or April 2008.In Sept of 2010 I got it out we wanted to start trying for another child.In March of 2011 found out I was pregant the next day I missed carried and it was a bad miscarrage my body filled up with blood and I had to have surgery.Then in July of 2011 got pregant and in Aug of 2011 nothing formed in the sac so I had to take meds to[Show More]I had my son Jan 25,2008 and got the mirena either March or April 2008.In Sept of 2010 I got it out we wanted to start trying for another child.In March of 2011 found out I was pregant the next day I missed carried and it was a bad miscarrage my body filled up with blood and I had to have surgery.Then in July of 2011 got pregant and in Aug of 2011 nothing formed in the sac so I had to take meds to micarry again and now I havent been able to get preganet.It makes me wounder if this caused by the mirena.Im going to a different docter oct 3 to find out if theres something wrong.
SaraSeptember 23, 2012 at 4:15 am
So I got mirena June of 2009, had my son in April of 2009, I was able to feel the threads in July of 2009, but have not been able to feel them since then at all. I didn't know if this was normal or not, I know mirena is still there because of x-rays i have seen and being asked what it is. I have not had a period since my I got pregnant with my son at all. that was in June 2008 which was my last pe[Show More]So I got mirena June of 2009, had my son in April of 2009, I was able to feel the threads in July of 2009, but have not been able to feel them since then at all. I didn't know if this was normal or not, I know mirena is still there because of x-rays i have seen and being asked what it is. I have not had a period since my I got pregnant with my son at all. that was in June 2008 which was my last period. Is this normal not normal. i don't have insurance and haven't had any insurance since having my son in 2009. I'm concerned after hearing and reading all the comments and all the issues that have happened, that I need to get this thing out. I don't want nothing to happen to me as I'm a single parent. Is their any advice that anybody can give me at all. Im really scared and need some advice, Im reaching anybody out anybody who can give me any advice at all please.
BrandySeptember 21, 2012 at 9:54 pm
I had the Mirena IUD inserted about 8 wks after the birth of my daughter in 2008. The process was VERY uncomfortable. I didn't even make it to the follow up appt to make sure it was still in place. I was in so much pain within a week and a half of insertion that I went back to my doc. They checked to see if it was in place but couldn't find it. They decided to do an ultrasound but still couldn't f[Show More]I had the Mirena IUD inserted about 8 wks after the birth of my daughter in 2008. The process was VERY uncomfortable. I didn't even make it to the follow up appt to make sure it was still in place. I was in so much pain within a week and a half of insertion that I went back to my doc. They checked to see if it was in place but couldn't find it. They decided to do an ultrasound but still couldn't find it. They finally had to do an x-ray of my abdomen. It turns out that it traveled and decided to wrap itself around my appendix. I had to have laparoscopic surgery to have it removed. Needless to say, I am not a fan! If anyone is thinking of getting it, please don't! It's really not worth the risks both from it possibly traveling as well as side effects from the drugs in it..
TeresaSeptember 18, 2012 at 11:43 pm
she is handicapped and can't deal with a mensral cycle she can not stand up to get a pad on her. she has many things that is wrong with her like seizures a bag that she has to pee threw she has soliosis and cerebral palsy her immune system is shot. what more can i say.
teresaSeptember 18, 2012 at 11:30 pm
my daughter has this and i really need to know if a uti is causes by it. she is contantly in pain. been to the doctor 3 times in a 1/2 month with this and they keep giving her antidoctes which are not working. if anything bad happens to her does she have grounds to sue for giving her false imformation.
AnaSeptember 18, 2012 at 8:07 pm
Oh, and just to update from yesterday's post, I had an HSG last week to see what was going on with me for fertility, and they found scarring and inflammation. Only 2 options on what went wrong here. And one of them happens to be IUD. I've only had one type of IUD. Mirena.
AnaSeptember 17, 2012 at 5:42 pm
I had Mirena inserted in August 2003. It wasn't painful to me to have it inserted, but my PMS for the next 3 months was completely off the wall. My periods came about every 2-3 weeks if it felt like it and I wanted to just chop my partners head off all the time. After those first 3 months or so, my periods stopped. I hadn't realized that i started to have an increase in infections until way later [Show More]I had Mirena inserted in August 2003. It wasn't painful to me to have it inserted, but my PMS for the next 3 months was completely off the wall. My periods came about every 2-3 weeks if it felt like it and I wanted to just chop my partners head off all the time. After those first 3 months or so, my periods stopped. I hadn't realized that i started to have an increase in infections until way later down the line. In 2008, I told my dr (i had a different dr by then) at my annual that its time to have it removed and have another put in since it was working fine and I would like to continue it. She said that it's working just fine as is and there was no need to remove it then. In 2009, i was in a divorce and starting a new relationship and I was still nervous about having the same IUD in, so I had it removed by another dr. Again, No issues on removal. Small pain but fine after that. Soon after my fiance and I decided we would like to try having a baby. But my periods were not on a schedule yet. That was October 2010 when we decided to start trying. Here we are September 2012 and still nothing. What has mirena done to me. Oh and I already had 2 children before mirena. And was very fertile before. So why nothing now?
LaurenSeptember 16, 2012 at 8:47 pm
To any who have experienced bad side effects of mirena, I now know your pain. I am a 22 year old female, got the mirena inserted may 15th 2012.about 6 weeks after my first child. The first month the side effects were non apparent, then it hit me like a ton of bricks. Migraines, more like torture the headache that never ended it lasted for 2 months straight relentless pain, ibuprofen,Advil, nothing[Show More]To any who have experienced bad side effects of mirena, I now know your pain. I am a 22 year old female, got the mirena inserted may 15th 2012.about 6 weeks after my first child. The first month the side effects were non apparent, then it hit me like a ton of bricks. Migraines, more like torture the headache that never ended it lasted for 2 months straight relentless pain, ibuprofen,Advil, nothing helped!! I went to 25 doctors from a neurologist to sitting in the er at two in the morning. Literally thought I was dying. Until I looked up the possible side effects it listed headaches, acne, severe weight gain, fatigue, low sex drive, and ovarian cysts. I decided to get it removed no questions asked. It's been a week and a half since its been gone and the headaches are gone for the most part, I am grateful to have found the culprit but want to warn others you could be that 5 percent that suffers
KellySeptember 12, 2012 at 4:51 am
I had two Mirena IUDs. 3 years after the second one was put in (8 years total on the IUD), I had it removed so my fiancé and I could try to conceive. 8 months later- no pregnancy, but I am experiencing such horrible anger/pms for two weeks before my period. I tried exercising, eating healthy, counseling, blood tests, all ineffective at helping me feel better or find out why I am feeling this way.[Show More]I had two Mirena IUDs. 3 years after the second one was put in (8 years total on the IUD), I had it removed so my fiancé and I could try to conceive. 8 months later- no pregnancy, but I am experiencing such horrible anger/pms for two weeks before my period. I tried exercising, eating healthy, counseling, blood tests, all ineffective at helping me feel better or find out why I am feeling this way. I finally made an appointment with a psychiatrist whom I saw today. She stated that coming off the IUD was doing to me what a hysterectomy does to some women. It has been getting worse each month- not better with time. She prescribed me a mood stabilizer, and it freaks me out that this is what it has come to. Am I going into early menopause? Can my fiancé and I not have children of our own now? So many questions still. :(
CherylAugust 28, 2012 at 1:32 pm
Hi, I've had my iud in since October of 2011 and haven't really had any issues with it except some pain occasionally during sex and constant bleeding for the first month or two but it was light, I've not had a period since December which is amazing but reading these stories has made me nervous, I've had bad hair loss and mood swings and depression along with some other things but I just figured it[Show More]Hi, I've had my iud in since October of 2011 and haven't really had any issues with it except some pain occasionally during sex and constant bleeding for the first month or two but it was light, I've not had a period since December which is amazing but reading these stories has made me nervous, I've had bad hair loss and mood swings and depression along with some other things but I just figured it was hormones, now I'm beginning to wonder, I've not had it check since it was inserted and haven't heard a thing from my doctor about it. Looks like I might be making a phone all to have it checked and possibly removed...thank you all so much for sharing your story with everyone
DeniseAugust 27, 2012 at 10:16 pm
I had an IUD put in 05' or 06' can't remember really. Having it put in did not really hurt me to bad, but about 5 days later I had the worst pain in my entire life in my pelvic regions. It passed that same day and didn't really think anything else about it. I went for my string check and my doctor said he did not feel it. He then did an ultrasound of my uterus and it was not there. Said it must ha[Show More]I had an IUD put in 05' or 06' can't remember really. Having it put in did not really hurt me to bad, but about 5 days later I had the worst pain in my entire life in my pelvic regions. It passed that same day and didn't really think anything else about it. I went for my string check and my doctor said he did not feel it. He then did an ultrasound of my uterus and it was not there. Said it must have came out. Me thinking it came out the day I was in so much pain. Never thought twice about it and went on with my life. Well just recently I went to the emergency room with severe abdominal pain and vomitting. The ER physician thought kidney stones, took me for an MRI. And guess what they found...... the IUD embedded in my abdominal wall along with a pelvic hernia. I am beyond furious. After doing some research on this it explains my chronic headaches I have been having the past few years along with stomach pain, possibly my chronic diahrea and back pain. I need help finding a lawyer in my area or even period that will take my chase especially with this being a 5 yr birth control it should have been taken out in 2010 or 2011 had it stayed where it was supposed to. My doctor never explained the risk of movement or any side effects for that matter. Please Help me.
JenniferAugust 27, 2012 at 7:42 pm
I had the Mirena put in after my son was born. While it was in I started to have ovarian cysts and migraines. I was nauseated everyday and felt pregant. I never felt like this until the iud was in.Every time I asked to have it removed the doctors would not do it. A year later I am out of the military and experience severe cramping and the iud fell out. My husband and I have been trying to conceiv[Show More]I had the Mirena put in after my son was born. While it was in I started to have ovarian cysts and migraines. I was nauseated everyday and felt pregant. I never felt like this until the iud was in.Every time I asked to have it removed the doctors would not do it. A year later I am out of the military and experience severe cramping and the iud fell out. My husband and I have been trying to conceive for 2 months with no luck.
DawnAugust 22, 2012 at 9:32 pm
I had the mirena put in Feb 2008 and removed sept 2010. When removed they had trouble removing it due to being attached to uterus and was very painful when finally done. I have been trying to conceive since with no luck and have 3 other children and had no problem conceiving them.
AmandaAugust 22, 2012 at 5:49 pm
Hi everyone! I had the mirena in for about 5yrs never had a problem until now. Well the first week in June 2012 I had to get my mirena redone so I went to my doctor in elkins,wv and they had a new doctor there the first thing he had me do was cough 2times and I never done that when I got the first mirena in and then when it was done I had bad cramps for 3days and I told my husband I dont think thi[Show More]Hi everyone! I had the mirena in for about 5yrs never had a problem until now. Well the first week in June 2012 I had to get my mirena redone so I went to my doctor in elkins,wv and they had a new doctor there the first thing he had me do was cough 2times and I never done that when I got the first mirena in and then when it was done I had bad cramps for 3days and I told my husband I dont think this guy new what he was doing and then I had to go back in june 28th,2012 for them to check it and the doctor checked it and I could here him saying uuh because I asked him to cut my stings alittle bit shorter if you know what I mean and the next minute I know is the doctor said it fall out and I was really upset because it should never have fallen out and now they r post to call me to replace it but its going on 2 months that I still have not recive it and no phone calls so I end up calling them a couple of times and they kept telling me that its not there fault that it has not come in yet they said the factory might be running show so I'm looking for another doctor to do it and I still upset about it and ever since its been out now I start gaining weight I was in a size 2 and now i'm in a size 7 pants. I just want to put my story out there.
TashaAugust 19, 2012 at 7:10 pm
I've had Mirena for 2 years and 2 months. I've had no problems that I can recall, lighter and irregular periods. No paid when inserted. Recently I'm having a lot of breast paid, cannot touch them, it hurts. I do have other issues that occured after Mirena, but not sure if it's related to Mirena. After all these comments, I'm going to have it removed ASAP. I am affraid to remove it, because of[Show More]I've had Mirena for 2 years and 2 months. I've had no problems that I can recall, lighter and irregular periods. No paid when inserted. Recently I'm having a lot of breast paid, cannot touch them, it hurts. I do have other issues that occured after Mirena, but not sure if it's related to Mirena. After all these comments, I'm going to have it removed ASAP. I am affraid to remove it, because of excessive bleeding (a friend at work had excessive bleeding for a long period of time, when she removed Mirena), but I don't want it in me anymore. Thank you all for sharing your experiences, that really opened my eyes.
chelsiAugust 17, 2012 at 9:47 pm
No doctor wants to address this issue I had the meraina put in in 2006 it was so painful I could barley walk for days then really bad infection and I have suffered with pain and nasty pid infections for 6 years I have cried to my doctor what's wrong with me I was fine before I asked so many questions got 2nd opinion all the same made me feel like an idiot tellin me to stop takin bubble baths and w[Show More]No doctor wants to address this issue I had the meraina put in in 2006 it was so painful I could barley walk for days then really bad infection and I have suffered with pain and nasty pid infections for 6 years I have cried to my doctor what's wrong with me I was fine before I asked so many questions got 2nd opinion all the same made me feel like an idiot tellin me to stop takin bubble baths and wash my panties in diff soap I MEAN REALLY A GIRL IN HER TWENTIES SHOULD HAVE AN INFECTION EVERY WEEK EVERY MONTH FOR YRS???? 3 WEEKS SMELLY NASTY DISCHARGES WITH CRAMPIN 1 WEEK WITH DOC PERSCRIBED MEDS WICH THEN GAVE MEBAD YEAST INFECTION FOR A WEEK WITH MY PERIOD WHICH WAS WAS SO PAINFUL STILL DOCTORS SHRUGG ME OFF MAKIN ME FEEL LIKE ITS ME??? I'VE BEEN HOSPITALIZED TWICE THIS YR 2012 THIS LAST TIME FOR FOUR DAYS SO SICK IN SO MUCH PAIN I CAN'T POOP OR EVEN PEE WELL AND WAS SENT ON MY WAS WITH PID FOR LIFE OVERIAN CIST AND TOLD TO FOLLOW UP WITH OB HA I ALSO HAVE SO MANY UNPAID MEDICAL BILLS TIME MONEY PAIN. WHO'S GONNA HELP THIS IS RUINIG MY LIFE MY RELATIONSHIP MY SELF ESTEEM MY KIDS. MY JOB??????????
LAKIESHIAAugust 15, 2012 at 3:59 pm
I am a mother of four boys ...prior to my last born I had the mirena placed in April of 2008. Once I had it inserted in it was painful for the first couple of days then the pain went away .BUT during intercourse it was VERY painful. after a couple of months maybe a year I started to have abdominal pain it was so serve I ended up going to the ER they diagnosed me with a cyst on my left ovary that n[Show More]I am a mother of four boys ...prior to my last born I had the mirena placed in April of 2008. Once I had it inserted in it was painful for the first couple of days then the pain went away .BUT during intercourse it was VERY painful. after a couple of months maybe a year I started to have abdominal pain it was so serve I ended up going to the ER they diagnosed me with a cyst on my left ovary that need emergency surgery ...I was scheduled maybe a week or two later to have surgery. The cyst was so big that it damages my ovary to the point I had to have it removed darning that removal of the cyst. Which was terrifying because my now husband and I wanted to have a child together. Not even two weeks after the surgery I suffered a PE (Pulmonary Embolism) which had me hospitalized for awhile and was on comedian (blood thinners) for 6 months. Not even a year later I had another cyst again I had to have removed during that last surgery I then had the mirena removed. With a 50% percent chance of becoming pregnant.... a couple of months later after the removal of the mirena we were fortunate my husband and I were able to conceive.....But because of the PE I had to have injections everyday and sometimes twice a day of blood thinners during my pregnancy. That was a horrible experience I ever encounter in my life time…. I have had to go through so much pain, financial struggles, depression; all because of a birth control which I was told would be a good birth control.
MichelleAugust 10, 2012 at 4:02 am
The data provided reflect the experience with the use of Mirena in the adequate and well-controlled studies for contraception (n=2,339) and heavy menstrual bleeding (n=80). For the contraception indication, Mirena was compared to a copper IUD (n=1,855), to another formulation of levonorgestrel intrauterine system (n=390) and to a combined oral contraceptive (n=94) in women 18 to 35 years old. The [Show More]The data provided reflect the experience with the use of Mirena in the adequate and well-controlled studies for contraception (n=2,339) and heavy menstrual bleeding (n=80). For the contraception indication, Mirena was compared to a copper IUD (n=1,855), to another formulation of levonorgestrel intrauterine system (n=390) and to a combined oral contraceptive (n=94) in women 18 to 35 years old. The data cover more than 92,000 woman-months of exposure. For the treatment of heavy menstrual bleeding indication (n=80), the subjects included women aged 26 to 50 with confirmed heavy bleeding and exposed for a median of 183 treatment days of Mirena (range 7 to 295 days). The frequencies of reported adverse drug reactions represent crude incidences. The adverse reactions seen across the 2 indications overlapped, and are reported using the frequencies from the contraception studies. The most common adverse reactions (≥5% users) are uterine/vaginal bleeding alterations (51.9%), amenorrhea (23.9%), intermenstrual bleeding and spotting (23.4%), abdominal/pelvic pain (12.8%), ovarian cysts (12%), headache/migraine (7.7%), acne (7.2%), depressed/altered mood (6.4%), menorrhagia (6.3%), breast tenderness/pain (4.9%), vaginal discharge (4.9%) and IUD expulsion (4.9%). Other relevant adverse reactions occurring in <5% of subjects include nausea, nervousness, vulvovaginitis, dysmenorrhea, back pain, weight increase, decreased libido, cervicitis/Papanicolaou smear normal/class II, hypertension, dyspareunia, anemia, alopecia, skin disorders including eczema, pruritus, rash and urticaria, abdominal distention, hirsutism and edema.
MichelleAugust 10, 2012 at 3:55 am
This is the link to the list of side effects & warnings with Mirena & several are ones that you need to report immediately if your Dr is listening go to another Dr & report it to the FDA maybe they will do something about it. http://www.drugs.com/sfx/mirena-side-effects.html
MichelleAugust 10, 2012 at 3:42 am
I had mirena inserted 14 months ago after my Drs told me I need to be on 2 forms birth control due to a cancer medication that could cause birth defects. I couldnt take the medication because it messed my heart. So i dont need 2 forms of bcontrol anymore. I did have clots heavy painful periods with low back pain, ovary pain, breast tenderness, nausea diarreah before mirena then it slowed down to j[Show More]I had mirena inserted 14 months ago after my Drs told me I need to be on 2 forms birth control due to a cancer medication that could cause birth defects. I couldnt take the medication because it messed my heart. So i dont need 2 forms of bcontrol anymore. I did have clots heavy painful periods with low back pain, ovary pain, breast tenderness, nausea diarreah before mirena then it slowed down to just a couple days a month with Mirena & on chemo. But Since June 24th, I'm having constant spotting for over a month now. The Drs did an ultrasound & said I have fibroids & my uterus is alittle swollen & they were going to take the Mirena out but worried about me getting an infection while on chemo so they put me on ortho pill with Mirena to stop the spotting. After 2 weeks on it, Im having terrible low back pain, nausea, diarreah, bleeding with grape size clots, ovarian cysts, & ovary pain so bad it feels like theyre going to explode. The Dr took me off the pill & said be ready for more bleeding so here I wait for what's next & once I'm off the chemo & can I'm getting Mirena removed. I can say way b4 the Mirena for about a year I was doing good. I took Promeva & it really helped balance me out. There's a lot of estrogen being put in certain foods thats making us sick & men have "moobs" man boobs. It's in most fast food. I ate more things at home. Easy stuff like salads fruits veggies organic chicken even canned, beans or sunflowers seeds almonds on my salads & I started to feel alot better & my periods were better. I took an advil or midol at the 1st sign of pain or headache and every 5-6hrs as needed & it kept my pain from getting out of control. My advice to any of you who are going thru this or have had or are getting the Mirena taken out or are now trying to get pregnant you NEED to balance your hormones & make sure your body has the vitamins & minerals u need thru good nutrition. Try to eat organic fruits & veggies whenever u can & get a hormone balancing product like Promeva. Its worth a try & it wont hurt u. It's not a drug like these ones the manafacturers like BAYER & the makers of YAZ keep lying to us & having our Drs push on us. It should be illegal what they are doing to us.Obviously, the Drs aren't listening or are getting kickbacks for pushing this crap. Maybe it's they're way of population control. All the crap they're putting in the food & these meds are a toxic mix that keeps them rich. Jerks. I'd like to say more than that but don't want cuss on here. I'm going to start Promeva now because I can't keep just bearing with the pain. Please try to eat mostly good foods & try to stay away from the fast food stuff I know it's hard but it's worth it to stop this pain & awful feeling. We are stronger than we think we are & after years of all this crap we can make it thru this & feel better. God Bless & well wishes to you all.
PriscillaAugust 9, 2012 at 4:21 pm
I am 35years old, mother of four, after.giving birth to my baby in april i got the mirena inserted about 6weeks after. Reading all the comments above I am sitting here at the doctors office waiting to have it removed, cause I am having all the same syptoms (back pains pains in my pelvic, headaches, having hard time falling asleep and staying asleep, mood swings, massive blood clots, n irregular p[Show More]I am 35years old, mother of four, after.giving birth to my baby in april i got the mirena inserted about 6weeks after. Reading all the comments above I am sitting here at the doctors office waiting to have it removed, cause I am having all the same syptoms (back pains pains in my pelvic, headaches, having hard time falling asleep and staying asleep, mood swings, massive blood clots, n irregular periods. I am in fear that it has did some damage to my uterus cause of the pains i been having for the past week, plus i too have no sex drive..
MichelleJuly 28, 2012 at 4:28 pm
I had Mirena inserted in late March 2012. Two days after insertion I felt as if I had a horrible UTI. My gyn called in an antibiotic, and the following day I tested negative. I had an ultrasound that showed the correct placement of Mirena. The symptoms of UTI and INTENSE pressure continued for a week, when I then insisted it be removed. It was removed. I continued to have the intense pressure[Show More]I had Mirena inserted in late March 2012. Two days after insertion I felt as if I had a horrible UTI. My gyn called in an antibiotic, and the following day I tested negative. I had an ultrasound that showed the correct placement of Mirena. The symptoms of UTI and INTENSE pressure continued for a week, when I then insisted it be removed. It was removed. I continued to have the intense pressure in my abdomen and bladder. I saw my gyn who placed me on another antibiotic and yeast medication. Still symptoms, I then saw a urogyn. My bladder was normal according to the urogyn, yet they placed me on yet another antibiotic. Four months later I had an ultrasound, and it was discovered I have a 6cm ovarian cyst. This is causing bladder pressure and also abdomen pressure. I have been through hell. I had not taken my antidepressant in many months, but due to this whole Mirena disaster I am back on it. Some days I can't get out of bed. I now wait to see if the cyst goes away on it's own, or if I need surgery to remove it.
JelJuly 27, 2012 at 4:39 am
I had my mirena inserted in public health care on 2008. After 2 years I called the clinic for pap smear. They told me they won't do it because I have it on. I went to another clinic the result came out abnormal. Now i'm waiting for my biopsy appointment if I have dysplasia or negative. I'm hoping to be negative i'm too young to die and have 3 kids to taking care. :-(
DawnJuly 23, 2012 at 2:45 pm
I had the mirena inserted August of 2011 and had it removed June 2012...I have never felt so terrible in my life....I have had every listed symptom and side effect....I gained 30 pounds in that short amount of time, had severe acne on my back...I never had one mark or pimple on my back previous..I have photos....was moody, depressed, foggy, suffered from severe hypotension..BP 80/60 caused me to b[Show More]I had the mirena inserted August of 2011 and had it removed June 2012...I have never felt so terrible in my life....I have had every listed symptom and side effect....I gained 30 pounds in that short amount of time, had severe acne on my back...I never had one mark or pimple on my back previous..I have photos....was moody, depressed, foggy, suffered from severe hypotension..BP 80/60 caused me to be orthostatic...the worst was the vertigo and dizziness which is still affecting me...have serious shooting pain in my left arm and my arm becomes numb. I have serious fatigue and severe joint and back pain. I felt like I was out of my mind...my hair was falling out in clumps. I cant believe I gained 30 pounds in 9 months....I am having issues now about how i look with extra weight and acne scars that I never had before. The best thing I ever did was have it removed. NEVER EVER AGAIN!!!!! My libido dropped and was in pain during intercourse...not enjoyable at all....ugh.....
CarrieJuly 23, 2012 at 2:42 pm
I've had the Mirena in aliitle over 4 years now. I had it put in after my second was born. At the 4 year mark, I noticed things were changing. Extreme fatigue, bloating, acne from head to toe, back pain, loss of libedo (that has been going on since I had it put in), and mood swings. Went to family doctor and OBGYN both told me the Mirena wouldn't be causing my problems. They referred me to an [Show More]I've had the Mirena in aliitle over 4 years now. I had it put in after my second was born. At the 4 year mark, I noticed things were changing. Extreme fatigue, bloating, acne from head to toe, back pain, loss of libedo (that has been going on since I had it put in), and mood swings. Went to family doctor and OBGYN both told me the Mirena wouldn't be causing my problems. They referred me to an Endocrinologist...now I have been diagnosed having Hypothyroidism! Putting me on a pill I would have to take for the rest of my life! I was hesitant for about 2 months about taking the pill...I was freaked out. Tired of feeling like crap everyday, I started taking the pill. I started having more energy, but that was it. They upped my dose and got my TSH levels right, but that's when everything got worse. Every symptom I had got worse. In addition, now my acne has turned into cystic acne. I have melesma on my cheeks now, impossible to cover up with make up. I have anxiety attacks. Extreme back pain, sometimes making it hard to even get out of bed. Depression, I find myself crying just thinking how my life was before I had the Mirena. NO health problems, clear skin, happy go lucky. Now, I feel like I'm walking around in a fog. After things had gotten worse after they upped my dose, I did go back and tell them about it...but they still insisted the Mirena had nothing to do with it and just to be patient and things would get better. That's when I started looking on the internet, trying to see if others were having problems with Mirena. I have been reading others post for days now. Feeling sick to my stomach. I have my yearly this week, I really want this Mirena out. Now, I'm scared to death what will happen afterwards, even shaking as I type this. I am 33 yrs. old with 2 kids.
CathleenJuly 20, 2012 at 6:55 pm
I had Mirena inserted in May 2010 and had it removed June 2012. It was very painful having it inserted when I first got it, it was not painful removing it. My doctor asked me why I wanted to have it removed and I had a laundry list of things that have been happening since having it placed. Like the other women my doctor asked me several times if I was SURE I wanted it removed. I gave hime the fol[Show More]I had Mirena inserted in May 2010 and had it removed June 2012. It was very painful having it inserted when I first got it, it was not painful removing it. My doctor asked me why I wanted to have it removed and I had a laundry list of things that have been happening since having it placed. Like the other women my doctor asked me several times if I was SURE I wanted it removed. I gave hime the following list. It started with my hair falling out, and lots of it. I joked that I could never commit a crime because I would at least leave 15 strands of hair at the scene in 5 minutes. I cleaned my brush everyother day. About 3 months into having the device I started to get REALLY bad yeast infections about a week before I started my period, and I needed a prescription to get rid of the infection because the over the counter stuff didn't work. I always had a smell and discharge. Then the crazy moodswings started... I was anxious all the time, couldnt sleep, cried about everything, irritable, and so angry the slightest thing would set me off. I gained about 10 lbs and I eat a very strict diet. I also got acne that was cyst like underneath my jaw bone every month. My relationship has suffered because I was always so angry with my husband about the stupidest things, and so self concious about my smell I didnt want him to touch me. I had horrible cramps and Migraine headaches too. I told my OB about all these things and he kept saying you need to give your body time to adjust. I was a week away from my period and the itching started again, I called my doctor that day and thankfully he got me in and I had him remove it. Today I am a basket case, I have been crying about everything and I flew off the handle at my boss for asking me a question. I have had Mirena out for a month and I feel so much better. All my symptoms have disappeared. I do want to make an appt with my OB because I feel my ovaries are still not right, I keep having pains from them. I had my 1st period without Mirena and it was extremely heavy, but I didn't get a yeast infection or kidney infection like I had been getting while I had Mirena. If I knew that I would have all these symptoms I would never have had it put in.
ChristyJuly 20, 2012 at 4:42 am
Having surgery on July 31st to have ovarian cyst (radiologist called it a tumor, doctor called it a cyst -- first ultrasound I had that the lab tech left the room to get the radiologist to come see the live feed) removed. Today my doctor told me that it is the kind that doesn't stop growing so it has to be removed. The ultrasound I had the other day also showed blood clot attached to or is right b[Show More]Having surgery on July 31st to have ovarian cyst (radiologist called it a tumor, doctor called it a cyst -- first ultrasound I had that the lab tech left the room to get the radiologist to come see the live feed) removed. Today my doctor told me that it is the kind that doesn't stop growing so it has to be removed. The ultrasound I had the other day also showed blood clot attached to or is right by mirena. Not sure if that is normal. Other symptoms: random nausea (no, not pregnant dr checked), unexplained gushes of blood (like someone turned faucet on then it goes off), whitish clumps of matter coming out, cramps, pain during sex, migraines. Because I just found out about sugery today I haven't been able to process if this is due to Merena or not. Will ask doctor at preop appointment on Monday.
JaimeJuly 17, 2012 at 4:41 pm
I am so happy that I found this website. I thought I was crazy. I had the Mirena put in 3 years ago. The insertion should have been a sign from the start. It took approximately 45 min of the doctor trying to get it in place before he decided I needed to be dilated for proper insertion. I was under tremendous pain and bled for days after. I started experience horrible mood swings and headaches. Whe[Show More]I am so happy that I found this website. I thought I was crazy. I had the Mirena put in 3 years ago. The insertion should have been a sign from the start. It took approximately 45 min of the doctor trying to get it in place before he decided I needed to be dilated for proper insertion. I was under tremendous pain and bled for days after. I started experience horrible mood swings and headaches. When I got the Mirena I had long beautiful hair almost to the bottom of my back. It was full and healthy. My hair started getting shorter and shorter without me cutting it. Finally my hair dresser asked what was going on because I had lost at least 50% of the volume of my hair and it was extremely dry and brittle. My hair now is to my chin and very very thin. All without cutting it. I was also experienceing horrible stomach pains, bloating, night sweats, and pains in my chest. I went to the doctor and had every blood test imaginable. Of course everything came back normal. I had an ultrasound done on my uterus and ovaries....normal. I has a hydascan done on my gallbladder...normal. I had an endoscopy .....normal. My doctor couldn't find anything wrong. In the meantime my hair continued to fall out and break off. My stomach pains were so severe sometimes I couldnt even walk. I become so bloated that I literally look 6 months pregnant at times. I have become depressed and unsociable. I was so upset, I would cry all the time. My husband was looking up hair loss in woman and came across the Mirena side affects. Once I started looking at them I realized it was everything I was experiencing. I have had the Mirena removed. My stomach pains have gone away 90% along with the extreme bloating. I still have horrible night sweats and headaches which I hope will go away soon. I haven't noticed any regrowth yet in my hair but it has only been 2 months since I had it removed. My doctor said it can take up to 6 months to get all the hormones out of my system. I have spent thousands of dollars on test and doctors visits along with vitamins, nioxin, etc not to mention the amount of emotional turmoil this has taken on me and my family. I pray that woman look at these posts before they have the Mirena put in.
LisaJuly 14, 2012 at 9:22 pm
As reading all of these comments it is making me wonder about my symptoms. I got the Mirena six weeks after the birth of my daughter in 2011. I had had the copper IUD after the birth of my son and had no problems whatsoever for five years. I have been experiencing severe dizziness for months now and have had numerous lab studies, ENT visits wtih a myringotomy tube put in my left ear and recentl[Show More]As reading all of these comments it is making me wonder about my symptoms. I got the Mirena six weeks after the birth of my daughter in 2011. I had had the copper IUD after the birth of my son and had no problems whatsoever for five years. I have been experiencing severe dizziness for months now and have had numerous lab studies, ENT visits wtih a myringotomy tube put in my left ear and recently a brain MRI with all being normal (thank God), but along with the dizziness I have severe fatigue, muscle weakness and achiness, horrible mood changes along with the cramping and not symptomatic UTI with bacteria in the urine. I mentioned this to my OB/GYN who said the Mirena was "ABSOLUTELY NOT" the cause of my symptoms, but now am wondering if this truly is the cause. I have been miserable for several months now. If the Mirena is causing all of these symptoms as well as the above symptoms, the FDA needs to put a stop to physicians inserting this device.
AmandaJuly 8, 2012 at 7:14 pm
I got the marina after I had my second child. I had the marina for 3 years then I got pregnant.. Had no problem removing mine expecially because it moved from my uterus .. All throughout the time I had it I never had any problems with it. Just sex was painful sometimes... Other then that no problems besides having another child which is a blessing but still happened when I thought I was protected[Show More]I got the marina after I had my second child. I had the marina for 3 years then I got pregnant.. Had no problem removing mine expecially because it moved from my uterus .. All throughout the time I had it I never had any problems with it. Just sex was painful sometimes... Other then that no problems besides having another child which is a blessing but still happened when I thought I was protected.
michelleJuly 6, 2012 at 5:09 am
o my i am 21 i have hot flashes and night sweats low sex drive constant pain sometimes it feels like my insides are falling out if you know what i mean or like contractions nausea during and after sex pain during sex when i do have sex some unwanted hairgrowth on the side of my breasts and ovarian cysts all the time for the cysts they put me on the pill and that tottally didnt work i just lost my [Show More]o my i am 21 i have hot flashes and night sweats low sex drive constant pain sometimes it feels like my insides are falling out if you know what i mean or like contractions nausea during and after sex pain during sex when i do have sex some unwanted hairgrowth on the side of my breasts and ovarian cysts all the time for the cysts they put me on the pill and that tottally didnt work i just lost my insurance and i cant afford to get it taken out without it and my doctor recommended it when i did have health insurance wish i would have not ever got it feel like its ruining my lif
KristinaJuly 5, 2012 at 10:19 pm
I am 45 years old and on my second mirena iud. I got my first one inserted after the birth of my daughter. The 1st one was great. I really don't remember anything to awfull. The second one has been a disaster. I I had to take an ambulance to the ER, I thought I was having a heart attack. All my tests were normal and was sent home. I was sent to my PCP who stated it was a paniac attack. I ha[Show More]I am 45 years old and on my second mirena iud. I got my first one inserted after the birth of my daughter. The 1st one was great. I really don't remember anything to awfull. The second one has been a disaster. I I had to take an ambulance to the ER, I thought I was having a heart attack. All my tests were normal and was sent home. I was sent to my PCP who stated it was a paniac attack. I have anxiety, heart palpitations,stiff neck, joint pain, spotting, racing heart, feeling warm, sweats, dry mouth, I now have borderline high blood pressure( I have always had perfect blood pressure) and no sex drive. I thought I was peri-menopausal but my GYN did thyroid and hormone tests and all were negative. After doing some research it's nice to know their are other women that are going thru this same thing. I thought I was going crazy!!! I have an appointment to get this thing out and I am just going to go thru the rest of my life the natural way.
KylaJune 21, 2012 at 3:18 pm
I had the Mirena inserted in December 2011 and had it removed April 2012. I had been on birth control prior to that since I didn't want to be pregnant. I'm only 23 and didn't want kids. The insertion was extremely painful and to make it worse my body rejected the first one while I was still on the table so they went and got another one and reinserted the new one. It was the worst pain I have ever [Show More]I had the Mirena inserted in December 2011 and had it removed April 2012. I had been on birth control prior to that since I didn't want to be pregnant. I'm only 23 and didn't want kids. The insertion was extremely painful and to make it worse my body rejected the first one while I was still on the table so they went and got another one and reinserted the new one. It was the worst pain I have ever had. I almost passed out while driving home because the pain was so bad. I couldn't even drive all the way to my house. I had to stop at my brothers to sit and rest because I was so lightheaded. I started to have severe cramping and sudden shoots of pain throughout my uterus. They would happen on a daily basis and the pain would take my breath away and make me double over. I was never a mean nor nasty person but once I got the mirena I seem to become bipolar and have major depression. I was horrible to my boyfriend. We fought all the time. I was never like that before. I went to the doctors regarding the pain and they told me it was not from the mirena but I didn't believe them because by then the strings have disappeared. They tried to get it out once but was unsuccessful so I had an ultrasound and they said it was exactly in place but I still wanted it out. They tried again and finally got it out with 2 doctors trying to remove it. Once I had it removed all the pain stopped but I still had severe mood swings and thought I was going crazy. They have since decreased and are slowing going away. I have been trying to get pregnant since the removal but have been unsuccessful. I do have periods so I am having a cycle but still no pregnancy and I'm afraid I have become sterile. It's not my boyfriend because he has been checked. I'm so scared that I will never have kids of my own now.
A. RoeslerJune 20, 2012 at 5:49 pm
I got the IUD insured March of 2008 after having my daughter. It was exteremly painful. I wouldn't walk or even hold my new boorn daughter. I started to get really depressed i was at the point i wanted to die. I never got my period which i thought was amazing. But no the pain got wrose and worse as the months went on. I started to feel like i wasnt a human anymore. My heart rate jumped to over 110[Show More]I got the IUD insured March of 2008 after having my daughter. It was exteremly painful. I wouldn't walk or even hold my new boorn daughter. I started to get really depressed i was at the point i wanted to die. I never got my period which i thought was amazing. But no the pain got wrose and worse as the months went on. I started to feel like i wasnt a human anymore. My heart rate jumped to over 110. And my Thyriod level become 0.01. Finally March of 2010 i had it removed and to this day i have so many issues that the doctors are unclear to what they are. Its a scary factor to know i cant work to provide to me and my daughter becauseof this device. I pray for everyone who is going thru with any of this.
VanessaJune 16, 2012 at 3:47 am
In Dec 2006 i had mirena placed in june 2007 i had emergency surgery as it had eroded out of my womb and was floating around in my peritinial cavity. In 2008 i was diagnosed with uterine cancer my ob wanted to only remove uterus but once inside had to do a full hysterectomy so i am supposed to take hormone replacement therapy for the rest of my life which because of recession can not afford. At th[Show More]In Dec 2006 i had mirena placed in june 2007 i had emergency surgery as it had eroded out of my womb and was floating around in my peritinial cavity. In 2008 i was diagnosed with uterine cancer my ob wanted to only remove uterus but once inside had to do a full hysterectomy so i am supposed to take hormone replacement therapy for the rest of my life which because of recession can not afford. At the time it was unheard of and no attorney would touch my case,shame on them. If it had been addressed at that time maybe fewer of you would have suffered. Please warn everyone you can not to make the same mistake my heart goes out to all who were victimized by this product.
KatrinaJune 14, 2012 at 6:01 pm
I had the mirena IUD put in 6 weeks after my baby was still born in January 2010. I thought it would be good for me since I was taking medicine for hypertension already. Less medication I have to remember. Later I started feeling depressed having panick attacks, migraines everyday, my husband said that he thought I was , memory lost of simple stuff. Now My lower stomach is always hurting and I ha[Show More]I had the mirena IUD put in 6 weeks after my baby was still born in January 2010. I thought it would be good for me since I was taking medicine for hypertension already. Less medication I have to remember. Later I started feeling depressed having panick attacks, migraines everyday, my husband said that he thought I was , memory lost of simple stuff. Now My lower stomach is always hurting and I have a itching and burning sensation on the inside of my stomach. I was scheduled an appointment and they couldn't find the string for the mirena. She refered me to recieve a sono two weeks they found after an hour or searching finally saying schedule a x- ray. When they pulled my file I already had an X-ray on file andright in my side sat the mirena. Immediately she says this is what's causing all your pain. I've also had 4 UTI's and as of yesterday 06/12/12 2 bacteria infections. My surgery is scheduled for July 12,2012. I'm still in a lot of pain, should I be worried of extra damage this can cause for another month.
BrendaJune 7, 2012 at 7:48 am
I had IUD inserted in 2010 and completely regret it. Never had I experienced so much pain. I had chronic abdominal pain for a long time -doctors weren't able to explain the cause for it. It wasn't til I was rushed to the ER that they discovered one of my ovaries had to be removed and not so long ago they discovered my tumors... I will never recommend Mirena to anyone...
DianeJune 5, 2012 at 6:34 pm
I have gained about 50 lbs while on the mirena and I still have the extra weight on me.. I was in horrible pain in my stomach I had a hard time walking... I started with a 24/7 ringing in my ears... I also had horrible pain in my arms and hands... After I took the Mirena out It took a while but my stomach pain is gone, my hands still go to sleep all the time but the pain has gone away.. The ringin[Show More]I have gained about 50 lbs while on the mirena and I still have the extra weight on me.. I was in horrible pain in my stomach I had a hard time walking... I started with a 24/7 ringing in my ears... I also had horrible pain in my arms and hands... After I took the Mirena out It took a while but my stomach pain is gone, my hands still go to sleep all the time but the pain has gone away.. The ringing in my ears will not stop, I have lost over 1/2 of my hearing .. (pretty sure this is from the mirena.. My daughter is know having problems after a few years on the mirena and she is at the doctors now getting it out.. We have been through hell... She is in Pain 24/7 her whole body hurts to the touch.. she has been treated for everything...put on cymbalta... pain meds, steroids, antibotics.. tested postive for ANA ... Sent to every kind of doctor been in the ER many times... Swelling up, breaking out with rashes... Headaches ... can't even open things cause her hands hurt so bad... She has went from 140 lbs to 221.... since the mirena has been put in.. She is 22 without a life... can't even go out with her friends... Sleeps all the time.
RahemaMay 22, 2012 at 3:56 am
And I forgot to mention the lower back pain, so intense that I actually went and bought a new mattress thinking that was the problem and mid day-early evening I cant stay awake!
RahemaMay 21, 2012 at 9:57 pm
I had Mirena placed about three years ago and I swear It took me at almost two years to REALLY blame myself for choosing Mirena....At first it was the constant discharge for SIX months straight I and I swear I kept saying if this doesnt change by the 6 month mark I'm getting rid of it, well it got a little better-discharge wise. Then the RAPID weight gain. I went from 140 to 190 in a matter of mon[Show More]I had Mirena placed about three years ago and I swear It took me at almost two years to REALLY blame myself for choosing Mirena....At first it was the constant discharge for SIX months straight I and I swear I kept saying if this doesnt change by the 6 month mark I'm getting rid of it, well it got a little better-discharge wise. Then the RAPID weight gain. I went from 140 to 190 in a matter of months, no sleep,the mood swings are the worst and for me I think the scars; marks on my face have really taken their toll, I use to really think I was cute...lol Now I have to make myself go ou in public because of the melasma on my face. Now the MORE serious issues I have had. I went to a ENT because of the constant post nasal drip...he immedialty refers me to a different Dr that deals with Thyroid problems, I had to have biopsys done last year they all came back fine,but the nodules are still there! It wasnt until I spoke with a stranger that I linked this all together. I still have the IUD in place and I really need to get it removed, its just the stories that I have read have me scared to do so. I called Mirena to ask them if Thyroid conditions are related and of course they said No, but reading the stories over and over online really had me wanting to look into this. My husband is at the end of his rope with me, I have no sex drive and I hate to see us going through this,my kids are almost afraid of me when I start my mood swings. The past 2 months when I get my period-YES I still get it, theres barley any blood, just discharge , and I was never a heavy bleeder, never had problems with cramping, but the past 2 months have been HORRIBLE, I get this wicked pain in the lower left side of my abdominal area, so bad that I am ready to go to the ER. I have a OB appt in a few weeks and I am going to request to have it removed. And I have to go back for anothe biopsy of my thyroid in a few months.
NikiMay 20, 2012 at 10:09 pm
Jessica if the doctor that put the Mirena in in the first place didn't warn you of the possible side effects and the risks that it can move around your uterus, I would suggest that she is liable for this. And also the fact that she found it "floating around" your uterus suggests to me that it is her fault for not removing it then and there. That sounds negligent if it was not in the right postion [Show More]Jessica if the doctor that put the Mirena in in the first place didn't warn you of the possible side effects and the risks that it can move around your uterus, I would suggest that she is liable for this. And also the fact that she found it "floating around" your uterus suggests to me that it is her fault for not removing it then and there. That sounds negligent if it was not in the right postion but she it left there. I would demand that she remove it free of charge with her insurance covering the other costs or you will sue her.
RachelMay 20, 2012 at 2:47 am
I has my Mirena inserted 8 weeks after I gave birth. My uterus was perfectly normal and I was very excited to not have to worry about birth control for 5 years. I believe I have a high tolerance for pain, so placing the Mirena was no problem, mind you it wasn't a skip in the park, but it was tolerable. Later that day I was having horrible cramps and just attributed it to the insertion. Then days a[Show More]I has my Mirena inserted 8 weeks after I gave birth. My uterus was perfectly normal and I was very excited to not have to worry about birth control for 5 years. I believe I have a high tolerance for pain, so placing the Mirena was no problem, mind you it wasn't a skip in the park, but it was tolerable. Later that day I was having horrible cramps and just attributed it to the insertion. Then days after, and more days after I was bleeding a lot. Having terrible cramps, to the point to being nauseous. Till I just have to give in. I have had the mirena 2 weeks, and went in to get it checked. My doctor couldn't find it. Went for an ultrasound. Couldn't find it. Had a CT scan. Found it! It perforated my uterus! No wonder I was in crazy amounts of pain! I had to have laprescopic surgery to have it removed! The thing that pisses me off the most, is that I not only have to pay to have it put in, but pay to get it removed! I had it for less than a month!!! Unacceptable.
AmyMay 19, 2012 at 8:08 pm
I had my IUD inserted in april 2009. After that i was all downhill mmy moods changed I could sleep at all I began to fight with everyone all the time. Dr.s gave me medication for depression anixety and sleep. I was a police officer and one day I complete lost it none the less in a parking lot at Applebee's one min I am crying uncontrolably another min I am yelling fomr the top of my lungs. unable [Show More]I had my IUD inserted in april 2009. After that i was all downhill mmy moods changed I could sleep at all I began to fight with everyone all the time. Dr.s gave me medication for depression anixety and sleep. I was a police officer and one day I complete lost it none the less in a parking lot at Applebee's one min I am crying uncontrolably another min I am yelling fomr the top of my lungs. unable to cope with the kids and the mood swings I lost custody of my kids cuz i just couldn't keep it together. I lost my Job as a police officer too. All the Dr.s told me I was usffering from PPD but i didn't feel that way at all. I even at one point had to check myslf into a Psyc ward but I couldn't control my sadness, temper, anger, anxiety and i mean it in a combo of all this. My son is 3 and I still dont feel right. MY body is not acting how it use to. I have a weird discharge that i never know when its going to pour out of me and a smell to accompany it. I have had in a matter of three year 12 yeast infections and they are hard to get rid of. I have had 7 uti's and 4 badder infections. each time i have visited my doctor whem i asked where this is coming from they never offered the Mirena as a reason. instead I was made to feel like i slept around or left with the assumption that I needed to take a good look at my husband. PEE after sex is what I am always told. BUt guess what i don't even have sex anymore the drive is gone. so i know that is not the cause of it. the headache I get are unbareable I was driving with my children in the car and i literally had to pull over and call someone to pick me up. vicodine could put a dent in the pain. so after much discussion my husband decided that he will just go under the knife and no more forms of birth control for us. My body still hasnt recovered from this and my echo and good name has been tarnished by a side effect that no one ever explained to me.
MistyMay 19, 2012 at 6:22 am
I got the Mirena shortly after my son was born. I did not like it at first, as soon as I got home from getting it put in, I started my period for 4 months non-stop after that I had no problems till about a year later when I got it removed. I got it removed because my husband and I wanted to have another baby, shortly after removal I missed 4 periods, hoping we conceived, so I went to my obgyn beca[Show More]I got the Mirena shortly after my son was born. I did not like it at first, as soon as I got home from getting it put in, I started my period for 4 months non-stop after that I had no problems till about a year later when I got it removed. I got it removed because my husband and I wanted to have another baby, shortly after removal I missed 4 periods, hoping we conceived, so I went to my obgyn because I was getting negative pregnancy tests and found out I have poly-cystic ovary syndrome and may not have anymore children again. I am a 22 year old mother to one child!
JessicaMay 17, 2012 at 7:06 pm
I had my IUD put in a little over 4 years ago. Six weeks after my daughter was born. when I went back for my check up 6 weeks later they couldn't find the strings. After poking and searching the doctor decided to try to find it by ultrasound. She found it floating around in my uterus. Then assured me that it was fine and was still working but may be a little difficult to remove when the time c[Show More]I had my IUD put in a little over 4 years ago. Six weeks after my daughter was born. when I went back for my check up 6 weeks later they couldn't find the strings. After poking and searching the doctor decided to try to find it by ultrasound. She found it floating around in my uterus. Then assured me that it was fine and was still working but may be a little difficult to remove when the time came. 2 years ago I started having unexplained abdominal pain , back pain, headaches, mood swings, severe cramping, among many many other symptoms. I went to see my regular doctor about the cramping and stabbing pains. After blood tests, urine test, x-rays, and eventually an ultrasound of my abdomin (with no insurance coverage). They could not find anything wrong and looked at me like I am crazy. (I was beginning to feel that way). So I went back to my OBGYN and she told me I wasn't crazy because they have had numerous complaints similar to mine. But that the only way to remove my IUD would be through surgery. My insurance only covers the placement and (conventional) removal of the IUD. She tried putting me on hormones for the mood swings and the tender breasts. It didn't work. I just kinda dealt with it for awhile. Now it is extremely bad. I can't sleep at night because it hurts to bad. Sex with my husband has become almost impossible because of the pain afterwards. It's like the better the sex the more the pain afterwards. The stabbing pains in my lower abdomin are getting worse and happen all day long. I have started spotting and I haven't had a period since 1 month after my IUD was put in. I returned back to my OBGYN and saw a diffrent doctor within the office. She seemed a little more concerned and wanted to try antibiotics and negotiating with my insurance company ( which I had already tried several times). Honestly I had gotten my hopes up this time. After she talked with the insurance company she received the same results I did. They will not cover the removal through surgery. Her next suggestion was to try the hospital and see if they would work with me on the payment of my surgery (which I have also tried before). The hospital wants $2500.00 down just to use the hospital (the remain $6000.00 to be paid out in monthly payments). Radiology and anstesiology have to be paid in full before the surgery. Pathology cannot give me an estimate up front, I just have to wait on the bill to come after the surgery.Totaling an estimated $10,000 before the surgery. I called MIRENA and was told the MAY reimburse me after everything has been paid in full. I am a single mother and can not cover these expenses. And I haven't even asked how much my OBGYN charges to perform the surgery. Please if anyone has had this same problem or may have suggestions please let me know.
SummerMay 17, 2012 at 12:33 am
I just had Mirena removed after the full 5 years. during that time I have had rashes, infections, eye blurriness. weight gain weight loss, I have had joint pain, muscle fatigue. Insomnia Anxiety. I have spent thousands on tests that fail to show much. I have been diagnosed with fibro. Mild RA. I have for 1.5 years paid for medication and visits back and forthto the doctor. I feel better for a few [Show More]I just had Mirena removed after the full 5 years. during that time I have had rashes, infections, eye blurriness. weight gain weight loss, I have had joint pain, muscle fatigue. Insomnia Anxiety. I have spent thousands on tests that fail to show much. I have been diagnosed with fibro. Mild RA. I have for 1.5 years paid for medication and visits back and forthto the doctor. I feel better for a few days and then crash again. WIthin 1 hour of having Mirena removed I started to sweat profusely and dropped 3 lbs in water weight in a day! 24 hours later I feel more like myself. I woke today with no painor stiffness and no hangover feeling. I have been alert all day and am amazed at the difference.
KimMay 9, 2012 at 6:12 pm
I first had the mirena for 4 yrs and started having bleeding, weigh gain, terrible migranes. Was told it was due to the fact the mirena needed to be replaced. Did that and now 10-14 day periords w the new mirena in, still headaches, foggy vision, head, thinking, severe mood swings....having it removed friday and am scared for the mirena crash.
NicoleMay 4, 2012 at 7:17 pm
The Mirena took a month of my life and I am slowly trying to get it back. Blood pressure spiked to 185/110, I had the shakes, I was foggy in my head, couldn't remember anything, irritable, hateful, no libido at all, slept maybe 4 nights in total for that month. I went to the emergency room and had it taken out. Now, 2 days post-mirena I am still foggy, though not as much and I am still having the [Show More]The Mirena took a month of my life and I am slowly trying to get it back. Blood pressure spiked to 185/110, I had the shakes, I was foggy in my head, couldn't remember anything, irritable, hateful, no libido at all, slept maybe 4 nights in total for that month. I went to the emergency room and had it taken out. Now, 2 days post-mirena I am still foggy, though not as much and I am still having the full body shakes. I hope this all goes away because it is scary. I did not gain weight, I actually lost, but that is not worth having to take blood pressure medication. I am happy to say that my blood pressure dropped almost back into the normal range 8 hours after removal. I just wish the shaking would stop.
KellyMay 2, 2012 at 10:38 pm
Reading this makes me so upset. 12 years ago, i struggled with my weight (i really liked to eat) and was up to 200 pounds at 5'6. I made a lifestyle change, got into the gym and began eating healthy. a year later, I was down to 129 pds and living with my new man who is now my husband. Maintaining my weight between 130-135 for 3 years, I decided to get Mirena. We don't want to have children s[Show More]Reading this makes me so upset. 12 years ago, i struggled with my weight (i really liked to eat) and was up to 200 pounds at 5'6. I made a lifestyle change, got into the gym and began eating healthy. a year later, I was down to 129 pds and living with my new man who is now my husband. Maintaining my weight between 130-135 for 3 years, I decided to get Mirena. We don't want to have children so in Aug 2005, I got my first IUD. I gained 20 pounds without changing my workout routine or diet. I figured it was "in love" weight and I began to work harder and never saw results. Loving the fact that I never had a period and accepting that I was going to always fight my weight and assuming that once you hit 30 your metabolism really slows down and no matter what you do you will gain weight (dumb I know), i got my second Mirena put in July 2010. I was engaged in August 2010 and loving life. Working out so much harder and counting calories to get down to 139 for my wedding day. I got gown to 145 (wow a entire 4 pds) in a year and when I had the final dress fitting, it fit like a glove. The final fitting was August 2011 and by October 2011 (my wedding day) I weighed 160 pounds. I gained 15 pounds and had to SQUEEZE into my dress that I paid a very pretty penny for. I sadly assumed again that it was "stress" or "age". I went to the doctor in December 2011 to get tested for a thyroid condition and she told me I was perfectly healthy. I cried in her office telling her that being "overweight" is not healthy. In January 2012, I counted calories, not consuming more than 1200-1500 a day and started playing roller derby. 5-6 a week of pure cardio and strength training. NO change in body and no weight loss. On April 6, 2012 I have the IUD removed. I have since gained an additional 5 pounds and retain water. I am still playing roller derby and I can't lose a pound. I was once a fat girl who was being treated for depression. I feel like i have become that fat girl again and I am back on anti-depressant medication. I know my symptoms are not as bad as everyone elses but I have been but I have worked too hard to become happy and healthy to have this drug take it away. No doctor thinks that a "birth control" can make it impossible to lose weight. Apparently, they do not listen to their patients. It has been a month and no changes.....
NicolletteMay 1, 2012 at 8:20 am
i had no prior femal issues i have 3 kids under the age of 6 so decided to get http://www.mirena-us.com/ after a few weeks of mild to sever pain above my pelvis bone and many obgyn apts i had it removed for the last year ive had same pain it finally got so sever had surgery 4-20-12 cycts and other things removed from overies and uterus have another apt with my dr this wends to see what eles th[Show More]i had no prior femal issues i have 3 kids under the age of 6 so decided to get http://www.mirena-us.com/ after a few weeks of mild to sever pain above my pelvis bone and many obgyn apts i had it removed for the last year ive had same pain it finally got so sever had surgery 4-20-12 cycts and other things removed from overies and uterus have another apt with my dr this wends to see what eles this thing did to me, my biggest fear would have to say the chance of not having more kids
latoyaApril 29, 2012 at 3:53 pm
i have had mirena for about 3 years. i have gain 45 lbs with know change in my diet. I have swollen feet and hands, irregular cycle, back pain, breakouts, headaches, weight gain, depression, low libido, pelivic pain and body aches. i havent had a normal cycle since i had mirena. Im going to have it removed this week and i know there is going to be some more problems so im preparing for it...
nikiApril 20, 2012 at 1:34 am
Margeaux, your doctor is either a liar or stupid if she said that "Mirena only releases hormones in the uterus". It doesn't matter where something is released in your body, it travels throughout your whole system. If you read the Mirena info leaflet it says that the hormones released by the Mirena are even found in breast milk. It's like getting a needle in your arm and the doctor saying the drugs[Show More]Margeaux, your doctor is either a liar or stupid if she said that "Mirena only releases hormones in the uterus". It doesn't matter where something is released in your body, it travels throughout your whole system. If you read the Mirena info leaflet it says that the hormones released by the Mirena are even found in breast milk. It's like getting a needle in your arm and the doctor saying the drugs only stay in your arm because that is where you got the needle. Absolute ridiculous rubbish!
MaggyApril 18, 2012 at 4:27 pm
I decided to get the mirena after having my 3rd child. I have never had ANY problem with birth control before and my doctor told me the Mirena was the newest and best thing out there so far. So I went forward with it. There was a lot of discomfort when having it incerted, felt a lot like having the contractions from birth. This lasted for several weeks.Then the pain went from contrtraction type pa[Show More]I decided to get the mirena after having my 3rd child. I have never had ANY problem with birth control before and my doctor told me the Mirena was the newest and best thing out there so far. So I went forward with it. There was a lot of discomfort when having it incerted, felt a lot like having the contractions from birth. This lasted for several weeks.Then the pain went from contrtraction type pain to sharp pain. Then the mood swings set it, the snapping, getting mean, it was pure hatred to everything and every one. The mood swings would tear me from pure fury to pure crying. I was miserable. And put my family in termoil - almost lost everything, my kids, my husband - I finily had the mirena removed and EVERYTHING stoped with in a few days! No more cramps, no more mood swings, and I was able to be apart of my family again!
reneeApril 15, 2012 at 7:21 pm
I have had two IUD's. This second one has been in for three years now. I am going blind. I have optic nerve damage and 50% visual field loss. The opthoneurologist is afraid to claim its the IUD's fault. He has told me that its likely to be the cause because there is no other reason for my vision loss. They put the IUD in to prevent migraines, it didn't work and caused me to have permanent vision l[Show More]I have had two IUD's. This second one has been in for three years now. I am going blind. I have optic nerve damage and 50% visual field loss. The opthoneurologist is afraid to claim its the IUD's fault. He has told me that its likely to be the cause because there is no other reason for my vision loss. They put the IUD in to prevent migraines, it didn't work and caused me to have permanent vision loss.
TeresaApril 14, 2012 at 5:34 am
I got the Mirena 2 months after giving birth to my daughter...... life after receiving mirena was a living ell. After Mirena was inserted I was spent plenty of days in the ER and nights in the hospital. They all thought it was just acid reflux. I told my regular obgyn about the problems (acid reflux, mood swings, weight gain and newly anxiety from the pain ) he told me it has nothing to do with t[Show More]I got the Mirena 2 months after giving birth to my daughter...... life after receiving mirena was a living ell. After Mirena was inserted I was spent plenty of days in the ER and nights in the hospital. They all thought it was just acid reflux. I told my regular obgyn about the problems (acid reflux, mood swings, weight gain and newly anxiety from the pain ) he told me it has nothing to do with the mirena. I request on numerous occasion for it to be removed he wouldn't remove it and other doctors wouldn't remove it bc they didn't insert it was it and I was having complications from imit. Finally I had yet another trip to the Er. Dr finally came to the conclusion the Mirena was causing all my constant visits and requested to my obgyn to remove the Mirena asap. Shortly after that Er visit I was hospitalize and had to have emergency surgery to have my gallbladder removed due to gallstones attack and intestinal problems. I had the Mirena removed as well. My life hasn't been the same since I the day I got the Mirena. :-(
MargeauxApril 13, 2012 at 6:00 am
I just got my Mirena removed to day after having it for a year . I had been planning on taking it out back in Feb. but when I went to the doctor and told her my complications she said that it had nothing to do with the Mirena cause the Mirena only releases hormones in the uterus. My complications were I hardly ever wanted to have sex and usually when my husband is ready I am but after the Mirena f[Show More]I just got my Mirena removed to day after having it for a year . I had been planning on taking it out back in Feb. but when I went to the doctor and told her my complications she said that it had nothing to do with the Mirena cause the Mirena only releases hormones in the uterus. My complications were I hardly ever wanted to have sex and usually when my husband is ready I am but after the Mirena for awhile I started to look at him like really I don't want to and that was starting to be a problem I wanted it in my mind but my body was not up for it. I got the Mirena in March 2011 in January I was buying jeans in a size 5 now I'm up to a size 11 in jeans and a xsmall/small in shirts and now to a large my body looks so big usually if I gained weight it was just in my stomach just a little pug but not my whole body and that just made me feel unattractive to my hasband and myself. I had back pains so bad in my lower back to where sometin=mes it was comfortably for me to sit down or when sitting down to lean back and feel comfortable. My pelvic area hurted sometimes like a real bad cramp. I felt it sometimes during intercourse but my hasband said that he didn't. Mood swings were something else sometimes I would be happy and then in a split second I'm mad and just really mad to were I was hollering at my kids and husband for small things, and then sometimes I would just feel so sad like something is just not right but I couldn't figure out why I was so sad or what was wrong with me why do I feel this way. But my doctor just kept telling me that she knew for sure that it is not the Mirena and that the Mirena is the best thing going for me, but I just decided to see for myself because don't no one knows your body like you the doctors are not with you or inside your body they can only go by what you tell them and what they have studied and doctors and scientist are humans just like us and make mistakes even though they would rather cover theres up make a patient seem wrong. But when I got my Mirena removed they took some swabs from me to take test of any complications or infections and will get back to me I pray there wasn't any just my mood swings,back pains, and decreased libido.
ChristinaApril 10, 2012 at 12:13 am
I hate this Mirena creation!!!!! I had the mirena in for about 1 year and three months after a year of having it in my hair started falling out and breaking off! Everytime I got into the shower I would cry because it was coming out sooo bad. My partner even noticed that it looked as he would say "Different" I was talking to my sister about it, and she said she had the same exact issues and that i[Show More]I hate this Mirena creation!!!!! I had the mirena in for about 1 year and three months after a year of having it in my hair started falling out and breaking off! Everytime I got into the shower I would cry because it was coming out sooo bad. My partner even noticed that it looked as he would say "Different" I was talking to my sister about it, and she said she had the same exact issues and that it was the Mirena she had it removed after the doctors said her white cell count was off. freked her out and she said her hair was falling out soooo bad too. so I had it removed a little over a month now and since then my MID BACK was severly hurting I couldnt get out of bed for a whole day! Ive never had back problems ever! and my hair is kinda growing back its sooo bad I have nice curly hair but since I had the mirena ive lost at least half of my volume in my hair it doesnt even look good when i wear it up. I am now going to do a hormonal cleane to get my body right again. I cant wait!
SuzanneApril 8, 2012 at 4:01 pm
I had the mirena ring in for two years. My last mammogram was clear 2 yrs ago. I have now been diagnosed with breast cancer. I believe they are related. I am looking for other women who have had similar experiences. Please contact me if you have experienced this or know anyone who has. Suzanne
HankApril 7, 2012 at 5:21 am
My girlfriend and i decided we were ready to have our first child oct 2011 so she had the IUD removed. in January i look her to the ER because she was having severe abdominal pain. turns out she had an ovarian cyst that ruptured and she had to get surgery that night. we also found out she was 3 weeks pregnant that night. She got a pap smear at OBGYN and it came back abnormal... she had the IUD for[Show More]My girlfriend and i decided we were ready to have our first child oct 2011 so she had the IUD removed. in January i look her to the ER because she was having severe abdominal pain. turns out she had an ovarian cyst that ruptured and she had to get surgery that night. we also found out she was 3 weeks pregnant that night. She got a pap smear at OBGYN and it came back abnormal... she had the IUD for about 10 months. We think the Mirena IUD is the cause of the ovarian cyst and maybe even why my girlfriend may have cancer in her cervix. If i lose my girlfriend and my baby to be i will have nothing. after leaving the military it is her who makes me feel human and alive. I love her so much and when i found out we were having a child it couldn't have gotten better. If i lose the two greatest things in my life because of pharmaceutics corruption to make billions... Just makes me sick!!!
RODNEYApril 4, 2012 at 12:20 am
MY WIFE AND I WENT IN FOR THE I.U.D N THE DR SAID IT WAS OK N SHE WASNT AT ANY RISK LIES NOTHING BUT LIES MY HAPPEN TO BE 5 WEEKS PRERG.. PRIOR TO THE INSETCTION OF THE I.U.D HE DIDNT PROPERLY CHECK MY WIFE WE LOST OUR ONLY DAUGHTER BECAUSE OF THE INCOMP.. DR
KellieMarch 29, 2012 at 1:42 pm
Having mine removed TODAY! Yay...I am way overdue though. My original appointment to have it removed was while my mom was nearing end of life and I just couldn't handle anything else. While I had mirena, I thought it was great...but I have to say, all the things I'm reading about, I experienced as well. I experienced post pardom depression before I had mirena so when it returned I didn't think m[Show More]Having mine removed TODAY! Yay...I am way overdue though. My original appointment to have it removed was while my mom was nearing end of life and I just couldn't handle anything else. While I had mirena, I thought it was great...but I have to say, all the things I'm reading about, I experienced as well. I experienced post pardom depression before I had mirena so when it returned I didn't think much of it. I did notice the weight gain but didn't contribute it to the IUD. I had cramps and some back pain but nothing major..sounds like I was one of the luckier ones. I hope everyone begins feeling better soon and I pray I don't crash after the removal as many have indicated they have. I do worry about that - and the fact that it's pinching now and causing some ovarian pain. I will visit this site again to see how everyone else is doing. Hopefully, everyone will find improvement in their symptoms soon!
AmandaMarch 22, 2012 at 5:59 pm
I cannot tell you how much better I feel after reading these posts from all of you. I have had MANY of these same problems myself, I really thought I was alone..that I was sick or going through early menopause(I am 36) for no reason. I had mine placed about 2 years ago,(excruciatingly painful and was in bed for four days bleeding) and in that time I have gained 25 lbs with NO change to my diet or [Show More]I cannot tell you how much better I feel after reading these posts from all of you. I have had MANY of these same problems myself, I really thought I was alone..that I was sick or going through early menopause(I am 36) for no reason. I had mine placed about 2 years ago,(excruciatingly painful and was in bed for four days bleeding) and in that time I have gained 25 lbs with NO change to my diet or activity level.My feet and hands tingle and go numb, I have developed insomnia, and I have been sick with more viruses and "bugs" than I ever have in my whole life. I get weird bleeding after intercourse, and sharp cramping out of the blue just above my pubic bone. I investigated PCOS, thinking it was the cause of my SERIOUS HAIR LOSS( I was told I have "female pattern hair loss"), even though my two older sisters, mother and 3 maternal aunts had NO issues with hair loss. I subsequently found out I have some kind of "growth" on my right ovary, (that was NOT there prior to the placement of this hideous iud), which is being followed with quarterly ultrasounds and hopefully will not require surgery.... My lab work and ultrasounds ruled out PCOS, iron deficiency, and thyroid problems, so I have been baffled as to why all of this is happening to me. I was told I was just going to have to buy Rogaine, and exercise more, along with prescription meds for the cystic acne. I have ZERO sex drive, and my self esteem took a long walk off a short pier...I am scheduled to have this HORRIBLE thing removed on the next week, and I can't wait,all though a bit afraid of the pain:( Thank god I am not alone in this..
SallyMarch 21, 2012 at 7:24 am
Mirena was recommend by my doctor for my severe menstrual bleeding. i thought I give try. when I gave birth to my second child in England the doctors there told me not to use any kind hormones. Here in the US i mentioned it to my OB/GYn. She reacted surprised and dismissed my comments. I was so desperate for relief that I decide to try the Mirena. After 5 month I begged my to doctor to remove it.[Show More]Mirena was recommend by my doctor for my severe menstrual bleeding. i thought I give try. when I gave birth to my second child in England the doctors there told me not to use any kind hormones. Here in the US i mentioned it to my OB/GYn. She reacted surprised and dismissed my comments. I was so desperate for relief that I decide to try the Mirena. After 5 month I begged my to doctor to remove it. I had all the side effects that they mention. After the removal I started to bleed. I checked it out and the information I got was that it can happen. After 11 days heavy bleeding I was rushed to the emergency room and received 7 units of blood. I was so weak they couldn't even perform surgery on me. After 3 days horrible days they could stop the bleeding.The day after my release I received an injection to put me on a temporary menopause to stop my cycle. When asked this happened because of the Mirena . They said no. I had 3 months later a hysterectomy at age 36. They told me it is a decision between life and death. I couldn't imagine going through that ordeal again. I still believe all this happened because of Mirena.
KarenMarch 20, 2012 at 10:08 am
I am 44 and had my Mirena put in 17 months ago. It was a painful procedure and the intense pain lasted a few weeks. I bled heavily for 6 months to the point where I was prescribed meds to stop it. I was told by my Dr that it was a great form of birth control and that 50% of all female Drs in their 40's had this device. I have been suffering with soreness and stabbing pain in my left ovary for [Show More]I am 44 and had my Mirena put in 17 months ago. It was a painful procedure and the intense pain lasted a few weeks. I bled heavily for 6 months to the point where I was prescribed meds to stop it. I was told by my Dr that it was a great form of birth control and that 50% of all female Drs in their 40's had this device. I have been suffering with soreness and stabbing pain in my left ovary for the last year, back pain on my left hand side. I still have periods though lighter, but my breasts hurt like crazy for 2 weeks out of 4 and this month, my left breast is agony, the a stabbing pain eminating from inside the nipple area to under my arm as well as the nomal tenderness and swelling. I had loads of bloodwork done 5 months ago and everything is normal. I feel so depressed and quick to tears. I am making an appointment with my Dr this week, I want this out regardless of what she says. I am convinced the Mirena is the cause of these symptons
JessicaMarch 14, 2012 at 6:01 am
I had Mirena for a few years with no problems. My period went away and I thought it was great. Well in March of last year on a plane from Fl to TX, I had an ectopic pregnancy rupture in my fallopean tube causing extensive internal bleeding. I had to go in for emergency surgery to get it removed and repaired. I had always had regular check ups to make sure it was correctly placed still. I had absol[Show More]I had Mirena for a few years with no problems. My period went away and I thought it was great. Well in March of last year on a plane from Fl to TX, I had an ectopic pregnancy rupture in my fallopean tube causing extensive internal bleeding. I had to go in for emergency surgery to get it removed and repaired. I had always had regular check ups to make sure it was correctly placed still. I had absolutely no indication I was pregnant since it stops your period. Mirena almost killed me, I would never recommend it.
JackieMarch 13, 2012 at 3:49 am
After having my 3rd child my doctor recomended the Mirena since we werent sure about having mor kids yet. At first I only suffered severe cramping and heavy bleeding all the time but a few months down the road i started having bad headaches and depression. Then I started having bad moodswings and rage. I would lash out at my kids and husband for no reason at all. I could not sleep at all durin[Show More]After having my 3rd child my doctor recomended the Mirena since we werent sure about having mor kids yet. At first I only suffered severe cramping and heavy bleeding all the time but a few months down the road i started having bad headaches and depression. Then I started having bad moodswings and rage. I would lash out at my kids and husband for no reason at all. I could not sleep at all during the night but during the day i felt so fatigued from not getting any sleep. Then my hands started tingling all the time like they do if they were to fall asleep. After about 6 months i noticed my hair started to get real thin and brittle and at about 9 months i looked in the mirror and you could see right to my scalp. I started googling my symptoms and could not believe all the blogs that came up from the mirena with people with the same problems. I demanded my dr take it out the very next day. He told me it will take at least 6 months with good nutrition and vitamins to even get my body close to where it was and be toxic free. This thing needs to be banned!!!
shellyMarch 10, 2012 at 1:42 am
I had the MIrnena in for about 3 months, i was sick for the whole 3 monhths i had it in, i had unbelievable discharge, abdominal pain, depression, mood swings, bleed pretty much the whole time,an awful odor, headaches and generally feeling sick 24 hours a day. i went to the ER and they told me i had an infection and gave me antibiotics, i felt good while on the meds and than about a month later i[Show More]I had the MIrnena in for about 3 months, i was sick for the whole 3 monhths i had it in, i had unbelievable discharge, abdominal pain, depression, mood swings, bleed pretty much the whole time,an awful odor, headaches and generally feeling sick 24 hours a day. i went to the ER and they told me i had an infection and gave me antibiotics, i felt good while on the meds and than about a month later i went to my gynecologist and asked to have it remove, they removed it and told me it was really infected cause of the odor and gave me 2 antibiotics, one for a bacterial infection and the other just in case it got in the blood stream as it was infected i think for the whole time it was in. i would not suggest anyone use this as a birth control as i think it does make us woman really sick. i have since i got it out, felt like a normal person again.
SandiMarch 10, 2012 at 12:14 am
I had the Mirena after going into preterm labor with my daughter in 2008. I decided to get Mirena until I was psycholoically ready to have another baby. In 2010 I decided I wanted to remove Mirena and try to have another baby. Because I did not have a period for the 2 years on Mirena I was not surprised when it didn't start right away. However it is 2012 and I have only had 3 periods and am now i[Show More]I had the Mirena after going into preterm labor with my daughter in 2008. I decided to get Mirena until I was psycholoically ready to have another baby. In 2010 I decided I wanted to remove Mirena and try to have another baby. Because I did not have a period for the 2 years on Mirena I was not surprised when it didn't start right away. However it is 2012 and I have only had 3 periods and am now inferitile without explanation. I called Bayer which was listed on my Mirena information and they took my complaint but stated they have no problem with infertility with Mirena and they were just filing the complaint but did not have to report it to anyone. The information was just for them. My little sister followed my lead and got the Mirena after me and now she has had it removed and still does not have a period and can not get pregnant. I have been told several times by my doctor that Mirena is not to blame however I have not had these issue before. I feel that my doctor does not care about finding the problem with me and wants to blame it on explained menopause onset but can not account for my period coming on 1 a year.
KassandraMarch 9, 2012 at 2:08 pm
I had the Mirena about 5 months after having our second child. From that point on, I had severe migraines that turned into migraines with an aura...it was so bad that I would loose vision in one eye. I had to be treated at the ER numerous times because over the counter meds would not help. Come to find out these migraines became so bad that there was a risk that it would cause a stroke due to t[Show More]I had the Mirena about 5 months after having our second child. From that point on, I had severe migraines that turned into migraines with an aura...it was so bad that I would loose vision in one eye. I had to be treated at the ER numerous times because over the counter meds would not help. Come to find out these migraines became so bad that there was a risk that it would cause a stroke due to the blood vessels in the back of my head swelling and putting pressure on nerves and so forth. (I was only 30yrs old) That was not the worst. I had to travel out of stat to attend training for 6 weeks--husband and kids stayed home. Within 3 days, I began to bleed, pass out, become dizzy, weak, and unable to eat. I ended up in the ER during my lunch hour and in emergency surgery within 45 minutes. This was very scary for me. I was alone, my husband couldn't reach make it to Florida in time to see me before my surgery. The doctor came back and said "Did you know you were pregnant?" I replied, "Must be a mistake, I have the Mirena and can't be pregnant for 5 years". She told me that this is very common in the ER and that I was pregnant but everything ruptured, I had severe internal bleeding, lost the baby, and if I did not have surgery in 30 minutes she may not be able to save me--my organs had begun to shut down due to the blood filling up the cavaties inside my torso/stomach. They called the hospital chaplain to say a prayer and talk to my husband on the phone when I went into surgery. Thank God I made it out okay but my baby did not and I no longer have my fulopian tube. It was horrible and I would NEVER recommend this product to anyone! I did file a complaint with the FDA.
shondaMarch 9, 2012 at 6:17 am
WOW...what an emotional overwhelming feeling...to read all your stories and finally not feel so crazy. i am 27 with two kids.i recieved the mirena iud jan of 2008 6 weeks after having my daughter. my doctor made this product seem amazing...effective birth control for 5 years and NO side effects i thought of course this sounds great!!! the initial recieving the iud wasnt that bad..a little discomfo[Show More]WOW...what an emotional overwhelming feeling...to read all your stories and finally not feel so crazy. i am 27 with two kids.i recieved the mirena iud jan of 2008 6 weeks after having my daughter. my doctor made this product seem amazing...effective birth control for 5 years and NO side effects i thought of course this sounds great!!! the initial recieving the iud wasnt that bad..a little discomfort but tolerable...well fastforward 4 years and i am MISERABLE...i have looked up my symptoms and even asked for advice on forums as to what could be wrong with me... headaches, hair thinning, extreme bloating at ALL times, headaches, abdominal pain, pelvic pain, shortness of breath, aching body and joints, NO desire for sex....emotionally im a wreck irritable, intense mood swings, depression, anxiety, extremely fatigued,weak all the time, emotional meltdowns, problems with affection.....YEAH i know all this happened AFTER getting the mirena. I found this site along with many more sites with women and the same stories and problems as me...needless to say i am in the process of finding a doc in my area to get this thing out of me!!!!!! I never thought about this being the cause of my symptoms/issues because my doctor only made this thing sound remarkable. I am nervous and excited to get it out...nervous that it wont be easy to remove since ive had it sooo long also that there could be organs or something damaged from it and excited at possibly/HOPEFULLY being the old,,happy...affectionat...healthy person i was before the iud, I hardly ever had a headache prior and felt good all the time was full of energy!!!this iud hs taken 4 years of my life and made them hell...and took from my kids the healthy vibrant mom they deserved.
MercyMarch 7, 2012 at 2:36 am
I had my first baby at age 20, he was born may 2011. The marena was recommended to me by my gynec. It was inserted after 6 weeks. In july of 2011 chronic pain on both sides of my body. in and out of the hospital no one could figure out what was wrong. this past Feb 2012 the pain hit me again. went to e.r. again and this time a gyne. called in. ultra sound revealed I had a hematoma cyst on the ovar[Show More]I had my first baby at age 20, he was born may 2011. The marena was recommended to me by my gynec. It was inserted after 6 weeks. In july of 2011 chronic pain on both sides of my body. in and out of the hospital no one could figure out what was wrong. this past Feb 2012 the pain hit me again. went to e.r. again and this time a gyne. called in. ultra sound revealed I had a hematoma cyst on the ovary. rushed to surgrey and now i am less one ovary/tube. I never recd risk factors or written info from my dr when inserted. Is this what has caused all of my pain? surgeon sd i would of died if i hadn't gotten in when i did. they left the iud in. i see my reg dr this month. im having it removed and consult an attorney. pretty scarry. i had no idea it cause an ovarian cyst. mine was a hematoma cyst. i could of bled to death.
RebeccaMarch 5, 2012 at 5:26 pm
I had the Mirena IUD in for 2 and a half years. I had it placed in November of 2009 right before the FDA issued a warning to Bayer about their false advertisment and for not disclosing the full risks about mirena. I had been experiencing severe depression, body aches and fatigue, irregular periods and bleeding, weight gain, breast tenderness, an ovarian cyst, serious back pain, and horrible achne.[Show More]I had the Mirena IUD in for 2 and a half years. I had it placed in November of 2009 right before the FDA issued a warning to Bayer about their false advertisment and for not disclosing the full risks about mirena. I had been experiencing severe depression, body aches and fatigue, irregular periods and bleeding, weight gain, breast tenderness, an ovarian cyst, serious back pain, and horrible achne. I had never experienced any of these, until I had the Mirena inserted. I just had the mirena removed last week, which took them over an hour of digging in my uterus BLIND SIDED to find it and remove it and I am now experiencing the Mirena Crash. It is awful. My body got, i guess "addicted" to the amount of hormones the mirena was producing and now my body is detoxing and I feel just horrible. My emotions are haywire, my body is hurting, I'm extremely exhausted, horrible headaches, I'm itchy, bad cramping, and much more. You would think that I'm recovering from being a drug addict with how I feel and look. I WOULD NOT recommend mirena for anyone.
kimberlyFebruary 23, 2012 at 11:33 pm
I got married at 19 and we had our first child not long after. Our second son came a few years later. We decided two were enough for now and reviewed our options. We chose mirena because it seemed like the best route for us. Now 4 years later I've been to the emergency room and my dr numerous times for ovarian cists. No one in my family nor I have ever had a history of female problems such as this[Show More]I got married at 19 and we had our first child not long after. Our second son came a few years later. We decided two were enough for now and reviewed our options. We chose mirena because it seemed like the best route for us. Now 4 years later I've been to the emergency room and my dr numerous times for ovarian cists. No one in my family nor I have ever had a history of female problems such as this. Now I am in pain most of the time and I wish I had never received the mirena.
AliceFebruary 22, 2012 at 7:57 pm
I had my first and so far only child in March of 2008 and at my 6 week postpartum visit discussed birth control with my doctor who highly recommended the Mirena. I had the Mirena inserted in May 2008 and within 2 months I was no longer lactating. At the time I was unsure of what was happening and assumed something was wrong with me. In the Fall of 2010 I had noticed other problems over the years a[Show More]I had my first and so far only child in March of 2008 and at my 6 week postpartum visit discussed birth control with my doctor who highly recommended the Mirena. I had the Mirena inserted in May 2008 and within 2 months I was no longer lactating. At the time I was unsure of what was happening and assumed something was wrong with me. In the Fall of 2010 I had noticed other problems over the years and did some research which showed me other women were experiencing the same issues while on the Mirena. My symptoms included violent mood swings, breast tenderness, nausea, periodic uterine muscle spasms, and increased pain and cramping during and around my menstrual. In January 2011 I decided to have the Mirena removed to severe and constant pelvic pain that increased while in a sitting position, tests later revealed that I had an infection in my uterus and was placed on an antibiotic. I waited a few months before trying to conceive and had my first positive pregnancy test on 6/19/2011, later I had my first miscarriage at 5 weeks on 6/28/2011 which resulted in an ER visit. Again we waited a few months before giving it another try and on 1/7/2012 we had our second positive pregnancy test. This time at 9 weeks I felt the familiar cramps and spotting and at my doctors office through ultrasound we found out that the fetus had stopped growing at 8 weeks and there was no heartbeat, that was on 2/16/2012. I've since then managed to pass the fetus and most other tissues on my own and am currently on medication to give it a boost and hopefully empty out the rest preventing a D&C. I have a follow up visit scheduled for Monday 2/27/2012 and fully intend to discuss yet again the possibility of the Mirena interfering. What I've seen is that a lot of women still have thinned uterine lining which is one of the ways Mirena prevents pregnancy and this prevents proper implantation of the embryo and leads to miscarriage after miscarriage. I'm going to ask my doctor to check the lining of my uterus and if she won't do it or tries to brush it off like before then I will be finding a different doctor.
AlexandriaFebruary 21, 2012 at 6:27 am
I failed to mention that while on the Mirena, I was treated for 3 cases of BV and nearly 12 yeast infections.
AlexandriaFebruary 21, 2012 at 6:23 am
I had Mirena inserted after the birth of my daughter in 2008. Prior to this, I had been on birth control since the age of fifteen due to the diagnosis of endometriosis. My ObGyn insisted I used Mirena as a replacement for traditional coc's since it was practically "worry free" and told me if I did not like the change or the convience Mirena offered, I could simply have it removed. At first, I [Show More]I had Mirena inserted after the birth of my daughter in 2008. Prior to this, I had been on birth control since the age of fifteen due to the diagnosis of endometriosis. My ObGyn insisted I used Mirena as a replacement for traditional coc's since it was practically "worry free" and told me if I did not like the change or the convience Mirena offered, I could simply have it removed. At first, I felt as though Mirena was the most spactacular form of birth control on the market. I went on a health kick and actually LOST nearly 70 lbs. while on Mirena. Innitially, I attributed my violent mood swings and severely negative behavior to post-partum depression. As the year progressed into the second year, I started to experience the darker side of Mirena. Prior to the use of Mirena, my face was nearly perfect (despite the occasional blemish). Now my face is permantently scarred from my fight with severe cystic acne. In fact, from the end of 2010 on, I had been seen for a number of ailments ranging from severe anxiety and depression, heart palpitations,severe weight-loss, cystic acne and (worst of all) alopecia (thats right, I lost 1/3 of my hair). Innitially, I was tested for hormone imbalances as well as hyperthyroidism; both of which came back normal. I was perscribed Buspar for anxiety (which made me feel worse). It got to the point (I call it my dark days) where all I could do was curl up into a ball and cry. My whole family (including my fiance') was afraid to interact with me in fear of my unstable demeanor. To be quite honest, if it weren't for my daughter, I would have ended it all then. On a hunch, I took it upon myself to get the real facts on Mirena and came across a few forums claiming the severely negative side-effects, as well as testimonials from women who experienced identical symptoms as me. My doctor assured me that it was NOT the Mirena. Despite his protest, I YANKED THAT SUCKER OUT BY MYSELF!!! A year has passed since the removal of the Mirena, and I'm happy to say, my hair has has grown back, my mood swings have subsided and I no longer feel the need to hide from my loved-ones. However, all is not back to normal (seeming to be long-term effects of a silicone- based IUD). I now have joint pain and inflamaton as well as symptoms of accute fibromialga. Honestly, I do not wish to be compensated. I just want this particular method of birth control to be taken off the market, so that women are no longer lulled into a sense of security by companies like Bayer; whose only goal is to sell their product as well as the hell that goes along with it. Bring this company to justice!!!!
SamanthaFebruary 20, 2012 at 8:07 pm
I had a child in 2004 after birth I had the copper iud put in for bc I went 5 yrs with no problems it was like it wasn't even there. When time came to replace it my doctor thought i should use the mirena taking her advice I had it put in the cramping and pain was way more severe then I remembered with the copper for the first couple of weeks I had severe cramping and was just uncomfortable altoge[Show More]I had a child in 2004 after birth I had the copper iud put in for bc I went 5 yrs with no problems it was like it wasn't even there. When time came to replace it my doctor thought i should use the mirena taking her advice I had it put in the cramping and pain was way more severe then I remembered with the copper for the first couple of weeks I had severe cramping and was just uncomfortable altogether. My doctor thought it would supress over time but things just got worse I started getting ovarian cysts that hurt so bad I would run to the er thinking that there was something terrible wrong in that area. Along with irregular bleeding and not being able to perform my job with the amount of pain I called my doctor and she thought I just needed to let my body get used to the hormones in it. well as time went by over the year and half period I had it I gained 30+ lbs was moody and overly depressed finally one day I had I guess what I thought was a nervous break down but the doctors thought I was either depressed or had severe anxiety I was in and out of the er cause I litterally thought I was loosing my mind I didin't want my husband to touch me and I didn't want to go anywhere I was at the bottom so with the pain still persisting I had it taken out and replaced with the copper iud in which I think I will be taking out this next week I still get sever cramping and lower back pain all the time and very heavy bleeding when I had the copper iud the first 5yrs I had no bleeding or cramping I just had no problems. All the mirena did for me was cause lots of medical bills and pain and sufering I would not recommend it to anyone. I still get severe pain on my left ovary alot not sure the cause my doctor isnt either. I dont feel that since I removed the mirena my problems have gone yet.
AshleyFebruary 17, 2012 at 5:06 pm
I had the mirena put in back in November 2011. It was extremely painful when put in and days after. I went to the dr to get it checked out and the mirena was not in place. They did an ultrasound and couldn't find it. Had xray that same day and they could see it but could not tell where it was exactly so then back to the dr for another 4d ultrasound and they still couldn't see it. They scheduled a [Show More]I had the mirena put in back in November 2011. It was extremely painful when put in and days after. I went to the dr to get it checked out and the mirena was not in place. They did an ultrasound and couldn't find it. Had xray that same day and they could see it but could not tell where it was exactly so then back to the dr for another 4d ultrasound and they still couldn't see it. They scheduled a mri which I had to wait 2 weeks until I they could fit me in. After all that trouble they still could not see it and had to do exploratory surgery on Dec. 19th. The iud was found in my omentum. The whole time I had the iud in my body it was painful... my dr said that it should not hurt and that it probably wasn't the iud causing the pain. I know that it was the Mirena iud causing my pain. I had to spend $155 to have the Mirena put in, $175 for the MRI, $20 for my Dr. to read the MRI, $150 for the surgery,(I have not gotten the bill for Anesthesia), I am sure there are some I am forgetting too. Not to mention the money I spent on gas going back and forth to the dr and my time. I have three children so I also had to have someone to watch them while I went to appts. My body still does not feel right. I am breastfeeding my baby and just a couple of days after getting the Mirena put in my milk supply dropped tremendously. If you are considering getting the Mirena IUD don't! It is not worth it! I can't tell you how much stress it caused me and my family while I was going through this.
lanaFebruary 17, 2012 at 4:26 pm
im 23 years old, i had the mirena in for about a mounth. I blead the hole time and had exstream amount of pelvic pain. The mirena acully came out of me on its oun. I had a PID, i had to go to the hospidel for the exstream amout of pain i was in. found out i had ovaerin sists, blatter infeactin. nothing but pain and problems. I wouldnt recomend this to my worst enemy. im still stugling with pelvic [Show More]im 23 years old, i had the mirena in for about a mounth. I blead the hole time and had exstream amount of pelvic pain. The mirena acully came out of me on its oun. I had a PID, i had to go to the hospidel for the exstream amout of pain i was in. found out i had ovaerin sists, blatter infeactin. nothing but pain and problems. I wouldnt recomend this to my worst enemy. im still stugling with pelvic pain. i hope they stop making this iud the hole time i was moody and the whole works.
FeliciaFebruary 11, 2012 at 8:55 am
After the c-section with my son that ruptured my uterus, my docter said to me it would be best for me to not have anymore atleasted for awhile. So I started looking for birth controle, I have herd someone say that Mirena was the best. So I talked to my docter, and it was decided. I would have it put in. And there it started 4/20/11. The pain was so bad just from having it put in its self, a week a[Show More]After the c-section with my son that ruptured my uterus, my docter said to me it would be best for me to not have anymore atleasted for awhile. So I started looking for birth controle, I have herd someone say that Mirena was the best. So I talked to my docter, and it was decided. I would have it put in. And there it started 4/20/11. The pain was so bad just from having it put in its self, a week and a half later still the same pain. Me thinking its just because its a new bc and all, but the pain never went away. Not knowing what was going on with me. The side effects I believe to be experiencing are as followed. contraction like pain in my lower pelvic area lower back pain bad headaches dizziness hurts during sex burns when I pee (often) tender breasts oder and discharge numbness and tingling in the hands and feet (sometimes) excessive abdominal pain I have been to the ER, they gave me some antibiotics and sent me home. I went to the docter a few weeks later and they gave me more antibiotics and some ibuprofen. I asked her if it could possiabaly be the Mirena, she said no, but I really truely think it is.
NiveaFebruary 11, 2012 at 2:00 am
Wow, after all this reading I really need someone to contact me about this Mirena. Im only 38 and have experience earlly menopause to other possible problems that I cant or any doctor can explain. Mood Swings Night Sweats when I get sick. gain weight badly constant feeling of discharge but nothing but clear liquid. tired. fatigue. contant been sick feeling. heart lpalpatitons contast going to pe[Show More]Wow, after all this reading I really need someone to contact me about this Mirena. Im only 38 and have experience earlly menopause to other possible problems that I cant or any doctor can explain. Mood Swings Night Sweats when I get sick. gain weight badly constant feeling of discharge but nothing but clear liquid. tired. fatigue. contant been sick feeling. heart lpalpatitons contast going to pee to the bathoroom blood in my urine unexplainable. finding some blisters in my scalp libido really bad I just want some answers to why Mirena doesnt give you warnings about early possible menoupause..
KerryFebruary 10, 2012 at 12:52 am
I am 43 and I had the Mirena in March 2010 due to terrible periods. After about a month l started to get boil type pimples on my face, which l thought was strange as l have never suffered with pimples before in my life. These pimples were so painful that my whole face as sore. They were huge, green and ugly. So l went to my GP and he prescribed antibiotics for the next 3 months. These meds did abs[Show More]I am 43 and I had the Mirena in March 2010 due to terrible periods. After about a month l started to get boil type pimples on my face, which l thought was strange as l have never suffered with pimples before in my life. These pimples were so painful that my whole face as sore. They were huge, green and ugly. So l went to my GP and he prescribed antibiotics for the next 3 months. These meds did absolutely nothing. Every day l had a new visitor on my face. Then l started to experience other strange things happening to my body, which turned my life upside down. Things like: Heart palpitations Unable to sleep Boils on my shoulders Walking in a slow pace with no energy Sweats – Day and night Not wanting to leave the house Anxiety Depression Constant thrush No appetite Panic attacks – One really bad attack driving home from work, had to call an ambulance So I went back to the GP as my life was going downhill rapidly and didn’t know why. I had a great life and there was no reason for all this to be happening to me. My wonderful GP tells me that l need to go on antidepressants and l need to go talk to someone (skeletons in the closet).....WHAT umm NO l don’t! So l go home and tell my hubby and he says all this started to happen after your Mirena procedure you should look it up. WHAMMO........ Went to a new GP and demanded to get it out ASAP. This GP told me that she takes the Mirena from women every week due to side effects. REALLY, hmmm not surprised. I decided that when l get the Mirena out to get the ablation procedure as l don’t want a period and l would give that a go. Had the procedure 3 months ago now. Woke up with no bleeding at all and apart from cramps felt great. Still today l have not had a period. Still healing from the acne have scars now and l have a little anxiety but nowhere near as severe as what it was. My test results came back saying l had an infected uterus and that the Mirena was imbedded low and cased this. The GP gave me a script of antibiotics but l didn’t have to use it as l felt great. I would never advise any women to get the Mirena, it’s just not worth the side effects.
BriennaFebruary 8, 2012 at 8:04 pm
I had the Mirena IUD put in place shortly after having my son. This Mirena device was inserted January 16, 2011. After insertion, severe cramping occurred and I felt my uterus trying to "expel" the device. I experienced a lot of bleeding and pain for at least two weeks following the procedure. I still had periods on and off for the majority of the year. Some symptoms I have experienced and are st[Show More]I had the Mirena IUD put in place shortly after having my son. This Mirena device was inserted January 16, 2011. After insertion, severe cramping occurred and I felt my uterus trying to "expel" the device. I experienced a lot of bleeding and pain for at least two weeks following the procedure. I still had periods on and off for the majority of the year. Some symptoms I have experienced and are still experiencing are weight gain (upwards of 40lbs), constant discharge with odor, irritability/anxiety, headaches (feels like it's behind my eyes), high blood pressure, and mild to moderate mood swings. I have spoken to my OBGYN and they have informed me that there is no need to worry as long as the device is still set in place properly (with ultrasound confirmation). I feel as though I should have chosen a different birth control method, and I am scared for future problems such as infertility issues, uterine damage, and prolonged hormone effects.
CarrieFebruary 8, 2012 at 4:00 pm
I had the Mirena IUD put in on Jan 19th 2012 and was in severe pain and bleeding for 3 weeks. I went in for the recheck and the IUD had tore through my uterus. I had surgery on 2/6/2012 to have it removed by laproscopy. I was not told by the doctor the risk of complications- only how great this IUD is... most no period, good for 5 years, does not interfere with breastfeeding, low hormone dose so s[Show More]I had the Mirena IUD put in on Jan 19th 2012 and was in severe pain and bleeding for 3 weeks. I went in for the recheck and the IUD had tore through my uterus. I had surgery on 2/6/2012 to have it removed by laproscopy. I was not told by the doctor the risk of complications- only how great this IUD is... most no period, good for 5 years, does not interfere with breastfeeding, low hormone dose so side effects are minimal. Within a day of receiving the IUD my milk supply was less than half of what I was producing despite increased pumping to try and increase my supply. Three weeks later my milk supply is gone completely. My left knee has had five surgeries and I do have arthritis, but my knee was doing great all through the pregnancy and I was not experiancing any problems. After I got the IUD my joint pain was intolerable and I could barely walk around anymore. This is only my second day out of surgery and my knee pain is gone. This was a horrible experiance and I do not recommend this as an option for any woman. Come to find out after reading, the risk for tearing through the uterus is increased with breastfeeding. It would have been nice to know this before they put this thing in me. If I would have known my risk was greater I would have waited to try this option until after I stopped breastfeeding.
LeahFebruary 2, 2012 at 4:13 pm
I got the mirena in early 2008 after being convinced by commercials and my gynecologist that it was the best form of birth control available with little side effects. My doctor explained that there would be a little pain on insertion and the first couple of months I might be a little hormonal but after that I would be fine. He insisted that his wife had it and loved it. Because I only had one c[Show More]I got the mirena in early 2008 after being convinced by commercials and my gynecologist that it was the best form of birth control available with little side effects. My doctor explained that there would be a little pain on insertion and the first couple of months I might be a little hormonal but after that I would be fine. He insisted that his wife had it and loved it. Because I only had one child, I questioned about how soon I could get pregnant if I ever decided to have it removed. My doctor informed me that immediately upon removal I would be able to conceive. I read the little info pamphlet I was given, and nowhere on the booklet did it inform me of all the complications I would have, and nowhere did it say that it could cause infertility otherwise I would have never decided to try it. Immediately upon insertion I almost fainted from the pain. I started having cramps and lower back pain and spotting immediately. The first couple of months I spotted constantly. My lower back hurt constantly the entire time I had the mirena. My joints hurt, my feet swelled, and I was constantly irritable. My pelvic area always hurt, and I would have sharp pains where my ovaries are. Half the time I felt like I was pregnant, and even invested in numerous pregnancy tests. After a few months of having the mirena, I started having anxiety and depression issues. I had never had a migraine in my life, and I began to have migraines for days at a time. I began having problems with my joints and back, and was told by a doctor that I had arthritis at 26. I don't even know how much money I spent going to the doctor for numerous issues the entire time I had the mirena. Eventually I became so unstable that I would have anxiety attacks leaving me scared to leave my house. I went to the doctor and was diagnosed with agoraphobia, but was never informed that the mirena could be the issue. I was prescribed medication after medication, and nothing helped. Things just got worse. I was to the point where I didn't want to live. I was advised to see a counselor, and he sent me to a mental hospital. I was in the hospital for a week having to take a temporary leave of absence from my job. When I returned to work two weeks later, I was treated as if I was incompetent to perform my job duties and as if I was crazy. A short time after this I was fired. A couple of months after this, after almost a year of having the mirena, my husband and I decided that I should have it removed so we could try to have another baby since I wasn't working anyways. I had the mirena removed in December of 2008. Shortly after having the mirena removed, I no longer used all the mental meds I was prescribed. Although most of the issues eased once it was removed, nothing has really completely left. I still have joint pain and back pain. I still have occasional migraines. I still have occasional anxiety attacks, but nothing like when that object was inside my body. I haven't used any form of birth control since, and here three years later, I have yet to become pregnant. All these years, I have felt like everything was my fault. The other day a friend informed me that he had heard on the news about some mirena users being infertile. Immediately I started doing my research, and began to put two and two together. All the medical issues I have had since the mirena are the same things other women are complaining about. This is not just coincidence. I almost lost my sanity, my family, and I lost a great job. I have problems now that I never had before. My husband and I have been trying to have a baby for 3 years, and I never had a problem conceiving before. I'm extremely concerned now of the possibility that I may never have another child. And at the same time, I am scared to even go to the doctor to find out if I am no longer fertile. After all the emotional damage I have endured which was apparently caused by the mirena, I'm not sure if I can endure not being able to conceive. I am sooooo angry. I feel like I have been deceived by my doctor, the fda, and bayer. I feel like I have been robbed of soooo much. How could they not say that this product could cause life debilitating issues??? Why didn't they inform me that I may not be able to have kids afterwards?? Why were they allowed to make the decision that this product was safe without giving us all the true facts? If I had known that there was even half a percent that I might not be able to conceive I would never have decided to put this evil object in my body. Why were so many young women blinded of the real facts? Why were doctors so quick to say that there were no serious side effects if any at all?? I thought this is supposed to be a free country. Is freedom of choice not included?? Why did others make the decision that the benefit of mirena outweighed the risks? Why didn't we have the option to know the risks?? These people should be ashamed of themselves. They can never make up for what they have taken away from so many women. Shame on all who knew and did nothing!!! Look at all the lives you have taken in your own hands as if you are God!! Nothing can ever repair the damage caused in the lives of so many people. Bayer should spend the rest of its corporate life repaying all those whose lives didn't matter to them, all those whom they viewed as dollars signs instead of human beings.
CarolFebruary 1, 2012 at 7:14 pm
I have had the Mirena IUD for 7 years. I got it because I was having non-stop heavy bleeding for like 80 days straight. The IUD stopped the bleeding almost immediately. I have gained 70 pounds, have occasional night sweats, ovarian cysts, and really bad fatigue. I never linked it to the IUD until I started reading all the comments. I had the IUD removed a little over 2 weeks ago because I had[Show More]I have had the Mirena IUD for 7 years. I got it because I was having non-stop heavy bleeding for like 80 days straight. The IUD stopped the bleeding almost immediately. I have gained 70 pounds, have occasional night sweats, ovarian cysts, and really bad fatigue. I never linked it to the IUD until I started reading all the comments. I had the IUD removed a little over 2 weeks ago because I had a blood clot in my leg and 1 in both lungs. Well, today I am going to mental health for intake because about 7 days after I had the thing removed, I started having thoughts of self-harm. I will be in partial hospitalization for at least 7 days. Right after having it removed I gained a ton of water weight, so much that my clothes barely fit. Now that seems to have passed and I've lost 2 pounds. I didn't have the psychological difficulties while I had the IUD, but I have been on an anti-depressant the whole time I had the thing in. Now I have to be on a more potent anti-depressant because of the thoughts. Actually, when I went to see my primary care doctor and told her about the thoughts and that I thought it was a result from having the IUD removed, she didn't agree with me. Then she went on-line and started reading all these posts on different web sites from women who have had the Mirena-crash. She believes me now:) Well, I'm off to go check into the psych ward...unbelievable.
KristinJanuary 18, 2012 at 7:06 pm
I had my merina iud inserted in 8/2010 and after months of my memory getting worse and worse and fighting with my husband and yelling at mychildren and blurred vision, anxiety attacks, and generally feeling like I was going crazy I checked into my symptoms and found all of you ladies who thankfully shared your stories. My doctor intended to start my on anti depressants anti anxiety stuff etc. I [Show More]I had my merina iud inserted in 8/2010 and after months of my memory getting worse and worse and fighting with my husband and yelling at mychildren and blurred vision, anxiety attacks, and generally feeling like I was going crazy I checked into my symptoms and found all of you ladies who thankfully shared your stories. My doctor intended to start my on anti depressants anti anxiety stuff etc. I had merina removed today and I hope this solves my problems.
MegJanuary 16, 2012 at 10:01 pm
I had the Mirena IUD put in 6 weeks after my 2nd childs birth in June 2009. I had no problems with it at first. In December 2010 I got my first period since being on the IUD. After that I got a period every 2 weeks. So I waited it out thinking since I had also just finished breastfeeding my son it was because of that and it would get normal. After a year and having the most painful horrific heavy [Show More]I had the Mirena IUD put in 6 weeks after my 2nd childs birth in June 2009. I had no problems with it at first. In December 2010 I got my first period since being on the IUD. After that I got a period every 2 weeks. So I waited it out thinking since I had also just finished breastfeeding my son it was because of that and it would get normal. After a year and having the most painful horrific heavy periods I could ever imagine to the point I could barely walk I went to the Dr in December 2011 to get the IUD removed at that point I was convinced it was something with the IUD what else reason could there be? My Dr could not find the strings and I said well I have never been able to feel the strings I am sure the Dr who put it in cut them too short. She DUG with a hook for 20 minutes trying to find it and couldnt, I thought I was going to throw up from the pain. She ordered a transvaginal ultrasound and it still wasnt found. Then I had an x-ray done which finally showed that the IUD had perforated and gone into my abdominal cavity. Now, next week I have a Dr appt to meet a new Dr and schedule a laparoscopic surgery to have it removed. My husband and I were really hoping for another baby and now I am scared to death this will make me infertile.
CrystalJanuary 11, 2012 at 9:03 pm
I got the mirena iud back in september of 2005. Not only was it a painful insertion, but I had the numerous issues right off the bat. I experience the random week long "break thru" bleeding but that was warned about. What I was not told about was that I would then start getting severe migraines, some related and some not related to high blood pressure. I started getting cysts on my ovaries, some t[Show More]I got the mirena iud back in september of 2005. Not only was it a painful insertion, but I had the numerous issues right off the bat. I experience the random week long "break thru" bleeding but that was warned about. What I was not told about was that I would then start getting severe migraines, some related and some not related to high blood pressure. I started getting cysts on my ovaries, some that ended up having to be surgically removed. I developed numerous kidney stones, to date I've had 7. I had the iud removed in October of 2008 but the damage was irreversible. Here I am in January 2012 having to go for surgery for a stent in my renal artery and reading amonst things on here it seems the mirena could have been the start of it all causing the stenosis in the first place.
KerriJanuary 10, 2012 at 9:29 pm
I had my daughter on 1/6/10 and a few weeks after that got the mirena and at first everything was fine then about a month later I started having chest pains, shortness of breathe, I have been hospitalized and I have been diagnosed (sp) with sevre depression and many more side effects. I am not sure what to do but I would like some advise I still have mirena but after reading all the side effects t[Show More]I had my daughter on 1/6/10 and a few weeks after that got the mirena and at first everything was fine then about a month later I started having chest pains, shortness of breathe, I have been hospitalized and I have been diagnosed (sp) with sevre depression and many more side effects. I am not sure what to do but I would like some advise I still have mirena but after reading all the side effects that it has caused is making me think.
becJanuary 9, 2012 at 5:42 am
I had my mirena put in after my 2nd child in 2007 and after having numerous problems - abdominal pain ,internal stabbing sensations , extreme tiredness, lowered sex drive , pain during sex, spotting after sex , yearly ultrasounds on stomach/bladder, monthly large swollen lymph glands under arms , continual sporradic periods,continual UTI,s and UTI symptoms ,foggyness, lethargy and irritability asw[Show More]I had my mirena put in after my 2nd child in 2007 and after having numerous problems - abdominal pain ,internal stabbing sensations , extreme tiredness, lowered sex drive , pain during sex, spotting after sex , yearly ultrasounds on stomach/bladder, monthly large swollen lymph glands under arms , continual sporradic periods,continual UTI,s and UTI symptoms ,foggyness, lethargy and irritability aswell as systemic candidais I finally had it out today. I hope this is a new beginning for me .
KellyJanuary 7, 2012 at 5:12 am
Had Mirena inserted by Kaiser doctor in July 2009 (6 weeks after birth of my son). An like mist everyone else it was the worst pain going in. I also felt ill all afternoon and vommitted from the pain. In July 2010 my husband and I decided to try to give our son a brother or sister so I went to have it removed. After the Dr not being able to feel the strings it was found that the Mirena perfora[Show More]Had Mirena inserted by Kaiser doctor in July 2009 (6 weeks after birth of my son). An like mist everyone else it was the worst pain going in. I also felt ill all afternoon and vommitted from the pain. In July 2010 my husband and I decided to try to give our son a brother or sister so I went to have it removed. After the Dr not being able to feel the strings it was found that the Mirena perforated my uterus and was in my abdomen. Had laparoscopic surgery to have it removed in September (earliest I could get in). I became pregnant in November 2010 but miscarried January 2011. I had a D&C and have been trying to get pregnant ever since. I go in beginning of my cycle to see if my tubes are blocked. I'm thinking the whole IUD perforation and abdomen surgery may have caused scarring and infertility
AmandaJanuary 5, 2012 at 6:51 pm
I had the mirena in for 3 years...it made me have severe Acne which I can't get rid of even after having it removed. The acne has spread to my arms and legs and stomach and chest and back. Also severe weight gain which seems impossible to get rid of. I also have very abnormal periods after having it removed...I will have my normal period but after it ends about a week later I will start to have[Show More]I had the mirena in for 3 years...it made me have severe Acne which I can't get rid of even after having it removed. The acne has spread to my arms and legs and stomach and chest and back. Also severe weight gain which seems impossible to get rid of. I also have very abnormal periods after having it removed...I will have my normal period but after it ends about a week later I will start to have another one with lots of brownish colored discharge. So for about two weeks I can't be intimate with my husband. Then in another two weeks I have it start all over again. I have also had abnormal cells on my cervic not knowing it the mirena was the cause of it. All I know is the company Bayer didn't exclose these severe symptoms before or after I decided to get the mirena!!! If I knew this was going to happen I would never had gotten this contraceptive!!!!
HollyJanuary 5, 2012 at 3:19 pm
I am 56, got this mirena coil, as part of HRT, as I had an early menopause, which I maintain was caused by my thyroid problem, but anyway. I got this put in, not too bad pain on 20/1011 by my gyno, just handed a leaflet, nothing to worry about. My life has been hell since then, and if only someone in the UK would bring about a lawsuit, I would happily, put Bayer behind bars, and the drs, who pus[Show More]I am 56, got this mirena coil, as part of HRT, as I had an early menopause, which I maintain was caused by my thyroid problem, but anyway. I got this put in, not too bad pain on 20/1011 by my gyno, just handed a leaflet, nothing to worry about. My life has been hell since then, and if only someone in the UK would bring about a lawsuit, I would happily, put Bayer behind bars, and the drs, who push this device. Even as a layperson doing research, I know that I would not recommend to anyone with anxiety problems, but they just give it to everyone. I am getting this out this afternoon hopefully, will update as to what the outcome is.
tinaJanuary 1, 2012 at 6:15 am
Had the Mirena inserted on10/7/2009 and it was painless and no side effects. It lessened the duration and flow of my menstral cycle so that was a plus. Weight gain was minimal but I can attribute that to my age and my metabolism slowing down so it was not significant enough to really be concerned. I have read many posts of terrible side effects and feel for those women that have went through th[Show More]Had the Mirena inserted on10/7/2009 and it was painless and no side effects. It lessened the duration and flow of my menstral cycle so that was a plus. Weight gain was minimal but I can attribute that to my age and my metabolism slowing down so it was not significant enough to really be concerned. I have read many posts of terrible side effects and feel for those women that have went through that but thought it was important to say that all people react differently to medications and procedures.
ElleDecember 30, 2011 at 8:49 pm
I got Mirena 8 weeks after my 3rd child was born, nearly 2 years ago. I have had nothing but grief from it. Hurt like a bitch going in, I bled for 3 months straight, couldn't walk for days afterward. Painful sex, horrendous cramping and bleeding, reverse periods, mood swings, back pain, leg cramps, headaches. It's just awful. I am waiting until my insurance kicks in to schedule an appointment, but[Show More]I got Mirena 8 weeks after my 3rd child was born, nearly 2 years ago. I have had nothing but grief from it. Hurt like a bitch going in, I bled for 3 months straight, couldn't walk for days afterward. Painful sex, horrendous cramping and bleeding, reverse periods, mood swings, back pain, leg cramps, headaches. It's just awful. I am waiting until my insurance kicks in to schedule an appointment, but I am afraid that when I go to have it removed, I will receive horrible news about surgery or possibly even a hysterectomy! I do want more children in the future, and I am scared to death that it may not be possible...I also suffer from Muscular Dystrophy. My doctor said this would be the best solution to preventing pregnancy, and i was uncomfortable with it all along, but she assured me it was safe and effective. Doesn't seem so safe and effective to me...
AnnonymousDecember 27, 2011 at 8:39 am
I have a terrible doctor. 8 meeks after my first son was born, My doc encouraged me to get the mirena because my health insurance covered it. He told me it was awesome eveything that the doctors rave about. So I agreed. He told me he would insert the Mirena during my first post-pregnancy pap. When I got there for my pap, he only screened me for STDs he didnt do a cervical scraping for cancer and[Show More]I have a terrible doctor. 8 meeks after my first son was born, My doc encouraged me to get the mirena because my health insurance covered it. He told me it was awesome eveything that the doctors rave about. So I agreed. He told me he would insert the Mirena during my first post-pregnancy pap. When I got there for my pap, he only screened me for STDs he didnt do a cervical scraping for cancer and he put the mirena in and didnt even document he put it in (as a my new doc says). It was very painful and he said it was normal, but this didnt feel normal something felt wrong, but i trusted him and went home. 2 months into it I was having Violent mood swings, hair loss in the right places new hair growing in the wrong places lathargy Depressed random spells of crying Weight gain heart palpitations Severe acute sharp pain and cramping night sweats anxiety in social situations forgetfulness I never had these before, I thought it was mirena right away went to the doc and he said it wasnt the mirena i was crazy and that it was post partum depression and proceeded to tell me I should seek help in the community (mental health, counselling etc) WTF?! Maybe I was crazy, I went home. Things just kept getting worse I started having my period every 10 days. and severe PMS and bad pain. I went and saw another docter, she told me my body just needs to adapt. give more time. Then i started spontaniously lactating, i was sure now that i was pregnant and the mirena was interfereing with a pregnancy, i went to the walk in clinic in town and he tested me and it was negitive but he told me to get checked out. I scheldualed anohter pap and iud removal and then something hapened that shocked me, it was stuck! and it wont come out! now I need a series of tests, why didnt my doc offer to check me out after all the nagging, why did they say these things are normal and i have to adapt? My relationship is taking a huge dive down the toilet because of my episodes of depression and anxiety. and its all because this thing ruined my life!!! if things are bad, im never gonna have a brother or sister for my son.. Im really upset and dont know if this is the docs fault or my own for getting it... :(
CarrieDecember 26, 2011 at 11:01 am
Oct. 9th was my first post. This is my update...Made my obgyn take my Mirena IUD out Oct.16th after having it for 3yrs. My Charlottesville dr. confirmed that it was the mirena causing all my problems..hiatal hirneas, internal bleeding, bleeding ulcers, gastritis, 4 internal bleeding hemroids, a tumor on my liver, confusion, head aches, bloating, weight gain, extreme fatigue, hair loss, vomiting, m[Show More]Oct. 9th was my first post. This is my update...Made my obgyn take my Mirena IUD out Oct.16th after having it for 3yrs. My Charlottesville dr. confirmed that it was the mirena causing all my problems..hiatal hirneas, internal bleeding, bleeding ulcers, gastritis, 4 internal bleeding hemroids, a tumor on my liver, confusion, head aches, bloating, weight gain, extreme fatigue, hair loss, vomiting, mood swings, anxiety, numbness in my extremities, a rash on my back and chest, acne, not being able to sleep, severe pain in my lower back, stomach, chest, legs, and under my shoulder blades, throwing up blood, fever, and now drug induced Lupus!!
NikiDecember 22, 2011 at 11:33 am
I didn't have the Mirena even though I was pressured to have one. Luckily I am old enough to remember the Dalkon Shield to never get an IUD. Can the attorneys find out about the incentives that the doctors are getting? Never in my life have I heard so many doctors rave about a product so much, deny that it causes any problems AND refuse to remove it. Not just on this website but many others. And[Show More]I didn't have the Mirena even though I was pressured to have one. Luckily I am old enough to remember the Dalkon Shield to never get an IUD. Can the attorneys find out about the incentives that the doctors are getting? Never in my life have I heard so many doctors rave about a product so much, deny that it causes any problems AND refuse to remove it. Not just on this website but many others. And never have I heard of doctors getting angry if a patient refuses a drug or treatment but so many doctors get angry if women refuse the Mirena, including my own. I am sorry but what form of contraception should invoke an emotional response from a doctor unless it is impacting on them in some (financial) way?
IrnaDecember 22, 2011 at 12:46 am
First of all Seasons greetings. I've had the Mirena for four years now. I'm an EMT, have been for the last 18 years. In July of this year i woke up one morning with severe, debilitating back pain, swollen ankles, blinding headaches, joint pain, unable to get to my bathroom without assistance. I've been sent for an MRI, and X-rays with no negative results. Last month I had my pap and mammogram done[Show More]First of all Seasons greetings. I've had the Mirena for four years now. I'm an EMT, have been for the last 18 years. In July of this year i woke up one morning with severe, debilitating back pain, swollen ankles, blinding headaches, joint pain, unable to get to my bathroom without assistance. I've been sent for an MRI, and X-rays with no negative results. Last month I had my pap and mammogram done and was sent for a sonogram of my breasts. It showed 2 cysts in the right and one in the left. I'm due for biopsy on January, 11th 2012. I haven't been to work since July 9th, and have been confused as to what's wrong with me. The pain in my joints is unbearable, my lower back, pelvis and entire right upper and lower leg feel like they're breaking and sex with my husband is near impossible. Those headaches are still with me relieved slightly for a short time with prescribed meds. I don't know what to do. All I can say is my quality of life has been severely affected by this implant and someone needs to do something to help the countless women who were grossly under informed about this device from the pits of hell!!
Cindy AnnDecember 20, 2011 at 7:10 pm
After having the Mirena in place for a year and a half, I finally could no longer take irregular bleeding, and monthly back and side pains, along with the constant wetness from the 2 weeks of bleeding that would leave me vulnerable to yeast infections. My doctor kept promising me that my period would start to lighten and then eventually vanish, and I would no longer bleed. While it did lighten ini[Show More]After having the Mirena in place for a year and a half, I finally could no longer take irregular bleeding, and monthly back and side pains, along with the constant wetness from the 2 weeks of bleeding that would leave me vulnerable to yeast infections. My doctor kept promising me that my period would start to lighten and then eventually vanish, and I would no longer bleed. While it did lighten inintensity, it just spread out over 2-3 weeks each month. I have now had it out for about 2 years, and my cycle still runs 2 weeks of bleeding a month (before it was regular 5-7 days- just very heavy). I am still fighting the constant yeast infections from wetness from the prolonged period, coupled with the wetness of ovulation each month. I wish my doctor had NEVER reccomended Mirena and I wish I had never seen a TV commercial showing such a happy woman with the Mirena- it sure did not leave me happy.I am still dealing with it today.
vickiDecember 17, 2011 at 3:15 am
My daughter has 2 children and going to school. She has a triple major which at the college she attends has never had any one with a triple major in 50 years. She decided to have the mirena inserted to prevent another pregnancy due to her busy schedule and the fact they were happy with having 2 children. She's had this for about a year and a half. Since having this placed, she's experienced mi[Show More]My daughter has 2 children and going to school. She has a triple major which at the college she attends has never had any one with a triple major in 50 years. She decided to have the mirena inserted to prevent another pregnancy due to her busy schedule and the fact they were happy with having 2 children. She's had this for about a year and a half. Since having this placed, she's experienced migrane headaches daily, spotting almost all month every month, depression, and forgetfullness. She went to the doctor yesterday to have this removed finds out it's not there. Two ultrasounds were performed and it's nowhere to be found. Luckily they had sense enough to use alternative methods of birth control and not get pregnant. After this news they get news today her finace's company he works for burned to the ground. They now have no source of income. Bad news 2 days in a row not to mention a week before Christmas. So now they have the burden of no income in addition to what happened to the Mirena. I would not reccomend this form of birth control to anyone. She has had nothing but problems. She now has major health issues and someone needs to take care of this.
terraDecember 16, 2011 at 5:41 pm
I had the Mirena inserted after my second son was born in March of 2010. My husband and I were losing our health insurance and wanted something semi-permanent in place in case we decided to have more children later on. I had almost two years of a good experience, it prevented me from getting pregnant again, but... yesterday I just found out I am 8 weeks pregnant and on top of that the DR couldn't [Show More]I had the Mirena inserted after my second son was born in March of 2010. My husband and I were losing our health insurance and wanted something semi-permanent in place in case we decided to have more children later on. I had almost two years of a good experience, it prevented me from getting pregnant again, but... yesterday I just found out I am 8 weeks pregnant and on top of that the DR couldn't find the stupid Mirena inside of me with an ultrasound. He couldn't find the strings to take it out and he said it could have passed out of my system but he said theres not much I can do at this point. I cannot believe how ineffective this device is... its terrible and if my posting this story keeps even one woman from deciding to get it than its worth it.... just say no!!!
brittneyDecember 14, 2011 at 5:28 am
I had mirena put in a year and a half ago after i had my son. I have been problems ever since. And my doctor just kept telling me it was normal so finally i just couldnt take it anymore and went to a hospital and they did blood tests and a ct scan and found out i have an ovarian tumor which they think is cancer and i probably will have to have a hysterectomy. And now im looking to start a lawsuit [Show More]I had mirena put in a year and a half ago after i had my son. I have been problems ever since. And my doctor just kept telling me it was normal so finally i just couldnt take it anymore and went to a hospital and they did blood tests and a ct scan and found out i have an ovarian tumor which they think is cancer and i probably will have to have a hysterectomy. And now im looking to start a lawsuit bc this is horrible. Do not get it. And something needs to be done. Bc women dont deserve to go through these horrible things.
CourtneyDecember 5, 2011 at 6:03 pm
I had the Mirena put in 9 months ago 6 weeks after my 2 child was born. MY sister had the Mirena and had said it was great for her and that it stops your periods and you don't have to worry about paying each month for bc pills. It sounded like a good option for me and I knew I didn't want anymore kids until 4 or 5 years down the road. So I talked to my doctor about it and she said that it was an e[Show More]I had the Mirena put in 9 months ago 6 weeks after my 2 child was born. MY sister had the Mirena and had said it was great for her and that it stops your periods and you don't have to worry about paying each month for bc pills. It sounded like a good option for me and I knew I didn't want anymore kids until 4 or 5 years down the road. So I talked to my doctor about it and she said that it was an excellent choice for me and that she had had it and then 5 years after she had it she got it removed and got pregnant right after. They handed me a pamphlet and told me to read through it. They didn't force me to read it or talk to me about all the side effects. They just here's a pamphlet. They put it in with a little bit of pain and I was in pain for about 2 months with it off and on but they said that was normal. I have not been able to lose my baby weight very fast and I exercise constantly. I have had awful acne, horrible headaches, bloating in my belly, really oily hair, and still have had a period every month since I got it put in and they last for about 5-6 days. About a month ago I felt sharp stabbing unbearable pains in my belly, lungs, ribs, back and pelvic area. The pain just got worse after day 3 and I decided to go to the OBGYN and see what was up. I had heard of IUD's being lost. I have no health insurance. I had a pregnancy test, infection test, ultrasound, and pelvic exam and they found nothing wrong. The pain got worse that day so I went to the ER because I thought it could be an appendicitis. I got blood work and a CAT scan, with NO insurance. They found nothing wrong with me. I finally realized that it was my Mirena causing me pain so I went in to have it removed and she couldn't get it out, the nurse practitioner couldn't find the strings and told me someone else would have to try to get it out n 2 weeks. So I waited for 2 weeks off and on in pain and I finally couldn't take the pain anymore so I went in again and after a lot of pain and time they were finally able to get the strings and pull it out. They got it out last week and I am starting to feel much better but am worried pains might come back with my next period or many periods to come. I am also worried that if we try again to have a baby we will have trouble. I never had troubles with my first 2. This device is horrible and I wish someone had talked to me face to face about all the risks and then I may have not go it put in. I will be paying hospital bills for the rest of my life due to this thing.
CherieDecember 4, 2011 at 2:07 am
Has anyone heard back from this web site? I posted months ago and have not heard anything. Why would they made this site for us to post on to try and get help for them not to do anything to help us. Whatever I guess they don't think we matter just like our doctors made us feel. Everyone is the same as long as it is only benefical to them they will help. I am sorry that we all had to go through suc[Show More]Has anyone heard back from this web site? I posted months ago and have not heard anything. Why would they made this site for us to post on to try and get help for them not to do anything to help us. Whatever I guess they don't think we matter just like our doctors made us feel. Everyone is the same as long as it is only benefical to them they will help. I am sorry that we all had to go through such a horrible ordeal and no one care. I pray for you all and hope none of you end up having to have a hystorectomy like me because of some stupid birth control.
lynNovember 23, 2011 at 7:27 pm
i had a iud in for years iv allso had lots of pain so i wonted it taken out so i went to my doctors to have the iud taken out but the string snaped off thay couldent take it out the doctor said i needed to go hospital to have it removed so i did thay put me to sleep and thay got the iud out they said i was better haveing a new iud put back in because of my heavy bleeding befor so i had a new on[Show More]i had a iud in for years iv allso had lots of pain so i wonted it taken out so i went to my doctors to have the iud taken out but the string snaped off thay couldent take it out the doctor said i needed to go hospital to have it removed so i did thay put me to sleep and thay got the iud out they said i was better haveing a new iud put back in because of my heavy bleeding befor so i had a new one put back in still haven pain so two on kepted on saying it was the iud thay would say no its not now iv found out i have blood in my urine had a camra in my bladder and found part of a iud or hol one so now iv got to wait to see whot there going to do now its been six weeks ago now and im still wating to here whot there going to do so please if any one gone thow the same could you please let me know has im going thow hell now o yes the new iud is still in the right place so it got to be the old iud
DawnNovember 20, 2011 at 1:53 am
I have had the Mirena for seven years and my body has gone to hell, I have High Insulin Restitant, pre-diabetic, chest pains, aniexty and panic attacks low blood sugar, passing out, I have always had a high metabolism and now that is gone and have been putting on weight. I have never had any problems and have never taking no medicine I am 34 years old. I was never told about these side effects tha[Show More]I have had the Mirena for seven years and my body has gone to hell, I have High Insulin Restitant, pre-diabetic, chest pains, aniexty and panic attacks low blood sugar, passing out, I have always had a high metabolism and now that is gone and have been putting on weight. I have never had any problems and have never taking no medicine I am 34 years old. I was never told about these side effects that this mirena was causing me, I just had it removed 3 weeks ago and I am so pissed that this IUD has caused this and now its going to take forever to get my body back to what it was 7 yrs ago. This company should take responsibilty at what the Mirena is causing in these women.
MariaNovember 20, 2011 at 12:32 am
Oh let's not forget the symptom my husband loves the most...NO SEX DRIVE. Zero. None. Its insane.
MariaNovember 20, 2011 at 12:24 am
February I gave birth to twins. I begged to have my tubes tied during the cesarean. Dr. Refused. She pushed and pushed Mirena oh don't tie ur tubes u will regret it Mirena is amazing and would be perfect for u. It was all BS! But I had it inserted inApril anyway. I have Hashimotos disease (hypothyroid), so when I started having all these absurd symptoms I attributed them all to my thyroid. I perso[Show More]February I gave birth to twins. I begged to have my tubes tied during the cesarean. Dr. Refused. She pushed and pushed Mirena oh don't tie ur tubes u will regret it Mirena is amazing and would be perfect for u. It was all BS! But I had it inserted inApril anyway. I have Hashimotos disease (hypothyroid), so when I started having all these absurd symptoms I attributed them all to my thyroid. I personally didn't have too much trouble with insertion. Was fine at the 2 week ultrasound apt and as far as I knew Mirena was all it was cracked up to be. Now its November and my thyroid is pretty much under control and my symptoms are worse. (Hair falling out, severe back ,shoulder, abdominal, and pelvic pain, weight gain, numbness/tingling in arms and hands, twitches in my fingers and legs, hot flashes, blurry vision, heart palpatations, the shakes, crying for the dumbest things, eposides of violent rage and anger, bloating, late/"reverse periods" now with the pain in my ovaries and the anger I felt towards my son today I decided to google "episodes of anger associated with Mirena IUD" and stunbled upon this site. Well I'm convinced. It has to be Mirena. I'm a person that usually doesn't trust what drs say and I research research but for whatever reason I trusted my ob and am now furious. With myself, with Bayer, and my drs office. My prayers are with everyone of you. I def know the feelings of being crazy on top of the constant physical pain. I'm getting this thing out of me asap!
AundriaNovember 16, 2011 at 9:45 pm
I got Mirena first in July of 2007. It came out in January 2008, so I had another one in. I still have it, but the strings are gone, and a catscan and xray show that they are in my uterus. I no longer have insurance so no one will remove this thing! I have pelvic pain constantly, and sometimes I have to be seen in the ER for it it gets so bad. I just want it out! If I could dilate my cervix I woul[Show More]I got Mirena first in July of 2007. It came out in January 2008, so I had another one in. I still have it, but the strings are gone, and a catscan and xray show that they are in my uterus. I no longer have insurance so no one will remove this thing! I have pelvic pain constantly, and sometimes I have to be seen in the ER for it it gets so bad. I just want it out! If I could dilate my cervix I would remove it myself!
ClaireNovember 16, 2011 at 12:41 am
AFter 4 months of having the Merina and suffering severe side effects of depression, mood swings, anxiety, weight gain, acne, dizziness, lethergy- I found these sites and put everything together and had it removed yesterday. I had been complaining to my doctor that I had experienced these things and he denied them having anything to do with the Merina. One day after its removal I am so relived th[Show More]AFter 4 months of having the Merina and suffering severe side effects of depression, mood swings, anxiety, weight gain, acne, dizziness, lethergy- I found these sites and put everything together and had it removed yesterday. I had been complaining to my doctor that I had experienced these things and he denied them having anything to do with the Merina. One day after its removal I am so relived this horrible thing is out of me. I was just about ready to beg for anti-depressant medication and see a therapist- I did not understand what was wrong with me. I ahve not been myself since I started taking this and I wish I realised it was not me, the stress of life or anything that was causing this than the damn Merina. I have reported this to the TGA. Please report your negative side effects of this drug - 1800 020 653. I have also made a complaint to Bayer, its manufactor. I encourage other women to do the same- only with formal reports can you make a difference. I am so angry and sad this has done this to me.
KIMMENovember 15, 2011 at 12:28 am
I had my Mirena placed in Nov 2008 and just had it removed today 11-14-11. I experienced the heavy bleeding for months after it was placed (3 or 4 months) and was told by the doctor and nurses that it was normal. I have read hundreds of women who have had horrible out comes from this devil IUD....Two weeks before I had it removed I have had heavy cramps with no bleeding, headaches, acne, mood swin[Show More]I had my Mirena placed in Nov 2008 and just had it removed today 11-14-11. I experienced the heavy bleeding for months after it was placed (3 or 4 months) and was told by the doctor and nurses that it was normal. I have read hundreds of women who have had horrible out comes from this devil IUD....Two weeks before I had it removed I have had heavy cramps with no bleeding, headaches, acne, mood swings, back pain, sore breast, dizzy, lightheaded, nausea, weight gain 40lbs, swelling in my joints, symptoms of HBP. My husband and I are going to me trying for another baby soon so I hope this hasn't messed anything up for us. Is there a time limit on this law suite? Being that I just had mine removed and have yet to see any side effects yet beyond what I have listed above. Please contact me with this information as soon as possible.
AshleaNovember 14, 2011 at 10:47 pm
I am 22 years old. I had the Mirena placed in Oct 2008 at my 6 week check up after having my son(which only took me one month to conceive). I had non stop bleeding, cramping, back pain and terrible pain during intercourse and bleeding after intercourse. It hurt to walk, change my sons diaper or even bend over to pick something up off the floor. I had it removed in Jan of 2010 because I was tired o[Show More]I am 22 years old. I had the Mirena placed in Oct 2008 at my 6 week check up after having my son(which only took me one month to conceive). I had non stop bleeding, cramping, back pain and terrible pain during intercourse and bleeding after intercourse. It hurt to walk, change my sons diaper or even bend over to pick something up off the floor. I had it removed in Jan of 2010 because I was tired of the pain it was causing me and wished to conceive a second child. I still have not become pregnant and suffer from ovarian cysts every month. I do believe Mirena has made me infertile. In March of this year I went to my OBGYN to try and figure out why I hadn't became pregnant yet; he had bloodwork ran on me and it showed that I am not ovulating because my progesterone levels are too low. My doctor then put me on Clomid for a month which made my progesterone levels drop even lower. We did not continue with the infertility treatments because of the costs so we are continuing to try naturally with no infertility drugs, but it doesn't seem to be working. I would give anything for just one more child. I feel that the Mirena has robbed me of that privilege.
bethNovember 13, 2011 at 1:36 am
I had the IUD After i had my first son and I had heavy bleeding and cramping so I got the IUD removed about 3 Years later i had my son and I went through so my birth control and my doctor said i should try the IUD one more time becasue the IUD is a great birth control so i got it after the first 3 days the IUD had fell out and i was in alot of pain the T part had turn a whole 360 so after i went t[Show More]I had the IUD After i had my first son and I had heavy bleeding and cramping so I got the IUD removed about 3 Years later i had my son and I went through so my birth control and my doctor said i should try the IUD one more time becasue the IUD is a great birth control so i got it after the first 3 days the IUD had fell out and i was in alot of pain the T part had turn a whole 360 so after i went to the doctor and she pulled it out.So i was spoting here and there for about 14 weeks the first 3 weeks i was bleeding and cramping and each week after the pain go worst. So i went to the doctor because i was so weak and was not my self the doctor told me well your pregnant i said what how can i be pregnant if i been bleeding she said the baby fine after she took test then i was still not feeling well and about a week later my baby was growing in my left tube and due to being 14 weeks my tube was not able to save. so i got pregnant 3 times after that and i had 3 more Ectopic Pregnancy in my right tube i think when the IUD had fell out had damage my tube is there any one out there that went through the same situation please commet
latashaNovember 12, 2011 at 2:15 am
i had the iud for four years. and it have been giving me problems like dischargin and having ordor behind it and i tried to get it out for three years and they wouldnt get it out for all that time
AmberleyNovember 11, 2011 at 3:01 pm
I had the IUD put in after my second child, after only 6 months I had to go to a different Dr to have it removed but the Dr who put it in refused to take it out after only that short a time. I had to have it removed because I had literally been on my period for 6 solid months. It took my husband and I 3 years to get pregnant again, and we have decided that we wanted to expand our family and try on[Show More]I had the IUD put in after my second child, after only 6 months I had to go to a different Dr to have it removed but the Dr who put it in refused to take it out after only that short a time. I had to have it removed because I had literally been on my period for 6 solid months. It took my husband and I 3 years to get pregnant again, and we have decided that we wanted to expand our family and try one more time for a little girl, after almost a year and a half of trying I went to my Dr to find out what the heck was going on, only to find out that I have polycycstic ovaries and now have to take fertility drugs to try to temporarily fix this problem, if I am able to get pregnant again it will be my last because my Dr will be doing a hestorectomy afterwards so I will no longer be in so much pain from the cysts bursting. I pretty much live in pain everyday and it does alot more than just irratate me that it could have been avoided if my Dr at the time had not talked me into getting this form of birth control.
JadeNovember 10, 2011 at 11:46 pm
I had my mirena put in 6 weeks after my 3 year old was born in 08. It was very painful to insert. I bleed for six months after with only a couple days rest in between. I gained weight I couldn't lose. In dec, of 10, I went to have it removed and they could not find it. So the next day I had an pelvic ultrasound and it was not there. They finally found it by my right kidney. On January 5, 20[Show More]I had my mirena put in 6 weeks after my 3 year old was born in 08. It was very painful to insert. I bleed for six months after with only a couple days rest in between. I gained weight I couldn't lose. In dec, of 10, I went to have it removed and they could not find it. So the next day I had an pelvic ultrasound and it was not there. They finally found it by my right kidney. On January 5, 2011 I had to have surgery to get it removed. It was very painful. Would not recommend it to anyone!!!
JessicaNovember 6, 2011 at 11:23 pm
I got my iud back in sept 2008. All its been is trouble ever since. I have bad cramps all the time sex is painful, I dont have the urge for sex anymore. When I do have sex I get bad cramps, one time it hurt so bad I was curled over for hours, almost went to the er. I have had bad mood swings and just havent been the same since I got this horrible device. My obgyn is the same, gave me no warnings a[Show More]I got my iud back in sept 2008. All its been is trouble ever since. I have bad cramps all the time sex is painful, I dont have the urge for sex anymore. When I do have sex I get bad cramps, one time it hurt so bad I was curled over for hours, almost went to the er. I have had bad mood swings and just havent been the same since I got this horrible device. My obgyn is the same, gave me no warnings about side effects and made it sound like it was the best thing. Its nice to know that there are others. I also have had quite a few yeast infections from this.
stephanieNovember 5, 2011 at 2:55 pm
First i started having sharp pains after two and of half years. My ob did ultrasound said i have small fibroids.Doctor said Not to worry as of now This spring finally started taking meds for severe mood swing. Still having them..Lately i been feeling as if my iud has shifted. Well last night things changed went from a scratchy feeling to my what i think is my uterus falling through my vagina wa[Show More]First i started having sharp pains after two and of half years. My ob did ultrasound said i have small fibroids.Doctor said Not to worry as of now This spring finally started taking meds for severe mood swing. Still having them..Lately i been feeling as if my iud has shifted. Well last night things changed went from a scratchy feeling to my what i think is my uterus falling through my vagina wall. Its the weekend so will be taking it easy til i can get to the doctor. Thought mirena was the best thing for.me after four years may turn out to be the worst thing ever. Will repost after results.
StefanieOctober 31, 2011 at 3:12 am
I have had my mirena for 3 yrs. It was extremely painful going in and my dr...like the otthers did not inform me of all of the side effects but made it sound like the greatest birth control in the world. At my 1st annual after having it my dr informed me that she could not find the strings. She said this was not a big deal sometimes they curl up into your cervix but sent me for an ultrasound and x[Show More]I have had my mirena for 3 yrs. It was extremely painful going in and my dr...like the otthers did not inform me of all of the side effects but made it sound like the greatest birth control in the world. At my 1st annual after having it my dr informed me that she could not find the strings. She said this was not a big deal sometimes they curl up into your cervix but sent me for an ultrasound and xray..which my insurance did not cover...just to tell me that it was in the right place and I should leave it in (it may take a scope and anesthesia to get it out). I told my dr about severe pains I had after having sex with my husband that had me doubled over. She stated that it may have been an ovarian cyst that ruptured...as if that was a common thing to not be concerned about. I have recently been having cramps(feel like contractions) abdominal bloating and depression and mood swings. When I called my dr to have it removed the nurse was rude to me and called me back saying that the dr refused to take it out and that i have to wait the full 5 years. I am going to schedule with a new dr..even tho I've been with the same 1 for 10 yrs. I am terrified of what's to come.the pain of removal, possibly having it surgically removed(I have NEVER had surgery in my life) and not to mention infertility or worse a hysterectomy...I am only 28 yrs old, mother of 2 girls and only been married for 4 yrs n would like to try again...as soon as this lawsuit is underway please post!!
StephanieOctober 29, 2011 at 2:31 pm
I have been researching this as well and was shocked to find I am not the only one out there having these "unexplainable" side effects. I also found a website where literally like 100 women were saying they just removed it themselves. I understand the risks of that (unlike when I had it inserted), but I think if I'm careful and listen to my body while trying self removal, all will be okay. I pl[Show More]I have been researching this as well and was shocked to find I am not the only one out there having these "unexplainable" side effects. I also found a website where literally like 100 women were saying they just removed it themselves. I understand the risks of that (unlike when I had it inserted), but I think if I'm careful and listen to my body while trying self removal, all will be okay. I plan on trying it tomorrow. Here's the link: http://community.babycenter.com/post/a5600795/self_removal
NancyOctober 24, 2011 at 7:32 am
I had my Mirena put in January 2010 by the nurse practitioner at my ob office. It was very unconfortable procedure not to mention I was always bleeding I was advised that this would control my irregular bleeding but, this never go under control I called my dr office and they would perscribe me birth control pills. I finally got sick ofthe dr makking me feel like I was over reacting that I went to[Show More]I had my Mirena put in January 2010 by the nurse practitioner at my ob office. It was very unconfortable procedure not to mention I was always bleeding I was advised that this would control my irregular bleeding but, this never go under control I called my dr office and they would perscribe me birth control pills. I finally got sick ofthe dr makking me feel like I was over reacting that I went to another dr and I leared that My mirena was out of my uterus and in my ilium per a xray as we were unable to locate with a ultrasound. I went in to have it removed n friday which was a week after the x ray and the dr had to get another xray due to the fact that it had traveled to my Pancreas, not to mention that my uterus is split open from where I believe the iud was pushed through by the Nurse who incerted it. I am not sure if I can have any other children at this point
LeighOctober 19, 2011 at 2:25 am
Just wanted to pass along my Mirena story...Summer of 2008 I had my mirena put in. In April of 2009 found out I was pregnant and doc did an ultrasound which found no IUD in my uterus. She said it must have fell out. Never thought anything of it. After the birth of my daughter in the winter of 2010 had the Paraguard put in as I didn't care for the Mirena. Well last week I start feeling somethi[Show More]Just wanted to pass along my Mirena story...Summer of 2008 I had my mirena put in. In April of 2009 found out I was pregnant and doc did an ultrasound which found no IUD in my uterus. She said it must have fell out. Never thought anything of it. After the birth of my daughter in the winter of 2010 had the Paraguard put in as I didn't care for the Mirena. Well last week I start feeling something poking, thinking my paraguard shifted position. Had an x-ray yesterday to have the guy say to me you have 2 IUD's in? Low and behold my mirena has been "hanging" out all this time. Never had a problem or even felt it till now. Ugg, surgery is on the horizon for me :(.
AlesiaOctober 18, 2011 at 8:24 pm
I got the Mirena in 2008 after I had my first child and it was the most painful thing I have ever encountered. The doctor said I would have cramps for about twenty minutes, boy was he wrong. I had a menstrual cycle every other week or two weeks. I know why you can't get pregnant...because all I did was bleed. The doctor gave me medicine to stop the bleeding, that worked, but when I used the 30 day[Show More]I got the Mirena in 2008 after I had my first child and it was the most painful thing I have ever encountered. The doctor said I would have cramps for about twenty minutes, boy was he wrong. I had a menstrual cycle every other week or two weeks. I know why you can't get pregnant...because all I did was bleed. The doctor gave me medicine to stop the bleeding, that worked, but when I used the 30 day prescription the bleeding went right back. I always cramped and felt exhausted, but I guess when you are loosing that much blood you will feel tired. I had it removed in May of 2011 and I got a call from the doctor's office that I had gotten the Pelvic Inflammatory Disease and had to take a round of strong antibiotics. It took me three months to start a cycle after having it removed. If I had known this was going to be the result I would have never gotten the Mirena. I am now taking Yaz and just saw what was happening with it...I can't win for loosing with birth control!!
thereseOctober 17, 2011 at 10:35 pm
I also had to have a hysterectomy to remove the Mirena IUD.
ThereseOctober 17, 2011 at 10:33 pm
About 6 years ago, I also had to have a hysterectomy to take out the Mirena IUD. INo strings were visible and the only way to take it out was with a hysterectomy. Unbelievable!!!
RegeneaOctober 17, 2011 at 2:27 am
I had the Mirena put in Sept. 2009, my Dr said it was my last option before hysterectomy. I didnt have any insurance so my Dr. "donated" the mirena to me. The insertion itself was pure agony, after about 20 mins, plus 30 mins rest time I was sent home. Since then I have been in constant lower abdominal pain, lower back pain that is almost more than I can bear, headaches, hair loss and uncontrolabl[Show More]I had the Mirena put in Sept. 2009, my Dr said it was my last option before hysterectomy. I didnt have any insurance so my Dr. "donated" the mirena to me. The insertion itself was pure agony, after about 20 mins, plus 30 mins rest time I was sent home. Since then I have been in constant lower abdominal pain, lower back pain that is almost more than I can bear, headaches, hair loss and uncontrolable mood swings. I bleed for the first 6 or 7 months and then didnt have any periods until Oct 2011, and it was worse than before I had this horrible device put in. I have been to the ER several times and am dismissed with "unexplained" stomach pains. Had I known I would be going thru all this I would never had let my Dr talk me into it.
VanessaOctober 12, 2011 at 10:40 am
Wow!! After reading all the comments I'm really starting to freak out...I got my mirena placed in Jan. 2010 2 months after my daughter was born...thought it was the best thing ever I mean I got my periods regularly for about 3 months till it finally went away some spotting every now and then but I never had any side affects until now...it's been in me for over a year and it started about a month a[Show More]Wow!! After reading all the comments I'm really starting to freak out...I got my mirena placed in Jan. 2010 2 months after my daughter was born...thought it was the best thing ever I mean I got my periods regularly for about 3 months till it finally went away some spotting every now and then but I never had any side affects until now...it's been in me for over a year and it started about a month ago I get shaky, I haven't been sleeping, I'm always hungry I've gained about 10 lbs, I feel bloated, tender breasts, almost like if I'm pregnant again I'm starting to really worry I was told I have a tilted uterus so I figured that's why i could never really feel the strings...I'm really worried now because I am getting to the point were I look, act, and feel like I'm pregnant i feel like I'm going crazy like I'm just make believing all these things idk what to do anymore I need help...
jessicaOctober 11, 2011 at 3:48 pm
Got mine removed,had to use a camera to find it and was put on some good meds had a shot to numb the cervix,felt better 4days after and after 2 weeks I was almost back to normal...now at 3 1/2 weeks I'm great!Posted back on Sept.10,2011....It is not a good choice for all of us,my problem was how it had my hormones all out of whack.I guess some women need more and some need less, I needed less.So [Show More]Got mine removed,had to use a camera to find it and was put on some good meds had a shot to numb the cervix,felt better 4days after and after 2 weeks I was almost back to normal...now at 3 1/2 weeks I'm great!Posted back on Sept.10,2011....It is not a good choice for all of us,my problem was how it had my hormones all out of whack.I guess some women need more and some need less, I needed less.So glad it is gone along with all my problems!My husband even saw the difference in me after.Oh and another thing I lost 8 lbs with no change in my diet.I was not out of my mind after all!Best wishes girls.
CarrieOctober 10, 2011 at 3:12 am
OMG!! I've had the same thing going on and have been to my OBGYN, FAMILY, GASTROINTEROLOGIST, and the ER doctors..I was told it wasn't my MIRENA!! My OBGYN office was the first place I went. They are now sending me to Charlottesville in December. I'm having all the same symptoms as everyone else! It's very painful and I'm about to go crazy..I feel like I'm not getting anywhere and I've been in pai[Show More]OMG!! I've had the same thing going on and have been to my OBGYN, FAMILY, GASTROINTEROLOGIST, and the ER doctors..I was told it wasn't my MIRENA!! My OBGYN office was the first place I went. They are now sending me to Charlottesville in December. I'm having all the same symptoms as everyone else! It's very painful and I'm about to go crazy..I feel like I'm not getting anywhere and I've been in pain for far too long. Medical bills are piled up sky high and I'm a single mom so I'm working and feel like Hell! It's been a NIGHTMARE! And December is too long to have to wait..it's been almost a year! They keep trying to give me all these pills that don't work. I DON'T want pills that mask my symptoms..I WANT them FIXED and GONE!! I will be in my OBGYN's office ASAP and I'm telling him I want it removed NOW! WTF!! THANK YOU ladies for your post!! Has anyone gotten a Lawyer yet?
MelissaOctober 9, 2011 at 12:52 pm
I have had the Mirena for over a Year and have never stopped bleeding, I actually bleed about every 10 days now, have horrible PMS, no sex drive, suffer from depression and worst of all I have developed migraine headaches that I am Having to see a neurologist for now. I am seeing a new Gyn to see about getting it taken out now. It has been the worst thing I have ever been through!!
cherieOctober 8, 2011 at 3:07 am
Has anyone heard anything about this. Or have any of you found help?
KimberlyOctober 5, 2011 at 7:47 pm
I had the Mirena inserted in March, 2008. When I went for my follow up visit shortly after, I was told that the device had moved and that the strings were no longer visible. I was sent for an ultrasound which found the Mirena was still in my uterus and not to worry - but that I would need surgery to have it removed when I was ready. On Monday I spent the entire day in the ER with MS-like sympto[Show More]I had the Mirena inserted in March, 2008. When I went for my follow up visit shortly after, I was told that the device had moved and that the strings were no longer visible. I was sent for an ultrasound which found the Mirena was still in my uterus and not to worry - but that I would need surgery to have it removed when I was ready. On Monday I spent the entire day in the ER with MS-like symptoms. Numbness and tingling on my entire right side and face, headaches, etc. A CT Scan and MRI as well as all bloodwork came back normal. I googled my symptoms and found literally hundreds of other women with my same symptoms. I immediately called my Gynecologist and have scheduled the Mirena to be removed via hysteroscopy next week. I will also be requesting a blood test for Silicone poisoning. I can not wait to get this device out of me and hopefully have my symptoms relieved.
BonnieSeptember 25, 2011 at 2:18 pm
I had the mirena fitted in January last year, I suffered the usual pain after having this fitted and continued to have pain. I became pregnant in August 2011 with the mirena still fitted, only to find out that the coil was now on top of my bowel. I have had to undergo a termination as my family is complete, following this I had a mini stroke and was in hospital, it could have been related altho[Show More]I had the mirena fitted in January last year, I suffered the usual pain after having this fitted and continued to have pain. I became pregnant in August 2011 with the mirena still fitted, only to find out that the coil was now on top of my bowel. I have had to undergo a termination as my family is complete, following this I had a mini stroke and was in hospital, it could have been related although i don't know for sure. I am now scheduled in to have more surgery to remove the coil. The consultant who saw me has advised that he believes the coil was never in my uterus but had been pushed through the wall of my womb at the time of fitting! Not only that but because the hospital would not xray me while pregnant to see if the coil was still in place I was advised to use another form of birth control after the termination, I therefore had the contraception injection, so I now have two lots of contraception in my system which is causing tiredness, spots, and the most horrendous night sweats meaning that I get little or no sleep whatsover.
KelseySeptember 25, 2011 at 1:50 am
I had the Mirena inserted after my second child, November 2006. This November would be my 5 year mark, and it has been amazing. I had a little discomfort after inserting it, but after that I didn't have too many side effects- I didn't gain weight like I did with other birth controls, my periods lightened up to 4 times a year, and I didn't have to do anything with it, never felt it- it was easy! T[Show More]I had the Mirena inserted after my second child, November 2006. This November would be my 5 year mark, and it has been amazing. I had a little discomfort after inserting it, but after that I didn't have too many side effects- I didn't gain weight like I did with other birth controls, my periods lightened up to 4 times a year, and I didn't have to do anything with it, never felt it- it was easy! Then, last week I started have horrible pains in my pelvic area, and when I went to the hospital they told me I was pregnant. So I knew it was most likely a tubal pregnancy, and I had to get emergency surgery. They thought that the IUD may have punctured my uterus too because there was a lot of fluid in my abdomen. But after surgery found out it was an ectopic pregnancy up near my ovary. They said I was about 1 1/2 months pregnant, the placenta started forming & since the lining is so thin in the fallopian tube, it ruptured. I had 1/2 gallon of blood in my abdomen. So they removed my IUD (which was still in place & my uterus was fine, no puncture or anything), and they also had to remove my fallopian tube as well. I always loved the IUD, and Im considering getting it again, but Im a little scared. I mean it worked great for 4 years and 10 months, right?! The Doc just said that I would need to get it taken out at 3 years instead of 5 cause apparently our eggs/sperm is too strong for the lower hormone levels. Last year I asked my OB if I could take it out a year early because we are super fertile, and if there is a higher chance of getting pregnant in the last year, I WOULD. But she told me it was safe for 7 years, but 5 years is the limit. So that was just great. I'm a little scarred to get it again, but I don't trust any other form of birth control...
sherrySeptember 22, 2011 at 3:45 pm
I had the mirena put in in April 2011because of serious bleeding and cramping just one month later i was rushed to the hospital because i couldn't breath and my heart was racing i had a pulmonary embolism. the funny thing about that was the blood work said there was low lickly hood of a blood clot because the numbers were to low but sure enough with a cat scan i had one and spent several days in[Show More]I had the mirena put in in April 2011because of serious bleeding and cramping just one month later i was rushed to the hospital because i couldn't breath and my heart was racing i had a pulmonary embolism. the funny thing about that was the blood work said there was low lickly hood of a blood clot because the numbers were to low but sure enough with a cat scan i had one and spent several days in the hospital. my primary doctor said we should have it taken out but the ob/gyn refuses to take it out swearing the mirena isn't the problem. i still have it in and and i am still bleeding from it being placed. the ob says it is because i am on blood thinners and still refuses to take it out. i just don't know what to do.
EmilySeptember 19, 2011 at 8:57 am
When I first heard about Mirena, and did my "research" (which consisted of reading the brochure and going to the Mirena website), I thought "what a dream come true! A birth control with the least amount of side effects and you don't even have to think about it! Yay!" ...Little did I know it was going to be one of the worst decisions of my life. Since I have had it inserted in December of 2010, I h[Show More]When I first heard about Mirena, and did my "research" (which consisted of reading the brochure and going to the Mirena website), I thought "what a dream come true! A birth control with the least amount of side effects and you don't even have to think about it! Yay!" ...Little did I know it was going to be one of the worst decisions of my life. Since I have had it inserted in December of 2010, I have had: constant panic attacks, terrible depression, I'm always in a daze with my head in the clouds, I feel like I am going crazy! A lot of disassociation is brought on with this, and it is driving me nuts. My temper is out of control, I have numbness in all my limbs and face, debilitating headaches and migraines, blurred vision and spots in my eyes, heart palpitations, pain in my lower back and abdomen, no bleeding (but that is due to the fact that I am still breastfeeding, which I pray has no direct impact on my son...if it did, I would probably kill somebody...) EXTREME exhaustion coupled with over-sleep during the day and insomnia due to paranoia at night, ....basically everything terrible that everyone has been talking about. I am disgusted and hurt, and getting this horrific thing out of me as soon as possible. I am terrified now knowing what can happen while getting it removed and after removal, and I am so angry at myself for ever having made this stupid decision. I am scared I will not be able to have anymore children, or I will end up getting a terrible infection that can lead to things such as type 1 diabetes, or constant yeast infections. I don't want a hysterectomy.........I don't want anything! I'm so scared!! I hope it all goes well...but after reading everything, I doubt it will. This is a terrible device, and I am amazed that doctor's are still offering it. If this causes irreversible damage, you better believe I will do everything in my power to get Bayer to fix it. I can't believe this. I am disgusted, and really, really hurt :( I feel like I could cry, which wouldn't be a surprise since I burst into tears randomly anyways. Ugh. I was not well informed on this decision, only the positives. And the brochure made it sound like nothing could go wrong at all, or if it did, it was a very slim chance, and it was nothing about these terrible side effects. I feel deceived. I hope getting it removed will end this nightmare....if not....then I don't know what I'll do. I just want this to be over :(
KaraSeptember 18, 2011 at 7:17 pm
I am 22 and had my 1st child in November 2009. In Jan. 2010 I had mirena inserted. Four months later I ended up pregnant and my OB couldn't find the IUD and still to this day I have pelvic pain, pain during sex, hip and back pain. In the progress of trying to get the doctors to help me find out what is causing the pain.
amberSeptember 16, 2011 at 6:50 pm
I am 24 yrs old I had the iud placed in 2008 six weeks after my son was born. It has been three in a half years and I feel like crap. It hurt so bad getting it out in I almost passed out. I have had the same symptoms as everyone else. I tried sep 12 2011 to get the iud removed the doctor attempted three times and with that a vaginal ultrasound to make sure it was still in place. It was so painful [Show More]I am 24 yrs old I had the iud placed in 2008 six weeks after my son was born. It has been three in a half years and I feel like crap. It hurt so bad getting it out in I almost passed out. I have had the same symptoms as everyone else. I tried sep 12 2011 to get the iud removed the doctor attempted three times and with that a vaginal ultrasound to make sure it was still in place. It was so painful trying to remove it I could not take it any more. Well from what he said the "strings that are suppose to hang down has wrapped their self around the iud." But I talked to a nurse and she said they r not strings its little metal wire that hangs from the end of it. So there for Monday Sep 19 I have to have it surgically removed. So if I continue with problems headaches, hot flashes, pelvic pain and bleeding I'm going to try hard as I can to press for a law suit, I want another baby and terrified after reading it may damage me fertility wise!!! And i have not stopped bleeding since he attempted to take it out.
KatherineSeptember 16, 2011 at 2:24 am
I had the Mirena IUD inserted in June 2011. It was extremely painful and I bled for six weeks straight afterwards. It was at that point I became extremely depressed one morning to the point of thoughts of suicide. I figured I would give it at least six months to level off but after repeated bouts of depression, back pain, lack of a sex drive, foul smelling discharge, headaches, nausea, chest pain,[Show More]I had the Mirena IUD inserted in June 2011. It was extremely painful and I bled for six weeks straight afterwards. It was at that point I became extremely depressed one morning to the point of thoughts of suicide. I figured I would give it at least six months to level off but after repeated bouts of depression, back pain, lack of a sex drive, foul smelling discharge, headaches, nausea, chest pain, sharp pain in my left breast, a constant need to urinate, anxiety attacks coming in waves (I am becoming increasingly dependent on my Xanax which is something in five years I have NEVER done), insomnia, dizziness and a host of other problems I called my doctor yesterday (09/14/2011) and am scheduled to have it removed next Wednesday. While my doctor covered the basics I don't feel I was given as much information as I should have been to make an informed decision. How can I make an informed decision regarding my health with only half the truth? The last four months have been a nightmare and I am just ready for it to be over with. KDH
RosemarySeptember 15, 2011 at 4:45 am
! had the Iud mirena inplanted in January 2009 and got it removed in March of this year.I never thought my stomach problems, headaches,no interest in sex and skin discoloration would have been caused by the Iud. After I had it removed my stomach was still hurting. They did a ultra sound and I found out I have veins around my uterus.They wanted to wait to see if I started producing estrogen again b[Show More]! had the Iud mirena inplanted in January 2009 and got it removed in March of this year.I never thought my stomach problems, headaches,no interest in sex and skin discoloration would have been caused by the Iud. After I had it removed my stomach was still hurting. They did a ultra sound and I found out I have veins around my uterus.They wanted to wait to see if I started producing estrogen again because it was a major surgery if not. Well it's getting worse and I can't deal with the pain any more so I have a appointment with the obgyn for a consult on Tuesday. I don't think this should have even been but on the market with all the horrible side effects it has. I know everything has side effects but if I knew all of them I would have thought twice about getting it.
SamanthaSeptember 13, 2011 at 6:57 pm
I am 22 year old female i had my IUD Mirena inplanted in 2008 after the birth of my second child with in 14 month time period. with in 2 weeks after the iud was inplanted the pains n cramping never went away. I always have pain in my lower abdominal area. keep getting infections in the uterus all the doctor will do is treat me for the infections. Then was told i have ovarian cyst which started abo[Show More]I am 22 year old female i had my IUD Mirena inplanted in 2008 after the birth of my second child with in 14 month time period. with in 2 weeks after the iud was inplanted the pains n cramping never went away. I always have pain in my lower abdominal area. keep getting infections in the uterus all the doctor will do is treat me for the infections. Then was told i have ovarian cyst which started about 3 years ago they come n go doctor hasn’t dont anything about it. with in 6- 1year after having the IUD when i first was told i had a ovarian cysic the ER told me i had PID which i never had before having the IUD. i know all these are side effects but it is making me lose my mind with pain all the time cant have intercourse with out wanting to scream out in pain. was told my uterus has dropped doctor dont want to do crap about it. I asked to have the IUD removed n to be honest he acts like he really dont want to do anything about it. about every two months have vagianl discharge from having the infection in my plevic or uterus area. come on now whats is really going on with this IUD. Another thing is my other half claims he can feel it when we can have intercourse and the doctors claims that it is inserted fine has’t not moved n has cut the string short as it can go. in my opinion i think there can be something done.. anyone else having issuse with this?
JessicaSeptember 10, 2011 at 2:30 pm
So many problems,it would be just a repeat of most of what you have read on this site.This thing should be taken off the market.If all the side effects had been given to me back in 2008 I would have had to have been out of my mind to get it,but I was told and given info that it was perfect for me.The one concern they had was that putting it in she could poke through my uteris but that was rare an[Show More]So many problems,it would be just a repeat of most of what you have read on this site.This thing should be taken off the market.If all the side effects had been given to me back in 2008 I would have had to have been out of my mind to get it,but I was told and given info that it was perfect for me.The one concern they had was that putting it in she could poke through my uteris but that was rare and she would know instantly.She said all went well so I was not worried.Now it is 2011 and I am sick of all the problems I am having.I went to have it removed and they were unable to get it,they have now set me up with an entirely different doctors office to see if they have better luck, with all of my (rare) side effects it would be best to have it removed now.I do not think this device is a healthy choice for anyone. I am 33 and feel like I am in full blown menapause and about 43 or older.I pray my life gets back to normal when they get this thing out of me!
SLWSeptember 9, 2011 at 8:59 pm
I had my Mirena inserted two months after my pregnancy in 2006. In the summer of 2010 a dermoid ovarian cyst developed on my right ovary. My doctor had to remove my ovary but I still have my left ovary. Here it is a year later and I'm having the same exact symptoms a little worse this time on my left side. I am scared to death that I may lose my left ovary and won't be able to have anymore childre[Show More]I had my Mirena inserted two months after my pregnancy in 2006. In the summer of 2010 a dermoid ovarian cyst developed on my right ovary. My doctor had to remove my ovary but I still have my left ovary. Here it is a year later and I'm having the same exact symptoms a little worse this time on my left side. I am scared to death that I may lose my left ovary and won't be able to have anymore children. While I thank God for my beautiful little girl it breaks my heart to think I can't have at least one more child. I have an appt set up with my OBGyn. I see that Mirena says the ovarian cyst are a side affect but "they go away". They left out the part about they go away with surgery.
JanettaSeptember 9, 2011 at 3:12 am
i had the mirena inserted in Jan of 2008 6 weeks after having my 2nd son. my doctor said it would be best for me, the first month was hell! i wouldn't stop bleeding 10 super pads full in an hour and it wouldn't stop. i contacted my obgyn and asked him o remove it and he decided to put me on yaz birth control pills as well as the mirena. i took the pills at the same exact time everyday and it stopp[Show More]i had the mirena inserted in Jan of 2008 6 weeks after having my 2nd son. my doctor said it would be best for me, the first month was hell! i wouldn't stop bleeding 10 super pads full in an hour and it wouldn't stop. i contacted my obgyn and asked him o remove it and he decided to put me on yaz birth control pills as well as the mirena. i took the pills at the same exact time everyday and it stopped the bleeding but if i missed a pill be an hour i would instantly start bleeding again. after that month i asked my doctor to take it out again and he told me to give it another month so i did. it had been 2 months and if i didn't take the pill exactly at the same time it wouldn't stop. finally by the end of the 2nd month i made him take it out i couldn't handle it anymore. its been over 3 years that I've been trying to have another baby and have yet to conceive. i am 23 years old and i can't have anymore babies,. IT RUINED MY LIFE!!! i would NEVER in a million years allow a doctor to talk me into getting in again!
lizSeptember 9, 2011 at 2:39 am
I have had the mirena for 3 years. I just went to my gyno today to have the normal yearly pap and to have the mirena taken out. I had to go thru two different doctors and they had to put their tools in and poke around two different times to only find that they are unable to take it out due to the strings on the mirena going up into my cervix. now apparently since i didnt have either one of my chil[Show More]I have had the mirena for 3 years. I just went to my gyno today to have the normal yearly pap and to have the mirena taken out. I had to go thru two different doctors and they had to put their tools in and poke around two different times to only find that they are unable to take it out due to the strings on the mirena going up into my cervix. now apparently since i didnt have either one of my children vaginally my cervix is too small for their tools to go up into my cervix to try to fish this thing out. I am currently waiting for my doc to call me back so that we can go to the operating room to have them surgically remove this. . . now i wonder what wouldve happened had I waited to remove this "wonderful contraceptive" until my 5 wonderful years had been up. Also my sister who just had her 2nd child naturally had her mirena put in 8 weeks after her son had been born. It had been put in on Thursday by Sunday night she was going into the er with intense stomach cramps. They couldnt figure out what had been going on so they sent her home with antibiotics. The pains continues. Eventually she complained to her doc enough about the pains so he had her go to the hospital. They put air into her stomach so that they could find out what was going on;. She ended up needed emergency operations...thanks to the mirena it put a hole into her fallopian tube which was causing internal bleeding, cysts on her cervix which irritated her appendix, so the appendix had to be removed among other issues that had happened all thanks to this great little miracle device and for having it for less than a month. This device needs to be taken off the market and if there are any lawsuits against the manufacturers of this device please contact me because we would love to join that.
jenniferAugust 31, 2011 at 4:56 am
Im a mother of 2 and when i heard about the yasmin recalls and i went in to see the dr about an alternative to yasmin first thing he jumped on was the iud aka mirena and i told him no way in h*ll will i get one not after the research that i did into bc before i went in. Also a friend of mine had mirena and it killed her twin babys because she got pregnant with it in and i had another friend who ha[Show More]Im a mother of 2 and when i heard about the yasmin recalls and i went in to see the dr about an alternative to yasmin first thing he jumped on was the iud aka mirena and i told him no way in h*ll will i get one not after the research that i did into bc before i went in. Also a friend of mine had mirena and it killed her twin babys because she got pregnant with it in and i had another friend who had it and it left her so scared inside they said she could never carry again. She did have a miscarrage but awhile later she did succesfully have a little boy. Now that i did some more reading i am glad i didnt get it. For furture reference do some research before u make such a big choice it could save u alot of trouble.
charleneAugust 29, 2011 at 1:16 pm
I was on the mirena for over 3yrs. I gained over 30lbs and became very sensitive. I asked my doctor to remove it several times & she always had an excuse even blaming weigh gain on my thyroids which I have no problem with. Finally, I demanded it be removed so I was told my insurance may not pay for it. I gave them days to respond then I call doc office & said I had contacted my insurance s[Show More]I was on the mirena for over 3yrs. I gained over 30lbs and became very sensitive. I asked my doctor to remove it several times & she always had an excuse even blaming weigh gain on my thyroids which I have no problem with. Finally, I demanded it be removed so I was told my insurance may not pay for it. I gave them days to respond then I call doc office & said I had contacted my insurance so they removed it. She begin to tell me how the mirena & depo can make you gain weight & suggest a diet plan. They dont care about your health just making money. This weight is making me miserable.
JeneAugust 20, 2011 at 8:29 pm
In June of 2006 I had the mirena put in 8 weeks after my daughter was born, my doctor helped convinced me this was the birthcontrol for me due to the fact that I was young and was not at all ready for another baby anytime soon. Three and a half years later, my husband and I were finacially ready to have another baby so we decided to have the mirena taken out, here I am 2 years later and no baby. I[Show More]In June of 2006 I had the mirena put in 8 weeks after my daughter was born, my doctor helped convinced me this was the birthcontrol for me due to the fact that I was young and was not at all ready for another baby anytime soon. Three and a half years later, my husband and I were finacially ready to have another baby so we decided to have the mirena taken out, here I am 2 years later and no baby. I have started going to the doctor for fertility testing and so on. I am terrified they are gonna tell me I can't have anymore babies due to the fact that I chose to be responsible when I was 18. I am only 23 years old and can't have a baby with my husband. My daughter deserves to have siblings and it's not fair. I felt I was cheated out of my family and will not stop until I find out what is going on with MY body. I dont know what else to do....
CarrieAugust 20, 2011 at 7:58 pm
I have been married for 7 years and have two children. I received my Mirena in April of 07'. I don't recall any issues with the insertion or side affects until my last year. I started noticing mood swings, back acne (not normal), cramps, and breast tenderness. When I went to the doctor this last week, she told me it was probably because it was coming to the end of the Mirena's "life cycle" and [Show More]I have been married for 7 years and have two children. I received my Mirena in April of 07'. I don't recall any issues with the insertion or side affects until my last year. I started noticing mood swings, back acne (not normal), cramps, and breast tenderness. When I went to the doctor this last week, she told me it was probably because it was coming to the end of the Mirena's "life cycle" and the hormones being released were dying down. I decided to have it removed and a new one put in (yes, dumb) and that is when things got worse. She could not find the strings (which I was NEVER told to check.....I found that out when I researched my symptoms). She informed me that the strings either retracted or the device expelled itself. If the strings were retracted she told me that they would have to put me under and find them with a camera. I was moved to ultrasound where they did locate it, BUT it had embedded itself into the wall of my uterus. I now have to go get it surgically removed at our local hospital. I will not be getting another IUD again. Not to mention that I have since found out that the Mirena produces a synthetic hormone......no thank you. I would advise against the Mirena.....I have no idea why this would not happen agian or to anyone else???? I have read about SEVERAL cases like this now.
LaChicAugust 19, 2011 at 6:28 am
Hello gang, Im a 31 yr old chick, I have a 5 yr old mini me. I got the Mirena on Dec.20 2007, I thought it was gonna be great, not gonna have to take a pill everyday...whoohoo!!! everything was working out fine them BAM!!! im on the bathroom floor with paramedics around me, Im "coming to" speaking to everyone, saying hello, hi. IM totally confused as to what the hell happened. The doctors are desc[Show More]Hello gang, Im a 31 yr old chick, I have a 5 yr old mini me. I got the Mirena on Dec.20 2007, I thought it was gonna be great, not gonna have to take a pill everyday...whoohoo!!! everything was working out fine them BAM!!! im on the bathroom floor with paramedics around me, Im "coming to" speaking to everyone, saying hello, hi. IM totally confused as to what the hell happened. The doctors are describing to me that I just had a SEIZURE. Im thinking to myself "where the hell that come from"...I've never had one in my 31 years of living. The docs wanted to medicate me and send me home.(i didnt want it). A couple more months go by...and here goes another one(daydreaming out the window)back to the hospital we go again. they hook me up to the machines to try and cause a seizure with all the flashing lights...NOTHING!!!....WHY because im not epeliptic...hhhmmmm. this time I take the med's. Now im feeling tingling in my fingers, im feeling slow..a little stupid and cant concentrate at times, and forget words that Im liiking for while talking...very frustratng!!!!. Now a year later everything is cool then BAM!!! POW POW...I hit a car and a light pole due to having a seizure while driving(tear)!!! Then it hits me...all this CRAP starting after I got this DAMN IUD!!!! but these damn shitty ass doc's wanna say its irrelivant??? really??? oh im crazy huh??? this thing is cummin out!!!! bet that!!!
ArianaAugust 16, 2011 at 2:51 am
I am a 21 yr old single mother of 2. I had the mirena put in May 2010 after my daughter was born. I cannot take the pills and i had the nuva ring before but it broke.. The mirena was kinda my last option since condoms aren't fool proof either. In aug. i stopped getting my period which i thought was a good thing until i got bacteria infections every other month around the time i would be due for on[Show More]I am a 21 yr old single mother of 2. I had the mirena put in May 2010 after my daughter was born. I cannot take the pills and i had the nuva ring before but it broke.. The mirena was kinda my last option since condoms aren't fool proof either. In aug. i stopped getting my period which i thought was a good thing until i got bacteria infections every other month around the time i would be due for one. I started having severe pains daily from my stomach to my back and legs. I could barely handle intercourse as well witout bein in pain. In dec the pains kept gettin worse and more constant. I went to my obgyn and found out i had cysts ony ovaries more on the left though he gave me pain pills and sent me home saying "they'll go away its normal". well from dec to feb 2010 the pain kept on worsening then i started to bleed randomly and it rush out like nothing. I went back to the hopsital and they couldn give me an answer. I made a follow up apt with my obgyn and had gotten an ultrasound. i found out i had 4 cysts on my right ovary and 5 one my left with fluid all around my ovary from one bursting. I was givin beyaz birth control to take for 2 mths to suppress them. Well in april i go back to find out my right side was ok but my left side wasn't. The cysts had conjoined with one other to form a a cysts the size of quarters and my ovaries are the size of walnuts! thats not a big difference. On the 29th of april i had surgery for the removal of the cyst and scar tissue from a c section. Only a mth n half later went by til i was bask at the er for bleeding n severe pain. They ended up finding yet another cyst on my left ovary again! They say i may have had on burst. I was in so much pain they had to give me morphine which didnt help that much. This pain was worse then contractions. To this day i have em sometimes i cant move my legs and my back cramps up to my shoulders. in july i went back to dr for bleeding yet again and the pain i was having. He wanted to make sure my iud was in place and it was. During the internal ultrasound they found 3 more cysts on my left ovary and one was a bigger then usual now i have to do the beyaz process again. Its been a month and honestly its gotten worse i hardly sleep from that pain and my back pain is gettin horrible. The dr had said as far as the pain goes it could be the iud or my cyst or both.. I wanted it removed at the time but he talked me out of it esp since i really didnt have options.. I said ok maybe ill ok its just the cysts. a couple wks later i i kept going through pain and painful intercourse to the point to where im afraid to do anything.. It hurts like hell! I made another apt in then end of july to remove it but he talked me out of it again.. i started doing more research and realized almost everything physcially and mentally i been through the past year may have been caused by this damn iud! I made an apt july 26 to have i out and had to wait 3 wks to do so. well las thursday i went to get it out and couldn't!!!y? well he treid for 5 mins and used different tools with out any lucky because the strings weren't visible. They're embedded into my cervix so i think.. i didnt get sent for an ultrasound or nething jus goin by what he says. The funny thing is he said he needed to reach my uterus.. but for what?? now i need to have this surgically removed friday.. i have a feeling my dr knows its not in my cervix but in my uterus! the crazy thing is this damn thing causes more problems then sovling them! when i was at my drs there was a woman at the desk with bc pills and a pamphlet in her hand for the mirena, and two mirena iud boxes sittin on the nurses desk.. Im starting to wonder if drs offices profit from woman having this inserted. Is that why they didnt want me to take it out?.. i am in the process of filing a lawsuit against the company who makes the mirena and my dr for Mal practice. I have been through more problems then explainable. i pray for all you women that have been through what i have or worse! if you still have it in you get it out!! I'm tired of the acne , swollen feet and hands,migraines,mood swings and bacteria infections!! i am a very clean person that makes me feel disgusting!
JackieAugust 13, 2011 at 5:16 am
I am 24. I had my son at 18. The doctor said I should get an IUD because it was the "best new thing". I had it in 2 years. It was painful sometimes where I couldn't sleep, couldn't have sex, I couldn't MOVE. I had to find one spot/position to lay in that didn't hurt. I finally had it removed. IN THE ER. Now I have that unpaid medical bill. Then every once in a while I can't have sex because the pa[Show More]I am 24. I had my son at 18. The doctor said I should get an IUD because it was the "best new thing". I had it in 2 years. It was painful sometimes where I couldn't sleep, couldn't have sex, I couldn't MOVE. I had to find one spot/position to lay in that didn't hurt. I finally had it removed. IN THE ER. Now I have that unpaid medical bill. Then every once in a while I can't have sex because the pain gets worse and worse. I went to the doctor 5 times in the past 3 years. Now I have all those medical bills. Each time the doctors said they couldn't find anything and tell me why I was in sooo much pain. For the past week my lower stomach hurt so bad I cried everyday and night. I didn't know what was wrong. Yesterday the pain started at 3am. I figured it would go away if I could sleep. I went to class, sitting in class the pain started to get so bad everyone around asked if I was ok. My teacher said either I get someone to take my to the ER or she was going to call the paramedics. I spent the next 6 hrs in the ER. The doctor told me I am pregnant. It is an ectopic pregnancy. I would have to get rid of it before it ruptured my fallopian tube, which I could die from. I was then told that it is because of having an IUD. I might not ever be able to have another baby. I have never been so depressed in my life. I really hope that this wont happen again. I learned that 1/3 of women who have ectopic pregnancy will have it occur again and again. 1/3 might be able to get pregnant again. and 1/3 will never be able to have a baby again. IUD needs to be taken off the market!!! And doctors need to actually CARE about their patients and tell them the truth instead of "selling" what can cause more harm than good!!!
RobynAugust 12, 2011 at 10:00 am
The Mirena was inserted on June 27th, an ultrasound was given to make sure it was in the proper place and it was. That evening I was having sharp pains and lots of blood but it had stopped. Checked strings and they were long and I could feel them. Later that night, I felt them again and this time they were much shorter. I checked them around lunch time on June 28th and couldn't feel the strings at[Show More]The Mirena was inserted on June 27th, an ultrasound was given to make sure it was in the proper place and it was. That evening I was having sharp pains and lots of blood but it had stopped. Checked strings and they were long and I could feel them. Later that night, I felt them again and this time they were much shorter. I checked them around lunch time on June 28th and couldn't feel the strings at all. I called my doctor and made an appointment for 9am on June 29th. My doctor checked for the strings and couldn't find them either. She then gave me an ultrasound and saw a white line but wanted to make sure and sent me for an X-ray right away. All this time, my boyfriend is supposed to be at work. The X-ray showed that it was in my body but didnt clearly show it's location so an MRI was ordered. After 30 plus minutes in the MRI, it was seen that the mirena was in my uterus but had migrated into the muscle of my uterus and needed to be removed. I then went back to my doctors office, it was now after 5 pm, to have it removed. My doctor tried numerous times to pull it out and after a lot of pain and blood, it was seen that it was not coming out easily and it was causing me great pain. An appointment was made to have an operation that consisted of a camera into my uterus for the removal and that I would be put to sleep. I called my mother who lives 4 hours away to come so that she could watch my children and that my boyfriend could be with me at the hospital this also means she would be missing two days of work. I wasn't able to eat or drink anything after midnight that night and I had to be at the hospital at 1pm and the operation was at 3pm. Without food and drink, this made it difficult for me to properly breastfeed my 6 week old baby. The operation was unsuccessful because the mirena and perforated my uterus and was now in my lower abdomen. Another operation was made to go through my belly button and my lower abdomen to remove the iud. I was told that some of possible complications could result in more bleeding, infection and possible hysterectomy. Also, because of the anesthesia, I cannot breastfeed my infant for 12 hours now. A specialist was called in to assist my doctor in this removal. The operation was successful and I woke up in recovery at around 11 pm. I was offered to stay the night and declined due to my boys needing me home. I was told that i cant lift over 10 pounds for 3 weeks, meaning I cant pick up my three year old and give him a hug. I will now have scarring in my belly button that will never go away and an experience that I'll never forget.
AprilAugust 11, 2011 at 8:52 pm
Has anyone been contacted by a lawyer yet?
CHERIEAugust 7, 2011 at 3:29 am
I had the IUD placed for the use of birth control in Aug 2008, so I could not have children for a while. About four weeks after placement I begun to have many complications. I continually had severe abdominal pain. I went to the same doctor in which he said that the IUD was not in my cervix and then continued to order an ultrasound of my abdomen to make sure it was not in my cervix. They did not s[Show More]I had the IUD placed for the use of birth control in Aug 2008, so I could not have children for a while. About four weeks after placement I begun to have many complications. I continually had severe abdominal pain. I went to the same doctor in which he said that the IUD was not in my cervix and then continued to order an ultrasound of my abdomen to make sure it was not in my cervix. They did not see it in there. But did say that I concurred pelvic inflammatory disease(PID) from having the IUD. They prescribed me medication and placed me on the depo prevera so I could continue to have birth control. But even though I finished all antibiotics for the infection I continued to have abdominal pain. In which I was seen several times for. After that they begun to tell me that I was having pain due to the depo shot I was on. But the pain never got better, it seems as though it gradually got worse and even though I seen the doctor many times they would tell me they could not find anything wrong except for the fact that the depo can cause the issues I was having. I have been very depressed, crying for no reason, back pain, pelvic pain, abdominal pain, really bad headaches, irritability, angry, and just plain not myself. So I then decided that the pain was not worth being on birth control so I then decided to get a tubal ligation so I could stop having all the problems I was having. So in Dec. 2010 I got the surgery done which was a permanent form of birth control but I couldn't help to think that I could finally live my life without the pain I was having. But guess what, I was wrong. The pain begun to get worse and worse. On July 20, 2011 I went to my doctor with sever abdomen pain with sever back pain, nausea, vomiting, fatigue, dizziness, and unable to eat or drink causing dehydration. The doctor that thought I may have something wrong with my appendix, so she sent me to the emergency room to get blood work, IV fluids, and a CT scan to find out what was wrong. So after seven in a half hours of sitting in the emergency room waiting to find out what was wrong and why I was in so much pain I get told something completely unexpected. I have an IUD jammed in my intestine and there is nothing they can do until I got see my OD GYN. So I got home stress all night about what was going to happen next. I go see the OBGYN the next morning for him to tell me that I have to have surgery to remove the IUD immediately. WOW!!!! Now I have many questions in the back of my head the first of which is, "WHY ME"! Why did they not try to find this 3 years ago when the pain first began. All the times I went and seen the doctor why did he not look into it more? Why did they not see it when they did my tubal ligation in December? Why didn't they do a CT scan or X-Ray in the first place to make completely sure? Now I have to go in have the surgery and miss work that I cannot afford to miss. Plus be in this sever pain because they couldn't be more couscous. This is not fair to me and I think that this was an issue of negligence on their part. If they would have taken the time out to actually look into my pain in the first place would I be going through this now? Now I got my tubal ligation done and will never have children due to you misleading my thoughts. I knew there was something wrong and you kept making me feel that I was wrong. That there was nothing wrong with me. My life has been miserable due to your un willingness to say that patient comes first. Due all possible to make sure they are comfortable. " I WAS NOT COMFOTABLE" I was in constant pain and you made me feel like I was crazy. I now know I was not and it will be fixed. But it is not fair for me to have to go through this. You should make more time for your patients and make them feel like you care. Or how about this why don't you try to ACTUALLY CARE. Thank you for listening to my story and the questions that I wish could be answered and even the things I would love to say to them.
melissa hallAugust 4, 2011 at 12:51 am
i had mirena put in after my first child. had it in a little over a year and had it removed in dec, in hopes of having another baby. by august my periods completly went away and a couple af months later and after many test i was diagnosted with premature ovarian failure. from everything i have been reading many many women have had the same outcome after using the mirena. now i have to go to a fert[Show More]i had mirena put in after my first child. had it in a little over a year and had it removed in dec, in hopes of having another baby. by august my periods completly went away and a couple af months later and after many test i was diagnosted with premature ovarian failure. from everything i have been reading many many women have had the same outcome after using the mirena. now i have to go to a fertility specialist with no insuranse. my chances of having another baby are very low even with fertility treatments.
MonicaAugust 3, 2011 at 7:42 pm
I had the Mirena for almost a year, I have never had problems with migraine before. It didn't hurt to have it put in but I always had migraine or headaches that never seemed to go away, I was tired all the time moody and had abdominal pains. I had to fight with the dr to remove it I would never consider recomending this birthcontrol to anyone ever. Now that I've had it removed I feel 100% better t[Show More]I had the Mirena for almost a year, I have never had problems with migraine before. It didn't hurt to have it put in but I always had migraine or headaches that never seemed to go away, I was tired all the time moody and had abdominal pains. I had to fight with the dr to remove it I would never consider recomending this birthcontrol to anyone ever. Now that I've had it removed I feel 100% better tylenol works for any kind of headaches I get now, I'm no longer tired no pain like when I had this IUD. I hate the mirena and would like to see it removed from the market!
MarleeAugust 1, 2011 at 3:07 pm
This is in response to Jeannie's comment about Mirena - I have had this device in for almost three years. I have experienced only the lower back pain during ovulation each month. Other than that I almost forget it's there. I would however be concerned that a scan done did not pick it up -- hard to believe it was missed. I do know that they sometimes are expelled by the body but my doctor assure[Show More]This is in response to Jeannie's comment about Mirena - I have had this device in for almost three years. I have experienced only the lower back pain during ovulation each month. Other than that I almost forget it's there. I would however be concerned that a scan done did not pick it up -- hard to believe it was missed. I do know that they sometimes are expelled by the body but my doctor assures me she has not had this occur wih any of her patients. My advise to Jeannie is go to a different doctor and get another scan and opinion before relaxing about this. I would want to be sure. The one GREAT advantage of Mirena other than preventing unwanted / unplanned pregnancies is no heavy periods just light spotting. I haven't had to use a tampon or pad in almost 2 years. Love it!
JeannieAugust 1, 2011 at 1:48 am
Well...I had Mirena put in ablout 4 months ago (6 weeks post baby). I reall didn't experience many side effects other than headaches, severe abdominal cramping, lower back pain, shooting pains in my vagina and but areas, acne! I bled for 5 weeks and when I went back for my check up on the Mirena my doctor couldn't find the strings to the device!!!! I went for an ultra sound and they couldn't see i[Show More]Well...I had Mirena put in ablout 4 months ago (6 weeks post baby). I reall didn't experience many side effects other than headaches, severe abdominal cramping, lower back pain, shooting pains in my vagina and but areas, acne! I bled for 5 weeks and when I went back for my check up on the Mirena my doctor couldn't find the strings to the device!!!! I went for an ultra sound and they couldn't see it anywhere! After a series of procedures to find the IUD (hystoscopy, CT scan) the only logical conclusion they can come up with is my body expelled the Mirena! I was content with that! Until about 2 months ago I have started cramping again. Still having shooting pains in my vagina and butt area, lower back pains and really bloated feeling!!!! I am really starting to wonder....all the tests that they ran on me to look for the Mirena..could they have missed it somehow and it still be there imbedding itself in my uterus or worse my liver or something???? Could someone talk to me about this and ease my mind alittle bit on this situation? Do you think I should go back to the doctor and have more tests ran? Or do you think its just all in my head??!!!
LisaJuly 31, 2011 at 2:01 am
My IUD was placed in May of 2007, just 6 weeks afer my miscarrage. I agree with the rest of the women who say it hurts when the doctor is putting it in,however it hurt because your uterus was contracting and that's to be expected. I did have some discomfort and spotting for a few days after insertion but it went away. My periods subsides ,I did have some headaches and migraines but I have always [Show More]My IUD was placed in May of 2007, just 6 weeks afer my miscarrage. I agree with the rest of the women who say it hurts when the doctor is putting it in,however it hurt because your uterus was contracting and that's to be expected. I did have some discomfort and spotting for a few days after insertion but it went away. My periods subsides ,I did have some headaches and migraines but I have always had problems with headaches in the past. Myself, getting the mirena IUD was the best thing so far. I have to say that this was the best birth control method for me. I will be calling in a few months to schedule my appt. to have it removed and a new one placed. I'm hoping all goes well with the removal- although reading some of these post really scare me.
AlyssaJuly 28, 2011 at 10:34 pm
I was 20 when I got the IUD put in Feb. 2010.Even while I was pregnant my doctor talking like the IUD was the best birth control to have. After having my daughter, I was put on Iron Pills because I was anemic. After my 8 week check-up, my doctor put the IUD in. He told me it would only hurt for a day and should only feel like cramps and was only supposed to bleed for a few days. I bleed for 7 MONT[Show More]I was 20 when I got the IUD put in Feb. 2010.Even while I was pregnant my doctor talking like the IUD was the best birth control to have. After having my daughter, I was put on Iron Pills because I was anemic. After my 8 week check-up, my doctor put the IUD in. He told me it would only hurt for a day and should only feel like cramps and was only supposed to bleed for a few days. I bleed for 7 MONTHS after having it put in. I went back for a 3 month check up and told him I had really bad headaches, bad mood swings (the mood swings was so bad my husband and I could be talking normally and then all of a sudden I would snap and go off), unbearable cramps that would result in sleepless nights, back pain, weight loss (I am usually around 120 to 125lbs and I got down to like 108lbsish) and depression (I had never been depressed in my life but I didn't want to get out of bed, play with my daughter, clean, cook or anything). He told me it was just because I just had the baby 3 months before. I thought post-partum usually lasted the 8 weeks. He also told me that the bleeding should stop. I went home...sad, depressed, cramping and a awful headache. I went home and googled side-effects of the mirena and got forum after forum of woman with the same problems as me. I went back to the doctor 3 months later for my yearly and told him to take it out or I would find someone else too. He tried to talk me into keeping it in and saying all this was in my head and he had never heard of any of this or had any other woman complain about it. Needless to say, he took it out and that was in the July 2010. A year later I feel like my old self. I do believe bleeding, headaches, cramps, and etc that I had was a result in the mirena.
SarahJuly 22, 2011 at 10:46 pm
I had an IUD inserted in November 2010. After 6 months of bleeding on and off, I went in to have it checked. After doing a vaginal check, ultrasound, and x-ray, the IUD was found in my abdominal cavity. I had to undergo laprascopic surgery to have it removed, and I am EXTREMELY frustrated that I 1- was not warned about this risk 2- am paying for this surgery. I think that this happens more ofte[Show More]I had an IUD inserted in November 2010. After 6 months of bleeding on and off, I went in to have it checked. After doing a vaginal check, ultrasound, and x-ray, the IUD was found in my abdominal cavity. I had to undergo laprascopic surgery to have it removed, and I am EXTREMELY frustrated that I 1- was not warned about this risk 2- am paying for this surgery. I think that this happens more often than told it does. I don't know whether this is a placement issue with the doctor, or a malfunction of the IUD itself.
jessieJuly 20, 2011 at 7:35 pm
I had mirena implanted in the beginning of 2008 at my six week checkup after my c-section. i left it in for 2 years before having it removed. i gained 40 lbs and still have not lost it and had severe cramps the whole two years along with other complications. mirena should be taken off the market
ShellyJuly 20, 2011 at 6:04 am
There was some typos in my last comment..i have four kids wanted six..and mirena made me infertile not clomide sorry
SshellyJuly 20, 2011 at 5:59 am
I have kids already always wanted a big family already six...i come from a big family so does my husband..after my forth child I got mirena put in was told totally safe and I could get pg as soon as I got it removed...wel we decided to get it removed after having it almost a year started trying right away it's now ben two and a half years stiil no baby...even did a few rounds of clomid!!! I'm only[Show More]I have kids already always wanted a big family already six...i come from a big family so does my husband..after my forth child I got mirena put in was told totally safe and I could get pg as soon as I got it removed...wel we decided to get it removed after having it almost a year started trying right away it's now ben two and a half years stiil no baby...even did a few rounds of clomid!!! I'm only 27 and have gotten pg so fast in the past...i know clomid has made me infertile and I'm so angry!!!i wish I would have known.this bc is the devil..I'm so sad and sorry for all the women affected by this..i read all your stories and cried...i hope we can save o ther womenfrom this heart ache.
ChristinaJuly 18, 2011 at 8:30 pm
As the others that have posted on this page, I am a mother. I got my IUD in six weeks after my first child was born in 2007. I was told that I should still have my period but with less cramping. WRONG! I experienced a extremely lite period that same week. After that did not have another period and explained this as one of the side effects. As of the end of the year in 2008 me and my husband decide[Show More]As the others that have posted on this page, I am a mother. I got my IUD in six weeks after my first child was born in 2007. I was told that I should still have my period but with less cramping. WRONG! I experienced a extremely lite period that same week. After that did not have another period and explained this as one of the side effects. As of the end of the year in 2008 me and my husband decided to try to have another child. Once it was removed I have only had 3 periods on my own. When they are on my own I bleed for close to a month before it stops. I go to my doctor often to get a prescription to force my body to have a period. Which means that I pay close to $30.00 a month to have what should come naturally. Before Mirena, I had the perfect 5 day period, same date every month. My doctor is now testing my blood to see if I am even ovulating anymore; and to check my harmon levels. So now, I fear that the "choice" that I thought I still had to have another child has been stolen from me and my family. Where I did the responsible thing to prevent pregnancy till my husband and I where back on our feet; is what has broken my heart now. I feel as if someone has robbed me. I have been also put on depression meds, due to my up and down mood swings. If I try to pick up my now four year old I feel as if my uterus is going to rip apart. At the age of 26 I have lost hope.
VickyJuly 18, 2011 at 6:13 pm
I am 24 years old and just had to have sugary to have this AWFUL Merina IUD removed in trun I had to have my tubes tied because of all the scar tissue and infection that this nightmare caused me. I had awful nightsweats and insomnia. I also had bad mood swings some of witch got violent and others where I was so depressed I could not take care of my family. I had awful migranes and body aches and m[Show More]I am 24 years old and just had to have sugary to have this AWFUL Merina IUD removed in trun I had to have my tubes tied because of all the scar tissue and infection that this nightmare caused me. I had awful nightsweats and insomnia. I also had bad mood swings some of witch got violent and others where I was so depressed I could not take care of my family. I had awful migranes and body aches and massive bleeding spells with sharp pains to my pelvic area and overys. I can not believe that I am 24 and have had to have this operation and now can not have have children with the man I plan on spending my life with. This Merina IUD has caused me much pain and suffering and my hope is that this product be taking off the market so it can not ruin anyone elses life.
AprilJuly 17, 2011 at 8:26 pm
Hello. I am a mother of two beautiful baby boys who are 3 and 22 months. I had mirena put in during my 6 week post partum appt. It was painful going in and i bled for several days after. A few months went by and everything was fine. Then I started having severe migranes to the point that I would be huddled over the toilet because it would make me so sick. I could not even go to sleep because they [Show More]Hello. I am a mother of two beautiful baby boys who are 3 and 22 months. I had mirena put in during my 6 week post partum appt. It was painful going in and i bled for several days after. A few months went by and everything was fine. Then I started having severe migranes to the point that I would be huddled over the toilet because it would make me so sick. I could not even go to sleep because they were so bad. Then in the fall of 2010 I started cramping really bad and had tenderness in my abdominal area to the point that I could not even wear jeans so I made an appointment with my obgyn. When i went in she did an ultrasound and found a cist on my right ovary. She told me there was nothing that could be done and it would go away on its own. Since then I have had more cists and one time I was in town I started cramping so i came home to lay down hoping that it would go away but the pain only got worse. I was huddled up into a ball in the floor and had to be rushed to the hospital. It hurt so bad to move and walk and even worse to breath. When i got to the hospital they gave me a shot and the pain decreased a little. They sent me home. I have gained over 30 pounds and have had the moodiness, crying about EVERYTHING for no reason, breast soreness, migranes, bleeding for long periods of time strong and heavy, and now I have an infection. The string that is supposed to hang down is gone but yet they still say that everything is in place and it scares me that when I get it taken out, how are they going to get it out if there is no string? Something needs to be done and bayer should have to pay for what they have done to all these women and their lives. They can give them their money from their doctor appointments back but there is no way that they can give them their lives back. I plan on having more children with my recent boyfriend and if I am unable to have kids I will be furious. it is not fair to him and he should not have to pay for their mistakes. They are fake and misleading and it is rediculous!!! Something needs to be done to stop this and take it off the market. It is NOT SAFE and it defenitly IS NOT "the best" form of bc. I am so scared that this infection is going to get worse. I have to take care of my children and they need me! I want every one of these women to have their lives back and I know that I want my life back not only for me but for my family!
Kelli GJuly 17, 2011 at 10:34 am
I had my 1st Mirena insterted in January 2008 after my daughter was born (Sepember 2007). I was 19 and in college planning to continue to get my degree. I experienced many of the symptoms described (emotional outbursts, weight gain, fatigue, etc..) but attributed it to being a new mom and the stresses that go with it. When my daughter was 10 months old (7 months post insertion) the Mirena expelled[Show More]I had my 1st Mirena insterted in January 2008 after my daughter was born (Sepember 2007). I was 19 and in college planning to continue to get my degree. I experienced many of the symptoms described (emotional outbursts, weight gain, fatigue, etc..) but attributed it to being a new mom and the stresses that go with it. When my daughter was 10 months old (7 months post insertion) the Mirena expelled itself. I decided to take a home pregnancy test before calling my OBGYN to get a new one inserted, and much to my shock it came back positive. I had not had a period for almost 4 months at that point and had no clue when I may have become pregnant. I turned out to be only 5-6 weeks (based on a blood test), At the time my insurance had changed and the OBGYN I had used for my daughter did not accept my new insurance, so I was seen by a new doctor for this pregnancy. The pregnancy was a nightmare. Crazy morning sickness, no weight gain. Preterm labor starting at 23 and continued to have issues so I was placed on bedrest and was able to contiue the pregnancy to approx. 34 weeks when my water broke. My son was born in March 2009 weighing 4 lbs 14 oz and spent 2 weeks in the NICU on breathing/feeding tubes. One of the most difficult times of my life... I decided I was not ready for a tubal ligation, but wanted something long term. After being convinced by the doctors of the great results and feedback of Mirena and that I was likely improperly inserted.not checked properly, I had a second Mirena inserted. Again I got the much loved side effect of not having a period (my main reason for going with Mirena again) and life continued as normal...until October 2009. Deja Vue....My Mirena again was expelled so again I took a test, positive instantly, and called the doctor to schedule an appointment. My OBGYN was shocked! aparrently the odds that happening to me (twice) were apperently astounding. So I had an ultrasound to see if it was in the uterus and it was...again at about 5-6 weeks gestation. My doctor spoke with their "Mirena Rep" and told them about my situation and the had no answers. Of course... The third pregnancy was less eventful and my second daughter was born full term at 37 wks 1 day weighing 6 lbs 4 oz. I love my kids and wouldn't change a thing now but it was not in my plans to be a mom of 3 under 3 at 21 years old. My husband and I decided it was time for sterilization since we had had so many failures (my 1st daughter was concieved on Nuvaring and was growth restricted so I was induced at 38wks, she weighed 5lbs 12oz). At my postpartum check-up my OBGYN said she had another Mirena pregnancy who was at 9 weeks. So I know I am far from alone. Mirena is far from the wondering-drug it is advertised as.
CORTAEY BJuly 12, 2011 at 10:04 pm
I got the mirena inserted monday june 13,2011 it was painful my ob/gyn told me the pain should only last about 30 mins and feel like menstrual cramps, which i didn't know what that felt like b/c i'm one of the few woman who don't have menstrual cramps the whole process took about 5 mins when he was done i barely could move it hurt so bad that he had 2 come back 2 the room and check 2 c if i was ok[Show More]I got the mirena inserted monday june 13,2011 it was painful my ob/gyn told me the pain should only last about 30 mins and feel like menstrual cramps, which i didn't know what that felt like b/c i'm one of the few woman who don't have menstrual cramps the whole process took about 5 mins when he was done i barely could move it hurt so bad that he had 2 come back 2 the room and check 2 c if i was ok after he left out the pain continued for two days i returned wednesday june 15,2011 for an ultrasound to make sure everything was ok which it wasn't. He couldn't find the string i was sent to a near by hospital for an xray then a ct scan and the mirena had perforated my uterus all within a two day period of time that is crazy and unheard of today is tuesday july 12,2011 i had 2 go through surgery yesterday to get it removed and been bleeding ever since the day i got it inserted i feel weak and sore all over i have a 4 month and 3 yr old child that i have 2 push off on all my family member b/c of the pain i can't take care of them myself i been out of work 4 about 4 weeks now and on the verge of losing my job and apartment .... i wouldn't recommend this method of BC to anyone
StephanieJuly 7, 2011 at 5:11 am
I got the iud in 2008 I was bleeding for three striaght mths sixs mths after started exp. backpains when I wake up in.the morinings then.it seems as if every thing went back to normal no heavy periods no backaces till the begining of this year my period stay on for two weeks at a time my backaces are back there worse then before not only do I wake up with them it's hard to sleep at night because o[Show More]I got the iud in 2008 I was bleeding for three striaght mths sixs mths after started exp. backpains when I wake up in.the morinings then.it seems as if every thing went back to normal no heavy periods no backaces till the begining of this year my period stay on for two weeks at a time my backaces are back there worse then before not only do I wake up with them it's hard to sleep at night because of them if im standing in one position for a while like washing dishes it's painful and not to mention the other side effects( headace ,fatige, low iron tender breast, and moodswings tired all the time went to doc they said it has nothing to do with iud I dn't knw if it's true dn't knw what to do someone help me please sooo.... tired of going through tthis oh yea and weight gain
AliJuly 1, 2011 at 1:14 am
I had a Mirena inserted 4 years ago. Prior to that I was in good health...fit, running marathons, no medical issues to speak of. After the Mirena was inserted I started developing cystic acne, several dermatologists suspected the Mirena but I was told by the nurse practitioner who inserted it it was not. So I have been on antibiotics as well as tried tons of products to get rid of it since then wi[Show More]I had a Mirena inserted 4 years ago. Prior to that I was in good health...fit, running marathons, no medical issues to speak of. After the Mirena was inserted I started developing cystic acne, several dermatologists suspected the Mirena but I was told by the nurse practitioner who inserted it it was not. So I have been on antibiotics as well as tried tons of products to get rid of it since then without success. About a year after it was inserted, at my annual exam I told the nurse practitioner that I had what I thought was a gallbladder attack and described it to her. She said I did not fit the profile and she felt strongly that it was a bad gas attack, so I left it at that. Around that time I also had an unexplained weight gain (15 lbs or so) and saw an endocrinologist who diagnosed me with insulin resistance (no family history) and he put me on Metformin. A few months later I ended up in the ER with a severe gallbladder attack, 3 weeks later it had to be removed leaving me with all kinds of fun bowel issues now. A couple of months ago I gained 12 lbs in 2 weeks and am now up to a total weight gain of around 16 lbs...again, no idea why. And the weight gain is all around my mid section, hips and my breasts are sore...to the point that I asked my doctor to give me a pregnancy test (it was negative). I have an appointment later this month to get it removed.
ChristinaJune 29, 2011 at 2:43 pm
I went to my obgyn last week to have my mirena removed (at the end of the 5 years). They could not remove it (it was stuck). The next week I had to have an ultrasound where they took a ton of pictures to see where the IUD was/is. Going to the exam room I still thought the doctor would be able to remove it that day. Unfortunately, I have to have surgery to remove it. They're not exactly sure wh[Show More]I went to my obgyn last week to have my mirena removed (at the end of the 5 years). They could not remove it (it was stuck). The next week I had to have an ultrasound where they took a ton of pictures to see where the IUD was/is. Going to the exam room I still thought the doctor would be able to remove it that day. Unfortunately, I have to have surgery to remove it. They're not exactly sure where it is (it is embedded in my uterus) but they're not sure if it has adhered itself to anything else. I would not recommend.
AdrienneJune 20, 2011 at 5:45 pm
Sorry. I got my dates incorrect. I had surgery June 2011. Last week. I'm still on some pain meds after surgery. Oops.
AdrienneJune 20, 2011 at 5:43 pm
I had Mirena put in march 2011. I previously had been using the nuvaring which I thought was great but relatively expensive. I had talked to my dr about switching to Mirena and she thought it was amazing having it herself for 3 years. The pain during insertion was bad and I vomited in the office. After a few hours the pain went away and I was feeling better. About 10 days later I started having [Show More]I had Mirena put in march 2011. I previously had been using the nuvaring which I thought was great but relatively expensive. I had talked to my dr about switching to Mirena and she thought it was amazing having it herself for 3 years. The pain during insertion was bad and I vomited in the office. After a few hours the pain went away and I was feeling better. About 10 days later I started having extreme "cramps". I was having a hard time walking and standing. I had called the dr office and they told me my body was still getting used to Mirena and I shouldn't worry. I wasn't able to locate my strings either. But she was not concerned. I had my six week checkup and she was also unable to find my strings. I had an ultrasound and they could not locate the iud. They sent me for xrays where they found it in my lower abdomen. It had perforated my uterus and had to be removed asap. I had to have laproscopic surgery July 2011 to have it removed. And now the bills are coming in. This is not what I had signed up for! I thought I wouldn't have to think about birth control for five years! Nice! But after having surgery I will tell everyone I know not to get Mirena. It's not worth it.
ElizabethJune 16, 2011 at 2:46 am
I had the Mirena IUD put in in Jan 2010. I immediatly experienced a bloated feeling and gained 10 pounds. I also spotted nonstop. I went to the Dr. who told me it can take time to get used to it. I spotted for 6 months strait. Finnally it stopped and I had what seemed like cycles where I spotted for a few day in place of a regular period. At 3 months after insertion I started having a very strage [Show More]I had the Mirena IUD put in in Jan 2010. I immediatly experienced a bloated feeling and gained 10 pounds. I also spotted nonstop. I went to the Dr. who told me it can take time to get used to it. I spotted for 6 months strait. Finnally it stopped and I had what seemed like cycles where I spotted for a few day in place of a regular period. At 3 months after insertion I started having a very strage illness that caused me to wake up tingling in my hands and feet that would be follow by very severe nausea and throwing up for around 3-4 hours. I have had aver GI test known to man and am completely normal. 12 months post insertion I started having severe migrains during my "cycle" I have gained a total of 30 lbs without change in diet in the past 1 1/2 years. I have hair loss, cystic acne, extreme fatigue, loss of sex drive, joint pain, shooting pains up back and down legs from lower back, very low blood pressure, mystery nausea illness, dizziness. I had Mirena removed 6/8/11, I have experienced very eavy bleeding and blood clots for 2 days so far. The whole time I have been told by my OBGYN that Mirena can't cause these symptoms. After doing research I have found he is either lieing or ignorant. These things are dangerous and I fear I am still in for a lot more problems with this thing. DON'T GET ONE.
camelliaJune 9, 2011 at 4:43 am
I had the IUD put in 6weeks after my little girl was born in 2009. I have had pain the whole time it has been in but I thought it was normal. Then in june of 2010 I had about three weeks of real bad pain, I have no health insurance so I waited thinking it would go away. Then it got so bad I could not wait anymore so I went,and they could not find my IUD so now I have been to the doc about 4times a[Show More]I had the IUD put in 6weeks after my little girl was born in 2009. I have had pain the whole time it has been in but I thought it was normal. Then in june of 2010 I had about three weeks of real bad pain, I have no health insurance so I waited thinking it would go away. Then it got so bad I could not wait anymore so I went,and they could not find my IUD so now I have been to the doc about 4times and they put me on meds of infection well five days after being on the meds my mouth started getting sores all over,one night I was having trouble breathing so my husband took my to the ER and i was told it was a reaction from the med and that my mouth my not heal. So I am having the IUD removed june 10 and 2:45 and I was faced with the fact that I may never be able to have more children at 25 yr old how can you deal with someone telling you that you may never be able to have kids. please help this is not something anyone should have to go through
StaceyMay 28, 2011 at 3:24 am
Have been trying for almost a year to have a 2nd baby and think it is from the damage caused by the Mirena I had. Frequently had baseball size cysts that ruptured for months!!!! Ever since I had that I struggle to orgasm during intercourse and now I am afraid I cannot have another baby! I have no insurance and have been taking ovulation tests and do not think I am ovulating because of the damage f[Show More]Have been trying for almost a year to have a 2nd baby and think it is from the damage caused by the Mirena I had. Frequently had baseball size cysts that ruptured for months!!!! Ever since I had that I struggle to orgasm during intercourse and now I am afraid I cannot have another baby! I have no insurance and have been taking ovulation tests and do not think I am ovulating because of the damage from the bursting cysts which were caused by the chemicals in the Mirena. So angry and heartbroken!!! Tell every woman you meet NOT to have a Mirena!!!
HyperfatigueMay 19, 2011 at 6:58 am
I too, have experienced the fatigue, eye twitching that was way too frequent and lasted over a month, hot flashes, night sweats, 20 lbs. Weight gain, shortness of breath, heart palpitations, rapid heart rate/pulse, trouble sleeping, etc. I requested a TSH and T level testing, despite how much my NP tried to convince me I was depressed and not suffering from Hyperthyroidism as I suspected. My feeli[Show More]I too, have experienced the fatigue, eye twitching that was way too frequent and lasted over a month, hot flashes, night sweats, 20 lbs. Weight gain, shortness of breath, heart palpitations, rapid heart rate/pulse, trouble sleeping, etc. I requested a TSH and T level testing, despite how much my NP tried to convince me I was depressed and not suffering from Hyperthyroidism as I suspected. My feeling, as usual, was right-Hyperthyroidism, I mentioned that my family history did not include any Thyroid problems, and that I suspected it was the Mirena IUD. She assured me it wasn't. I went home and googled if the mirena could cause Hyperthyroidism, and came across several forums, blogs, and posts describing exactly what I was experiencing. I brought it up to my Endocrinologist at our first appointment, and she assured me there was no corralation between the two. I mentioned how it was odd that many women were going through the same symptoms as I was, and asked if there was enough research done on this device. She mentioned how the internet only provides you with a lot of misinformation, and shouldn't be used as a reliable source for medical advice. I again tried to explain to her that I found it odd that she could dismiss it so quick, and she said that most of these sights are competitors trying to make money by spreading false accusations. I let her know there was too many sites, too many women, and too many of the same stories for that to be the case, and she tried to talk me into a scan. I let her know that I would have my IUD removed the following day and have my TSH and T levels tested after 6 weeks again. She made me a follow-up with her at the end of July 2011. I will try and update with good news then, God willing. Best of wishes to everyone out there receiving the same quality of care, or lack there of.
MelissaMay 15, 2011 at 12:45 am
I had Mirena put in Jan. 2011. I have had several symptoms but thought it was always from something else. Night sweats, no energy, waking up in night, stiff neck, low back pain, sore muscles and joints, thinning eyelashes and recently a random sharp, breath taking pain in middle of back near spine that feels like bee stings and now very rapid thinning hair and hair loss which is very stressful a[Show More]I had Mirena put in Jan. 2011. I have had several symptoms but thought it was always from something else. Night sweats, no energy, waking up in night, stiff neck, low back pain, sore muscles and joints, thinning eyelashes and recently a random sharp, breath taking pain in middle of back near spine that feels like bee stings and now very rapid thinning hair and hair loss which is very stressful and depressing. I had my anual exam yesterday and told my Dr. about the thinning hair loss and she said I bet its your Mirena. We did blood work to rule out other things. Get results Tues. I have done nothing but research since my appt. and I believe I will be getting this removed asap no matter what blood work says. I did research before having it inserted but never came across this website. Really upset I didn't. I have history of ovarian cysts always with surgery, so am worried about getting them when this is removed. I also have migraines and am on daily medicines for these so hope removal doesn't make them worse. Thanks for your stories. I hope the best for everyone.
MonicaMay 13, 2011 at 3:29 am
I had my last child on June 26,2008 the iud was placed inside me in august of 2008 it was a very painful procedure and was very discomfortable months went by i gained weight cycles became heavier and irregular with clots and i had severe migranes and pelvc pain. My skin change leaving me with blemishes and acne in Feb.2009 my iud came out on its own in this huge clot that looked like a 4mo. fetus [Show More]I had my last child on June 26,2008 the iud was placed inside me in august of 2008 it was a very painful procedure and was very discomfortable months went by i gained weight cycles became heavier and irregular with clots and i had severe migranes and pelvc pain. My skin change leaving me with blemishes and acne in Feb.2009 my iud came out on its own in this huge clot that looked like a 4mo. fetus it felt as though i was going in labor i didnt feel any better until it was passed after that my cycles were worsening thick and slimy at time and running like water ranging from 7-25 days i kept getting sick blood count kept dropping no matter how many iron pills i took in march 2010 i discovered i was pregnant 2wks later i had a miscarriage my health kept declining in Nov2010 i found a new doctor that ran tests and found out i had fibriods, cysts, and really bad scar tissue i was told i cant have any more kids i had surgery in nov2010 to help with the cramping and bleeding it didnt work so in Feb2011 i was given another procedure called an abilation that was suppose to work it didnt also so now i have to go back in July2011 to have a hysterectomy and im only 25.
jennfierMay 10, 2011 at 6:10 pm
I have had it put in twice the first time at 19 years old and had removed within 6 months because of horrible mood swings and lower back pain. I just had my second daughter in september and had it put in within 2 months and nobdy told me that increased my chances of it going through and now at 21 it has gone through the wall of my uterus partially. i also have horrible mood swings and migranes t[Show More]I have had it put in twice the first time at 19 years old and had removed within 6 months because of horrible mood swings and lower back pain. I just had my second daughter in september and had it put in within 2 months and nobdy told me that increased my chances of it going through and now at 21 it has gone through the wall of my uterus partially. i also have horrible mood swings and migranes that are so bad i am having to go to the doctor and she said it will probably take surgery to get it out.
nMay 5, 2011 at 4:46 am
Sarah- go to urgent care at your local ER and they should be willing to take it out. I am fine now, a month after removal of the mirena the 'crash' effects have gone- I thought they never would! It really was a complete nightmare. I did everything I could think of- flushing my system out will lots of water, eating lots even when I had no appetite, lots of exercise and my life saver I believe- acc[Show More]Sarah- go to urgent care at your local ER and they should be willing to take it out. I am fine now, a month after removal of the mirena the 'crash' effects have gone- I thought they never would! It really was a complete nightmare. I did everything I could think of- flushing my system out will lots of water, eating lots even when I had no appetite, lots of exercise and my life saver I believe- accupuncture! I even went to church.i was never religious but it gave me a 'safe' place and a feeling that I would come through this. My poor husband and children that they had to endure this.
ClareMay 4, 2011 at 4:45 pm
I am in the third year of my second mirena. When it was time to have the first one out, my obgyn said my symtoms couldn't possibly be from my mirena. She even gave me a spendy prescription for facial hair. She also told me I should consider leaving my boyfriend if I didn't want to have sex with him, it must be his fault. I went ahead and got the second one put in, and have gotten much worse since.[Show More]I am in the third year of my second mirena. When it was time to have the first one out, my obgyn said my symtoms couldn't possibly be from my mirena. She even gave me a spendy prescription for facial hair. She also told me I should consider leaving my boyfriend if I didn't want to have sex with him, it must be his fault. I went ahead and got the second one put in, and have gotten much worse since. Same story, different woman with a ruined life. A couple of weeks ago I collapsed at work, and was having terrible chest pain. ER doc said acid reflux. The follow up doc said stress and anxiety. After seven years of headaches, nausea, chest pain, depression, anxiety, joint pain, weakness, no sex drive and my beautiful woman beard, I did my own research. I am shocked. I can't believe none of these "medical professionals" would make this connection. I plan to have mirena removed as soon as I can. I now have to tell my 18 year old daughter, who I forced to get a mirena a year ago, that mommy has probably helped mirena ruin her life. How many of these symptoms are irreversible? Do I get to spend the rest of my life being a giant bearded lady? Will I ever stop hurting? Will the damages to my relationships be forgiven? These people must be stopped. That can't be unaware of what they're doing to people with their "miracle birth control."
SarahMay 3, 2011 at 4:09 am
In Aug of 2010 I woke up to find a huge bald spot on the right side of my head the size of a hand. Freaked out I went to the doctor. I have been losing weight ever since. Originally at 130 pounds I know weight 103 pounds. I have had memory issues, night sweats, menstrual cycle every two weeks for the last 4 months, mood swings, high anxiety, and cramping on my right side that felt like my ovaries [Show More]In Aug of 2010 I woke up to find a huge bald spot on the right side of my head the size of a hand. Freaked out I went to the doctor. I have been losing weight ever since. Originally at 130 pounds I know weight 103 pounds. I have had memory issues, night sweats, menstrual cycle every two weeks for the last 4 months, mood swings, high anxiety, and cramping on my right side that felt like my ovaries where gonna pop. My hair has continued to fall out. Having 10% diffusing along with 4 bald spots. My doctor ran every test in the book he could think of until this last Friday he said maybe its your IUD. Now Im convinced because my boyfriend told me that ever since I had it put in he noticed my hair shedding more and the aggressive mood swings. This whole experienced ruined so much for me. I quit school even though I was so close to graduating because of the stress and my job was put on the line as I kept missing work due to feeling disoriented or like I was going to pass out. I literally thought I was terminally ill and the doctors just couldnt figure it out claiming that it was just stress. I rearranged my entire life to be "stress free" to no avail. I'm trying to have it removed but it will be two months before I can see an OB. I hope my hair will grow back and I cant get back to normal.
LBishopMay 2, 2011 at 7:19 pm
I have had the mirena in for a llittle over a year now. I started the weight gain approximately 4months after having it. I have tried every diet possible and seems I gain more everyday. I recently went to the doctors for an annual exam and made the decision to have the mirena removed. I was asked by the doctor is it was possible that I lost it along the way. I was in disbelief, then wondering coul[Show More]I have had the mirena in for a llittle over a year now. I started the weight gain approximately 4months after having it. I have tried every diet possible and seems I gain more everyday. I recently went to the doctors for an annual exam and made the decision to have the mirena removed. I was asked by the doctor is it was possible that I lost it along the way. I was in disbelief, then wondering could I be pregnant and thats why i'm gaining weight. I had to have an ultrasound in order to ensure it was still there. It was just embedded in the wall of my uterus. Now I have to have surgery to have it removed. Im not sure how long its been there but all the papers of side effects and things that could possibly go wrong depending how long its been embedded. Not happy with this birth control and would not recommend it to anyone.
BritneyApril 28, 2011 at 2:00 pm
I had Mirena in for a few months and then found out I was prego.... I must say we were very shocked and I was VERY depressed about it... I was losing my insurance at my employment and I thought having this would be great and NO WORRIES!!! Guess I was WRONG..... I called doctor and the didnt believe me... but I made and appt anyways and I SURE WAS pregnant!!!! Ok so what so we do??? leave it[Show More]I had Mirena in for a few months and then found out I was prego.... I must say we were very shocked and I was VERY depressed about it... I was losing my insurance at my employment and I thought having this would be great and NO WORRIES!!! Guess I was WRONG..... I called doctor and the didnt believe me... but I made and appt anyways and I SURE WAS pregnant!!!! Ok so what so we do??? leave it in or take it out... I choose to have it removed and doctor thought that was the best decision.. So I did a few days of pain and bleeding and thought I was gonna miscarry I didnt.. NOW THE STRESS WAS FINDING INSURANCE!!! I have to have c sections so i needed to find something I cant afford to pay for that.... I ended up having to quit my job and get medicaid for pregnancy... I ended up having my son June 28,2005 and i wouldnt change it in the world I LOVE HIM TO DEATH... But it sure did make things very hard!!!! This device when i hear people talk about it I SAY DONT DO IT!!!!!! My friend this week had to have hers out do to problems with it... Serious problems!!! Please take this off the MARKET!!!!!!!!
wesApril 26, 2011 at 4:04 am
My wife had to have hers surgically removed last yr and the doctor told her she will never had kids again. She is 24 yrs old. She recently got pregnant after a yr and just had an emergency DNC after 3 months of pregnancy this past Easter. Strongly convinced this product has put a burden on our lives. Angry.
RachelApril 21, 2011 at 3:00 am
I have had the mirena for almost 2 years, i have been experiencing left sided weakness in my upper extremity, tingling, cramps so bad that i feel as if i am giving birth to my mirena. Recently i found out that i have a "low lying cecum" where my colon has enlarged and is now sitting on top of my bladder and uterus. iam not if sure if it was caused by the mirena, but i didn't have any issues before[Show More]I have had the mirena for almost 2 years, i have been experiencing left sided weakness in my upper extremity, tingling, cramps so bad that i feel as if i am giving birth to my mirena. Recently i found out that i have a "low lying cecum" where my colon has enlarged and is now sitting on top of my bladder and uterus. iam not if sure if it was caused by the mirena, but i didn't have any issues before the mirena. i almost have been experiencing depression, shortness of breath, generalized weakness, hair loss, and hair growth on my face, an increase in acne and oil on my face; and i have never had acne. i have made an appointment with my ob and will be removing the mirena on wednesday in hopes that some of the problems will be alleviated. i will be having surgery on my enlarged colon in a couple weeks, is there anyone else out there that has experience anything else close to this? thank you!
nApril 18, 2011 at 1:44 am
i had the mirena put in on jan 28th. asked my doc twice to remove it- she said no and instead gave me seasomale birth control to take in addition to help with spotting. I said i really did not feel well. she said it would pass. well i ended up in the local hospital last week and it was subsequently removed by my doc and have been ging down hill steadily since. I feel like i'm losing my mind. I am [Show More]i had the mirena put in on jan 28th. asked my doc twice to remove it- she said no and instead gave me seasomale birth control to take in addition to help with spotting. I said i really did not feel well. she said it would pass. well i ended up in the local hospital last week and it was subsequently removed by my doc and have been ging down hill steadily since. I feel like i'm losing my mind. I am weepy and desperate all the time. i feel hopeless. is there any detox anything i can do ? I am seriously suffering the mirena crash. please help- anyone
AshliApril 18, 2011 at 1:37 am
Gosh!! This form of birth control has definitely impacted many woman in a awful way! I have only had the iud for 3 months. Luckily I had my yearly pap and when I did my Dr. Checked for my string and when he did he could not find anything. I since then have had a xray and it is in my abdomen. I would not recommend this to anyone! I too was told that is form of birth control was the way to go! I [Show More]Gosh!! This form of birth control has definitely impacted many woman in a awful way! I have only had the iud for 3 months. Luckily I had my yearly pap and when I did my Dr. Checked for my string and when he did he could not find anything. I since then have had a xray and it is in my abdomen. I would not recommend this to anyone! I too was told that is form of birth control was the way to go! I wish I was informed more!
DeniseApril 15, 2011 at 4:01 pm
Just so we're clear... I will be specific with my dates: I had the Mirena insterted on July 25 2008. Aside from the mood swings, no sex drive, pain during intercouse, acne, lower abdominal pain.. etc... In January of 2011, my hair started coming out in HANDFULS! I went and had blood work done immediately thinking i had leukemia or thyroid cancer.. My blood work came back normal.. Two days la[Show More]Just so we're clear... I will be specific with my dates: I had the Mirena insterted on July 25 2008. Aside from the mood swings, no sex drive, pain during intercouse, acne, lower abdominal pain.. etc... In January of 2011, my hair started coming out in HANDFULS! I went and had blood work done immediately thinking i had leukemia or thyroid cancer.. My blood work came back normal.. Two days later, I went to the gynecologist and discussed my situation with her, and she confirmed that it was my Mirena that was causing it... She immediately removed it. She also said that my hair should stop falling out within 10 to 14 days.. It did'nt.. I now have about 10% of my hair left.. With no sign of it ever stopping.. I go back today to see what other damage this has done to my body... Take this with a "grain of salt" if you will... But you've been warned!! I cannot tell you how depressing it is to have no hair:((
GlennaApril 12, 2011 at 10:52 pm
Thank goodness I was smart enough to do my research on this thing! I had my Mirena in for just over 18 months and I too had all the same symptoms. Zero libido, extreme fatigue, foggy headed and dizzy all the time, pubic acne (never had acne in my life!), brittle thinning hair, pain and a weight gain of 25 lbs with no change in diet or exercise. I too was attributing the symptoms to being a mom [Show More]Thank goodness I was smart enough to do my research on this thing! I had my Mirena in for just over 18 months and I too had all the same symptoms. Zero libido, extreme fatigue, foggy headed and dizzy all the time, pubic acne (never had acne in my life!), brittle thinning hair, pain and a weight gain of 25 lbs with no change in diet or exercise. I too was attributing the symptoms to being a mom of two small children. I thank God that others cared enough to post, otherwise I never would have thought it was the Mirena. I had it removed yesterday and like many others my doctor (whom I've seen for years and said Mirena was "living the dream") was suddenly rude to me and acted like I was crazy. I told her that I was 100% sure i wanted this thing out and she must have asked me at LEAST six times if I was sure. What is the deal with these doctors??? NEVER in my life have I told a Dr. I was unhappy with a perscription and they tried to talk me into continuing to use it!! Did these doctors get some kind of kick back that they now have to return because it was removed??? I don't get why she was so insistent that I keep it. Something's rotten here and I sure hope that someday they get to the bottom of it!!
MariaApril 12, 2011 at 6:35 pm
It appears that some of the information above couldn't possibly be correct. A woman who had the Mirena put in in August of 2005 should have had it out by now (it's been over 5 years) and any side effects she's experiencing may be due to that fact. There is also another woman who is quoting dates from 2011 that have not occurred yet (August?). It is hard to take this blog seriously when seeing [Show More]It appears that some of the information above couldn't possibly be correct. A woman who had the Mirena put in in August of 2005 should have had it out by now (it's been over 5 years) and any side effects she's experiencing may be due to that fact. There is also another woman who is quoting dates from 2011 that have not occurred yet (August?). It is hard to take this blog seriously when seeing information like this!!! There could be other reasons why some women experienced loss of fertility. Just because you got pregnant easily before doesn't mean that you will the 2nd or 3rd time. Kidney stones or gallstones are very common also and they can cause similar pain as what some of the women are describing. STD's are more common than one would realize and Mirena certainly won't protect against those. Personally, I have had Mirena in for 4 years now (after having 2 children) and have not had any problems. I did have spotting for 6 months after it was inserted which I was not happy about, but then my periods stopped (was told this may happen) and I've been thrilled not to have them. I am currently doing my research about the newest and best birth control methods out there... Will probably go with Mirena again, but still trying to decide. The info on this website certainly concerns me, but it needs to be taken with a grain of salt.
LouiseApril 11, 2011 at 4:44 pm
One major symptom that I have not seen much in the comments is pelvic floor prolapse, which I swear I have. It's causing me to want (and need) to go the bathroom again, right after I just urinated but just a dribble comes out. Also pelvic pain and fullness that is sometimes severe. I just read that one of the therapies for prolapse is estrogen replacement theory, which makes sense if the Mirena is[Show More]One major symptom that I have not seen much in the comments is pelvic floor prolapse, which I swear I have. It's causing me to want (and need) to go the bathroom again, right after I just urinated but just a dribble comes out. Also pelvic pain and fullness that is sometimes severe. I just read that one of the therapies for prolapse is estrogen replacement theory, which makes sense if the Mirena is causing my hormones to be unbalanced. But who wouldve thought that hormones and urination would be linked? I would liked to know if other women have also experienced this. It's like I can never quite pee completely, always feels like there's "more" but it can't come out because my bladder is collapsed so a lower area is closed off from the ureter. I believe this has contributed to the numerous UTI's and yeast infections I have suffered. I have also had the following problems while on the Mirena for the past 6 months: severe crying bouts, can't stop crying for a whole day which is debilitating and embarrassing; severe anxiety attacks that nearly ruined my entire life; difficulty sleeping theough the night, (I wake up in a sweat and anxiety a lot, can't go back too sleep); blurred vision, especially at night; headaches that were so strong it knocks me off my feet and I can't do anything until it passes. And I have never had migraines before this! I was willing to put up with the constant bleeding, but with all the side affects added together, I want to take this thing out. After reading the post-removal horror stories though, I'm anxious about the cysts and heavy bleeding. At this point it would still be better knowing it was out! I wish I had known this expensive device would make my life miserable.
kellyApril 11, 2011 at 1:04 am
I've had the mirena for 4 years. The insertion was pretty much painless. I had 3 to 4 months of irregular bleeding and spotting. Off and on I have had spontaneous period with the exception of the last 8 or 9 month in which I've had none. I was in love with the mirena! But in the last several months I have had an increase in anxiety to the point in just about resigning from my supervisory posi[Show More]I've had the mirena for 4 years. The insertion was pretty much painless. I had 3 to 4 months of irregular bleeding and spotting. Off and on I have had spontaneous period with the exception of the last 8 or 9 month in which I've had none. I was in love with the mirena! But in the last several months I have had an increase in anxiety to the point in just about resigning from my supervisory position. I have also had an increase in weight and fatigue that is unpredictible from day to day. I have an increase in muscle aches that feel like I'm getting the flu. While my symptoms are not to the severity of some on this site. I am certainly going to see if I am in menopause. I just turned 41 about 3 weeks ago. I do not have a family history of early menopause. I am glad that I do not want anymore children, but not happy of the idea that I may have to begin worrying about osteoporosis and breast cancer sooner than expected. I have a family history of breast cancer and have been getting my mammograms now for several years. Guess it's time to see my GYN!!!
KimApril 6, 2011 at 9:07 pm
I had the Mirena inserted after my second son in 2008 at the advice of my obgyn because of the limited side effects. I also had the side effects of headaches, back pain, severe pelvic pain, and lack of interest in sex. My husband also would be uncomfortable during sex because it would stick him. In 2010 I went to the Dr and had the Mirena removed because of the pain and because I was ready to try [Show More]I had the Mirena inserted after my second son in 2008 at the advice of my obgyn because of the limited side effects. I also had the side effects of headaches, back pain, severe pelvic pain, and lack of interest in sex. My husband also would be uncomfortable during sex because it would stick him. In 2010 I went to the Dr and had the Mirena removed because of the pain and because I was ready to try for a girl which was one of my biggest dreams. About a month after having the Mirena removed I began bleeding profusely and after visiting my obgyn we found that the uterus lining was over 3 times the thickness that it was supposed to be so I had to have a dnc and scope put in me. The found 2 pollips and removed them and started me on birth control to regulate my continued profusive bleeding in which did not work so in Jan this year I had to under go a total abdominal hysterectomy. I am only 27 yrs old and the chance of me ever having a daughter of my own has been robbed from me!! When they done the hysterectomy they left my ovaries because of my age but only a month later I began having severe pain and after visiting the er they found a huge cyst on my left ovary. I have never in my life had any type of cyst or female problem prior to this iud! The Dr scheduled me for laposcopic surgery to remove the cyst but when he got in with the scope they had complications and had to again cut me hip to hip because the cyst was entwined within my intestines. They had to remove my left ovary and cyst at that time. I still have my right ovary but I'm so scared of the damage that I know this Mirena has caused me mentally and physically! I have had to be cut hip to hip twice in just 2 1/2 months! I have lost my job, my car, and my life!!!! It has caused such a strain on my family and between me and my husband because I am in constant pain and he has to pick up all the slack! The Mirena has ruined my entire life!
RosaApril 4, 2011 at 7:35 pm
I had Mirena put in after my 4th child about 3 months after giving birth about 5 months ago. I first started with the heart palpitations and headaches. Went to the doctor and she said just to give it some time for my body to get used to it so I did. a couple months later I feel worse and my mind is cloudy and can't even think straight every single day. Then then the past few weeks I have been [Show More]I had Mirena put in after my 4th child about 3 months after giving birth about 5 months ago. I first started with the heart palpitations and headaches. Went to the doctor and she said just to give it some time for my body to get used to it so I did. a couple months later I feel worse and my mind is cloudy and can't even think straight every single day. Then then the past few weeks I have been having pain in my heart like someone is squeezing it and last night it got worse and the left side of my face got numb I swore I was having a heart attack. I went to the ER and they ran a bunch of tests and still they came out normal but I sure didn't feel normal. The Dr. said it could be related to anxiety and/or indigestion which I NEVER experienced in the past and was always able to handle my stress levels. I have an appointment this week to have it removed along with going over my ultra sound for bad pelvic pain I was having which I also believe was due to the Mirena.
JacquelyMarch 29, 2011 at 6:18 pm
I got the IUD placed 2006,when it was placed I experienced awfully bad cramping,it feels like you are in labor that went on for 3 monthes,I went in to see my doctor he said everything was in place so I left it alone the pain eventually went away.So know here it is 2011I went to get my IUD removed last Monday my doctor could not get it out,so he rescheduled a appointment for next week which was ye[Show More]I got the IUD placed 2006,when it was placed I experienced awfully bad cramping,it feels like you are in labor that went on for 3 monthes,I went in to see my doctor he said everything was in place so I left it alone the pain eventually went away.So know here it is 2011I went to get my IUD removed last Monday my doctor could not get it out,so he rescheduled a appointment for next week which was yesterday,know I am told I have to have surgery because they still could not remove it.they did the ultra sound and said it went through my uterus and its possible I would not be able to carry anymore children so I am forced to get my tubes tied
PetrinaMarch 28, 2011 at 3:10 am
My Mirena was inserted in 2008 and I wouldn't recommend it to others. I think the side effects far out-way the benefits. I've been suffering from mood swings, backaches, headaches, pain in the nerve of my tooth, acne on varies parts of my body, and again did I mention the ridiculous mood swing I have for what seems to be no apparent reason. I am overwhelmed by the smallest things that didn't u[Show More]My Mirena was inserted in 2008 and I wouldn't recommend it to others. I think the side effects far out-way the benefits. I've been suffering from mood swings, backaches, headaches, pain in the nerve of my tooth, acne on varies parts of my body, and again did I mention the ridiculous mood swing I have for what seems to be no apparent reason. I am overwhelmed by the smallest things that didn't use to overwhelm me.
AleidaMarch 28, 2011 at 2:41 am
I am in total shock reading all of these stories! This is actually my second Mirena IUD. I had one inserted six weeks after my first child because of some of the reasons stated prior, my doctor recommended it saying it was a great alternative to the pill and it had no real side effects. I now know that everything I experienced then and am now experiencing all over again have to be due to this t[Show More]I am in total shock reading all of these stories! This is actually my second Mirena IUD. I had one inserted six weeks after my first child because of some of the reasons stated prior, my doctor recommended it saying it was a great alternative to the pill and it had no real side effects. I now know that everything I experienced then and am now experiencing all over again have to be due to this thing. I am extremely moody, any little thing irritates me and I have fits of uncontrollable anger. I remember when I went to my OBGYN the first time I had the IUD and told her about my symptoms she immediately prescribed Prozac and said it was post partum. But it all is so clear now. I have the same moodiness now and the same uncontrollable anger! I have also had lower back pain that I would go to a chiropractor for due to the fact that some days it was hard for me to even walk. I have not gained any weight but I attribute that to being in the gym five times per week for almost two hours and that is just to maintain my current weight, I cannot any weight. I have also experienced sharp pains in my crotch/pelvic area and extreme fatigue. I have no problem falling asleep but staying asleep is unlikely. It is hard for me to make it through the day without wanting to go home and get in bed. I have taken B12 supplements but nothing helps. I have had the current IUD in since January of 2009 and tomorrow first thing, I am calling my OBGYN to get an appointment to have it removed. It is so weird how I never attributed any of these symptoms to the IUD, I guess I trusted my doctor instead of researching it further.
sarahMarch 17, 2011 at 6:14 pm
I had the mirena inserted in Oct of 09 and was told it was amazing. Yeah right! Never had a cyst in my life until I got that, and would have these pains right before my period was about to come where they would send me to the floor in so much agony that I couldn't breathe and my bf had to rush me to the ER where they told me I had a bunch of cysts, and that they were rupturing! I gained like 50lbs[Show More]I had the mirena inserted in Oct of 09 and was told it was amazing. Yeah right! Never had a cyst in my life until I got that, and would have these pains right before my period was about to come where they would send me to the floor in so much agony that I couldn't breathe and my bf had to rush me to the ER where they told me I had a bunch of cysts, and that they were rupturing! I gained like 50lbs and hated to have sex! I had it removed in June of 2010 for two reasons, 1 bc it was hurting and two bc we wanted to have a baby. In those 8 mos the mirena had already decided to attach itself to my uterus and caused a tiny scar. After getting pregnant they found that I had gallstones which I blame solely on the mirena as well because I never had a problem until I got that thing!!!
KeishaMarch 15, 2011 at 7:07 pm
My story is a little different than others. I got my first 1ud (mirena) after my daughter was born in 2007. Got if removed December 4, 2008. I got pregnant 26 days later Dec. 31. I guess I must have ovulated twice cause I got pregnant with twins. WOW. 6 weeks after the twins were born I got a second IUD put in. I was going to physical therapy cause my pelvic bone hurt so bad I couldn't even push a[Show More]My story is a little different than others. I got my first 1ud (mirena) after my daughter was born in 2007. Got if removed December 4, 2008. I got pregnant 26 days later Dec. 31. I guess I must have ovulated twice cause I got pregnant with twins. WOW. 6 weeks after the twins were born I got a second IUD put in. I was going to physical therapy cause my pelvic bone hurt so bad I couldn't even push a laundry basket across the floor with my foot without horrible pain in my crotch/pelvic area. My husband came home one day from work saying they had a discussion on the radio about mirena and he thought I should look into it. Now I have found all this info. YIKES...now thinking about what to do. I also have a pinching feeling in one of my ovaries every so often. WEIRD.
Lisa O'BrienMarch 14, 2011 at 5:31 pm
I have a friend that has an IUD that's traveled and embedded itself into her Uterus, which brought me to this site; after reading many of your comments and some other research regarding IUD's; specifically the one's made of "Copper" it appears to me that many of you may be suffering from Copper toxicity symptoms which include: •Acne •Allergies •Hair loss •Anemia •Anorexia •Anxiety •Attention defi[Show More]I have a friend that has an IUD that's traveled and embedded itself into her Uterus, which brought me to this site; after reading many of your comments and some other research regarding IUD's; specifically the one's made of "Copper" it appears to me that many of you may be suffering from Copper toxicity symptoms which include: •Acne •Allergies •Hair loss •Anemia •Anorexia •Anxiety •Attention deficit disorder •Arthritis •Asthma •Autism •Candida overgrowth •Depression •Dysmenorrhea •Male infertility •Prostatitis •Fibromyalgia •Migraine Headaches •PMS •Chronic infections •Insomnia •Racing thoughts •Neuralgia (nerve pain) •Sciatica •Hypertension •Hypothyroidism •Schizophrenia •Bipolar (Manic Depression)
MelissaMarch 12, 2011 at 6:01 pm
I had the Mirena inserted in 2008, recommended by my OB/GYN. She said all her patients were extremely happy with it and had no complaints. I was doing okay until 2009 when I had SEVERE abdominal pains and the DR found a grapefruit sized ovarian cyst. Had to have surgey to have it removed, six months later the pain returned. I knew it was another cyst and refused to go through another surgery, [Show More]I had the Mirena inserted in 2008, recommended by my OB/GYN. She said all her patients were extremely happy with it and had no complaints. I was doing okay until 2009 when I had SEVERE abdominal pains and the DR found a grapefruit sized ovarian cyst. Had to have surgey to have it removed, six months later the pain returned. I knew it was another cyst and refused to go through another surgery, so I decided to have the Mirena removed. When the DR tried to remove it, she could not. It was lodged in my uterus (which she said was another reason for the pain), so she needed to insert a scope to take it out. That procedure was a quick 30-second procedure and I was much better afterwards. But now my Insurance refuses to pay for the IUD removal. The DR charged my insurance $5000! For this simple procedure, a procedure I HAD TO HAVE, because she suggested I get this IUD. I've already paid to have it inserted, have surgery and to have it removed. The Mirena has caused me so much financial and physical distress. And the DR continues to push it on her other patients.
AudrieMarch 9, 2011 at 6:38 am
I have had the Mirena in for 25 months now, Dr. Recommended this bus birth control after my 1st daughter was born. Approximately 3-6 months after having it inserted the Sharp unbearable lower abdominal pains began. These pains are daily sometimes three times a day lasting a painful minute to two minutes each time. I have been to the WE three times regarding these pains, one time they stated the [Show More]I have had the Mirena in for 25 months now, Dr. Recommended this bus birth control after my 1st daughter was born. Approximately 3-6 months after having it inserted the Sharp unbearable lower abdominal pains began. These pains are daily sometimes three times a day lasting a painful minute to two minutes each time. I have been to the WE three times regarding these pains, one time they stated the reason was scar tissue from my c-section, next reason was unexplained, and last they say was from a yeast infection. I know there is something seriously wrong with me and glad to know thst i am not going crazy! I was healthy with no medical problems prior to using the Mirena. I still have it inserted and am looking to remove it soon. So scared that it ain't be found or i will have horrible side affects after removal. I have the weight gain. fatigue, moodiness as well but more seriously is tge sharp pains that at times have caused me to stop in my teacks and drop to my knees!
MistyMarch 7, 2011 at 2:20 am
I am so appalled at finding this site and reading all of these horrific experiences because what my OB/GYN also told me was an incredible form of birth control. I feel for every woman who has ever used Mirena, it is life altering and Bayer as well as the MD's pushing this contraceptive as a "great" form of birth control should be held responsible. I had Mirena put in after the birth of my last chi[Show More]I am so appalled at finding this site and reading all of these horrific experiences because what my OB/GYN also told me was an incredible form of birth control. I feel for every woman who has ever used Mirena, it is life altering and Bayer as well as the MD's pushing this contraceptive as a "great" form of birth control should be held responsible. I had Mirena put in after the birth of my last child in July 2003, I didnt know the small changes that began shortly after such as the start of migraines, depression, and anxiety were possibly from my Mirena, as these were never side effects I had been informed about. I didn't realize how this intrauterine device would change my life until my major problems began in January 2007. It started with excruciating lower abdominal and back pain that truly felt like labor contractions and the pain did not ever seem to let up. I went to my OB/GYN who did an MRI, he thought he saw a kidney stone. He sent me to a Urologist and here began the many months of painful tests and surgerys. They started by putting a stent in my ureter to dilate it and help me pass the supposed stone he found, two weeks later still in excruciating pain and no stone passing they removed the stent. So they then figure out the "stone" was just a calcification outside the ureter and not the cause of my pain. I was sent back to my gyno, I was given more MRI's and ultrasounds and when nothing could be found and my IUD was seen to still be in the correct position on ultrasound I was given narcotics and told to go home we do not know. From January 2007 until April of 2007 this was my life narcotics and a heating pad, I like the rest of these women could not do my job as a nurse and was on medical leave. I could not take care of my kids because I could not move, I was depressed and in pain 24 hours a day. Finally I went to another gynocologist in another town and he decided to go in Laproscopically, amazingly he did start finding the root of the pain. He first found a blood filled tumor on my left fallopian tube the size of a baseball, as well as large cysts inside and outside my left ovary. He had to remove my fallopian tube and ovary on the left side due to the scarring and damage that was done to them. After this surgery, pain still occurred, so this gynocologist wanted to take out my Mirena and test it for infection, up until this point my own gynocologist had not simply thought of this as an option. The results were shocking after testing the contraceptive he informed me it came back positive for Ecoli and that my entire uterus was full of Ecoli as well and I had to be on a series of antibiotics to cure this infection. As a nurse I am well aware that over time Ecoli can cause potential death, just astounding that this product is still allowed on the market. Everything seemed okay after both these treatments until the winter of 2009 and then the pain began again. In March of 2009 I finally did not have any other choice left but to have a full hysterectomy due to the fact that I had so much scarring on my uterine walls and the cysts on the outside and inside of my right ovary had gotten so large it was best to remove the ovary as well as my uterus. I was only 31 yrs old at this time and I want Bayer to know that I never had fertility problems or female problems before having this intrauterine contraceptive implanted as a choice of birth control without obviously being given all the serious side effects. Sadly my choice was taken away as to whether or not to have more children, I am so thankful that I did not have this put in before I gave birth to the two beautiful children I do have. This needs to be taken off the market so that there does not have to be anymore posts added to websites such as these.
JDMarch 6, 2011 at 6:33 pm
I am a 46 year young mother of one. Prior to Mirena I was in great shape, physically fit, slender working out with lots of energy and Fertile, regular 28 day cycle. I entered into a new relationship over a year ago and was extremely fertile. I was concerned about going back on the pill. My good friend who is a nurse said get the IUD it will be the best thing ever. I did not think to ask her w[Show More]I am a 46 year young mother of one. Prior to Mirena I was in great shape, physically fit, slender working out with lots of energy and Fertile, regular 28 day cycle. I entered into a new relationship over a year ago and was extremely fertile. I was concerned about going back on the pill. My good friend who is a nurse said get the IUD it will be the best thing ever. I did not think to ask her what kind (now I know she has the copper one if I had only looked into this further!) I had Mirena inserted in March 2011. By June I was suffering with bouts of what my OBGYN diagnosed as Group B Strep and Yeast. The pain in my pelvic region was intolerable. I also was have foggy brain, extreme fatigue, heart palpitations, nightsweats, and blurred vision. In August 2011 I had the IUD removed.I had begun working with a Naturopath in August to help alleviate the symptoms. Post IUD I have fought infections for six months, been on countless rounds of antibiotics and antifungals treatments. I continue to have foggy brain, extreme fatigue, mood swings, night sweats, heart palpitations, memory issues, blurred vision, phantom pains in my pelvic region like cramping and most recently have developed severe bloating that makes me looks like I’m pregnant. The worst is now the OBGYN is saying I am Premenopausal. My Naturopath was able to give me supplements three months out in November which that my period back and we tested and I was not premenopausal. Yet one month later no period again for the last three months and my Naturopath is thinking I am premenopausal . This devastates me because I was healthy and fertile pre Mirena. I have always wanted another child. Now thanks to Mirena, I may not ever get that opportunity. MY OBGYN got so fed up at not being able to control the infections she sent me to a Infectious Disease Specialist. He put me on a month long supply of antibiotics! I have another very close friend who is much younger who has lived through the same ordeal and nightmare. Although she suffered extreme depression in addition to all the symptoms I described. She luckily had her IUD taken out within three weeks, yet even today and testing and detox she Is not better and possibly premenapausal.
QuanitraMarch 2, 2011 at 4:45 pm
I am a user of Mirena for about 1 1/2 yrs now and for about 8 mths or so I have been feeling exhausted, having night sweats, insomnia, headaches, nausea, anxiety attacks, depression, abdominal pain, pelvic and anal pain, weight gain almost 50 pounds that I can't get rid of no matter what I do, back pain, chest pain, breast tenderness etc. I thought I was dieing from cancer or something. I constant[Show More]I am a user of Mirena for about 1 1/2 yrs now and for about 8 mths or so I have been feeling exhausted, having night sweats, insomnia, headaches, nausea, anxiety attacks, depression, abdominal pain, pelvic and anal pain, weight gain almost 50 pounds that I can't get rid of no matter what I do, back pain, chest pain, breast tenderness etc. I thought I was dieing from cancer or something. I constantly went from doctor to doctor getting tests done and not finding out what was wrong until I did further research about Mirena and saw all the side effects. I hope that to much damage wasn't done to my body I am only 28yrs old I am getting this thing removed ASAP!! God bless all the woman who have dealt with this situation. I wish you all the best.
KatieFebruary 28, 2011 at 2:04 am
I had a Mirena inserted in Dec. 2010. My doctor sold me on this "miracle" birth control. I have had continuous bleeding since. I have inquired many times only to be told that "bleeding" was normal. 7 days ago, I was rushed to the OR because I had an ectopic pregnancy and my fallopian tube ruptured. I lost both the embryo and right fallopian tube. My IUD – missing. I am now waiting on the result o[Show More]I had a Mirena inserted in Dec. 2010. My doctor sold me on this "miracle" birth control. I have had continuous bleeding since. I have inquired many times only to be told that "bleeding" was normal. 7 days ago, I was rushed to the OR because I had an ectopic pregnancy and my fallopian tube ruptured. I lost both the embryo and right fallopian tube. My IUD – missing. I am now waiting on the result of my xray to find out if I will be need surgery again this Spring to remove it.
TammyFebruary 25, 2011 at 8:23 am
I got the Marina October 2009 because I had heavy periods. I was told there was no side affects besides puncturing your uterine wall when inserting it or that it could slip out. As of now I still hve the Mirena and am estatic that come March 2, 2011 I will finally have this foreign object out of me. It has been a disaster!!!! Extreme weight gain, I look pregnant!! My breast went from a 36 D t[Show More]I got the Marina October 2009 because I had heavy periods. I was told there was no side affects besides puncturing your uterine wall when inserting it or that it could slip out. As of now I still hve the Mirena and am estatic that come March 2, 2011 I will finally have this foreign object out of me. It has been a disaster!!!! Extreme weight gain, I look pregnant!! My breast went from a 36 D to a 36G!! I am extremely exhausted most of the time. I have been spotting constantly since I got the Mirena inserted, oh yes they did say that could happen and not to call them until it went over a year. If I knew this would I would be spotting daily for a year and a half I would have bought stock in panty liners. I am now starting to have lower back pain, and I am assuming it is the enormous breast and weight gain that is putting to much pressure on my back. I just had a pap smear and hmmmm, I just got notice yesterday it is abnormal, and I am positive it is not vd. The V.A. can not remove this damn thing because if they could it would have already been out. If this appointment some how gets cancelled, I will flip out, I am about ready to rip it out myself!! I want my lfie back!!
JenniferFebruary 24, 2011 at 6:41 pm
I had mirena inserted Sept 2010, had it removed yesterday Feb 23, 5 months into having it, I felt like I was going to die. I wanted to die everyday for the past week. I was having continuous hot flashes, missed work for days. Pain in my abdomen and low back. Acne really bad despite many treatments. Depression got worse and worse to the point that I was so tired and just unhappy in general and dis[Show More]I had mirena inserted Sept 2010, had it removed yesterday Feb 23, 5 months into having it, I felt like I was going to die. I wanted to die everyday for the past week. I was having continuous hot flashes, missed work for days. Pain in my abdomen and low back. Acne really bad despite many treatments. Depression got worse and worse to the point that I was so tired and just unhappy in general and disinterested. Unable to play with my 2yr old son because my body pain got to the point of just doing nothing really was even painful. Once removed i was given premarin (conjugated estrogens) to help deal with the side effects of hot flashes, fatigue, etd, until the progestin levels come down to where I can just be on my natural cycle.
RachelFebruary 19, 2011 at 3:42 pm
I had the Mirena IUD inserted in November 2007 and removed in September 2009. I was told the insertion would be painless and I didn't need anyone to accompany me as a driver. While the Dr. was inserting the IUD I told her how much pain I was in and that I was going to either be sick or pass out. I did get sick and she informed me that if I couldn't handle the pain she would have to stop. Before I [Show More]I had the Mirena IUD inserted in November 2007 and removed in September 2009. I was told the insertion would be painless and I didn't need anyone to accompany me as a driver. While the Dr. was inserting the IUD I told her how much pain I was in and that I was going to either be sick or pass out. I did get sick and she informed me that if I couldn't handle the pain she would have to stop. Before I was even able to decide she continued inserting the IUD. I was in no condition to be driving 45 minutes by myself and was in so much pain I could barely stand. I later found out that the Dr. was supposed to offer me a local anistetic to help with the pain....well obviously that wasn't the case for me. In September 2009 I started experiencing horrible pains and felt like the IUD had moved and was pinching me. Shortly after having pains I started my period, which I had not had in over 2 years since I went straight from the Depo shot to the IUD. After having heaving bleeding and serious pains for two weeks I had it removed. When I went to the Dr. she did an internal ultrasound to see if it had moved as well as look for any cyst. The Dr. was unable to see my left ovary or the IUD. After searching for nearly 20 minutes, she decided to remove it by the string connected to the end of it. Thankfully it was still there but the pain was intense while she slowly yanked it out. In November of 2009 I started bleeding the first part of the month and continued for 28 days. Since my period was never really normal prior to using the IUD I wasn't to concerned. I should have seen a Dr. immediately. It is now February 2011 and I have yet to have another period since November 2009. I did see another Dr. back in August 2010 and test were done to determine that I am no longer ovulating and will probably never have children. The IUD has reuined my life. I do not have any children of my own and want the experience for myself.
LourdesFebruary 17, 2011 at 4:03 am
To Kimberly: The Mirena removal did not hurt. It took a couple of minutes. A day after, I started my period and it's been heavy for the last week. However, the bloating was gone immediately and the lower abdominal pain diminished.
SabrinaFebruary 17, 2011 at 4:01 am
I had the Mirena IUD inserted October 2009 and had it removed 10 months later. I have had quite a few of the same issues as the ladies posting here and on other sites. I never imagined when I had this device implanted that it would cause me to have mood swings and food cravings like being on PMS 24/7, that I would feel tired all the time, gain about 50 pounds, that I would have numerous problems w[Show More]I had the Mirena IUD inserted October 2009 and had it removed 10 months later. I have had quite a few of the same issues as the ladies posting here and on other sites. I never imagined when I had this device implanted that it would cause me to have mood swings and food cravings like being on PMS 24/7, that I would feel tired all the time, gain about 50 pounds, that I would have numerous problems with back, joint and abdominal pain, or that intimacy would be negatively impacted by lack of drive, dryness and pain/discomfort for both me and my husband. August 2010 we decided enough was enough and I had this god-awful thing yanked out and went right back on old-trusty Depo-Provera, what I should have done after the birth of my little girl in the first place.
LourdesFebruary 17, 2011 at 3:56 am
I have had the Mirena IUD for close to 5 years.Finally removed it and although I am bleeding profusely (which was expected), I feel a better knowing Mirena is out of me. I was told by my doctor that it was amazing birth control with no side effects and that it was almost painless to insert. It was not painless however, the problems were just about to start... Some of the side effects I exper[Show More]I have had the Mirena IUD for close to 5 years.Finally removed it and although I am bleeding profusely (which was expected), I feel a better knowing Mirena is out of me. I was told by my doctor that it was amazing birth control with no side effects and that it was almost painless to insert. It was not painless however, the problems were just about to start... Some of the side effects I experienced were irregular heart beats, lower abdominal pain, lower back pain, breast pain, horrible migraines, total fatigue, lack of energy, loss of sex drive,, and extreme bloating. Within a couple of months, I gained 28 lbs that I cannot shed, no matter what I try. In addition the above mentioned side effects, I was diagnosed with high blood pressure and I had deep depression, moodiness, bouts of crying, joint and muscle pain, had several pelvic and yeast infections and acne (at 48!!!). It has been 4.5 years of true hell. I'm still 28 lbs heavier and hope eventually to go back to my original weight. Something should be done so the COMPLETE side effect information is communicated to women BEFORE the Mirena is inserted. There should also be a complete warning issued. I also think that doctors are educated and given ALL the information. I am slowly going back to the way I was... an instant at a time. Still get bad migraines and some of the pains are still nagging. Hopefully once my system expelled all the hormones completely, I will feel better.
KimberlyFebruary 14, 2011 at 2:01 pm
I have had my Mirena since 2005. It did hurt when they put it in, the pain lasted a few days. It felt as if somebody had punched me as hard as they could a few times in my stomach as they were putting it in. I was approximately 135 pounds 6 months after I had it put in. I am now 175 pounds never have I weighed this much except when I was pregnant. My doctor and I have ran every text possible to s[Show More]I have had my Mirena since 2005. It did hurt when they put it in, the pain lasted a few days. It felt as if somebody had punched me as hard as they could a few times in my stomach as they were putting it in. I was approximately 135 pounds 6 months after I had it put in. I am now 175 pounds never have I weighed this much except when I was pregnant. My doctor and I have ran every text possible to see why it is I have gained all this weight and not been able to loose it. I look pregnant. My breast are tender and have enlarged by two sizes. I have back pain and migraines. Although the migraines I have suffered since a teenager they seem to have gotten worse. When I seen this thread I had to wonder if my issues were due to my mirena as well. I need to have mine removed but I am deathly afraid of thee pain I will feel when they remove it.
AmandaFebruary 11, 2011 at 11:15 am
I have had my Mirena for about 2 years now. I have recently started having breast pain. I thought that maybe because of a round of antibiotics to cure strep that maybe it caused a disruption in my hormone level - I don't have regular monthly periods anymore. This lasted for about a week then it went a away for about a week. I am now having the same symptoms and my breasts are full of cysts. I[Show More]I have had my Mirena for about 2 years now. I have recently started having breast pain. I thought that maybe because of a round of antibiotics to cure strep that maybe it caused a disruption in my hormone level - I don't have regular monthly periods anymore. This lasted for about a week then it went a away for about a week. I am now having the same symptoms and my breasts are full of cysts. I have also experienced severe cramping, fatique, insomnia, headaches, frequent urination (UTI-like symptoms without any actual infection), vaginal irritation, and now I am experiencing cyst-like acne, night sweats and hot flashes - I am 37. I knew the cramping was linked to the Mirena but it was not until googling "can mirena cause cysts in breasts" that I came up on this site as well as others that linked my other symptoms to my IUD. I have gone to the doctor of the last couple of years without being able to leave with any real diagnosis. I have been left feeling that it just is the way that you are supposed to feel when you start getting close to 40. Unfortunately, my insurance will not be in affect for another 2 months. I will definitely have it removed as soon as I am insured again. I will repost if there are any adverse side effects that may arise after removal. I am now very afraid of infertility and any other potential long term effects from this horrible device that needs to be recalled. I did my homework before the insertion and was told that it was very safe and that my gyno had been prescribing IUDs for years. At that time any IUD blogs were mostly for the coil and outdated information. Now, there is up to date blogs and far worse symptoms being mentioned than I could find before.
ValerieFebruary 5, 2011 at 3:42 pm
I had it put in January 2009 and ended up having it removed by my primary care doc sometime in March, I think. I have never been the same. If there is a lawsuit going on, I'd really like to join in. I tell every female I know that is thinking about getting it NO, it will ruin your life, it did mine.
JasminFebruary 3, 2011 at 1:09 am
I had the Iud Mirena inserted in December 2007. It was a painful insertion, now I constantly get yeast infections. I have terrible mood swings, I feel depression, I also have punching feeling on both of my overies. The headachs don't seem to go away and the lower back pain is annoying. Forget the sex drive that went out the door months after I had it put in. I've had it in now for almost 3 years [Show More]I had the Iud Mirena inserted in December 2007. It was a painful insertion, now I constantly get yeast infections. I have terrible mood swings, I feel depression, I also have punching feeling on both of my overies. The headachs don't seem to go away and the lower back pain is annoying. Forget the sex drive that went out the door months after I had it put in. I've had it in now for almost 3 years and im thinking of getting it taken out. What should I do? Help!
JessicaFebruary 2, 2011 at 9:35 pm
After seeing a commercial last night I was very concerned with the painful side effects I have had with the Mirena. I had mine put in a month after the birth of my third son, (feb 2005) in which I was not told about the very bad side effects I saw on the TV last night. I was never told it could cause infertility, Uterus Perforation, Migration, Expulsion, and Infection, along with all these things [Show More]After seeing a commercial last night I was very concerned with the painful side effects I have had with the Mirena. I had mine put in a month after the birth of my third son, (feb 2005) in which I was not told about the very bad side effects I saw on the TV last night. I was never told it could cause infertility, Uterus Perforation, Migration, Expulsion, and Infection, along with all these things I have experienced cramping, shooting pains, migraines, loss of libido, spotting of a funny color after sex, cramps with no periods, tiredness, sharp shooting pains in both my ovaries area and my anus area that drop me to my knees and ect........ I could go on forever most of which I have seen listed in all the woman above. I do not have insurance and have not been able to find anyone to take it out. I went to the ER last year with severe pains and cramping and discomfort and was told by the ER doctor it was not in his job description to take those things out, so I left in frustration. I do not have a job but now after reading this I am going to use my tax return to pay the 200 to get it taken out in the hopes that with all I have experienced with it they will be able to do so without surgery. If I am going to have to have surgery to take it out I do not ow what I am going to do. I am very scared and sick and tired of feeling this way but at least now I know it is not just me and I am not crazy these painful feelings are real and not just my imagination. I think these law suits are in most need and should be happening because if I was told all of the things they knew now about it I would have never never never taken it. I am also most concerned because me and my fiancée have three boys and since our youngest is in school we wanted to try for a girl after I finished school. Now I am afraid that will not happen. I am with you ladies I do not recommend this to anyone and feel very strongly about the fact this thing should be taken of the market!
MeredithJanuary 30, 2011 at 6:14 am
The pain and suffering from Mirena use, as reported by the women in this site as well as others, is appalling. I had the IUD placed in Sept. 2010, approximately six weeks after the birth of my son. I have experienced EXTREME fatigue, upper back/shoulder PAIN, cramping, side pain, no period, and look five months pregnant. I have been to chiropractor for several weeks. In past week,TWO trips to ER[Show More]The pain and suffering from Mirena use, as reported by the women in this site as well as others, is appalling. I had the IUD placed in Sept. 2010, approximately six weeks after the birth of my son. I have experienced EXTREME fatigue, upper back/shoulder PAIN, cramping, side pain, no period, and look five months pregnant. I have been to chiropractor for several weeks. In past week,TWO trips to ER for irregular heart beat, Blood tests are fine and no history of heart problems. I will have the Dr remove the Mirena ASAP!!!!!!
DebJanuary 27, 2011 at 6:58 am
I had the Mirena for 3 weeks back in 2008, and was perfectly healthy before having it. I'm a very athletic woman (44 yrs old). I had one previously with no problems, but the second one almost killed me. I felt like I had aged 40 yrs within a few days of insertion. I had severe backaches, muscle soreness and weakness, headaches, anxiety attacks, heavy sensation in my throat, extreme fatigue and t[Show More]I had the Mirena for 3 weeks back in 2008, and was perfectly healthy before having it. I'm a very athletic woman (44 yrs old). I had one previously with no problems, but the second one almost killed me. I felt like I had aged 40 yrs within a few days of insertion. I had severe backaches, muscle soreness and weakness, headaches, anxiety attacks, heavy sensation in my throat, extreme fatigue and tiredness, dizziness, etc... It's been just over 2 yrs since I had it removed, and i still don't feel 100%. The biggest issue I still have is muscle weakness, numbness and tingling in my hands and legs. I'm currently trying acupuncture and detox foot pads to see if it helps. My doctor still refuses to believe that Mirena could have caused all these problems. She referred me to an internal medicine dr, possibly checking for MS, but nothing came up. The acupuncturist could see that internally, my body was unbalanced and I had some horrible infection previously. To my surprise, many women I have spoken to about this IUD, they too had bad experiences.
KariJanuary 26, 2011 at 10:57 pm
I am finding that alot of women are reporting another issue with Merina. The issue being premature menapause caused by Merina. I am one such woman. I had Merina for five years and had it removed last March. I am now having the following symptoms; headaches heart palpations night sweats hot flashes decreased libido vaginal dryness mood swings. I went to my primary doctor and he stated I am in [Show More]I am finding that alot of women are reporting another issue with Merina. The issue being premature menapause caused by Merina. I am one such woman. I had Merina for five years and had it removed last March. I am now having the following symptoms; headaches heart palpations night sweats hot flashes decreased libido vaginal dryness mood swings. I went to my primary doctor and he stated I am in premature ovarian failure and I am only 38. I am finding alot of other woman who were put on Merian are also in full menapause at an early age. This side effect is not listed by Merina as well. I am wondering if there is a lawsuit in the works agaist Merina dealing with premature menapause. Have any of you had this problem as well? I am going to my obgyn in three weeks to confirm this diagnosis. Thanks
ShellyJanuary 25, 2011 at 3:39 pm
Hi I have had mine for about 3 years now and I have been to the doctor several times since they checked it the couple weeks after getting it and they have not been able to remove it without causing me serious pain and I need to have it removed via surgery. I am afraid that it may be attached to my uterus somehow. I unfortunately have not been able to have it removed yet due to no insurance. I like[Show More]Hi I have had mine for about 3 years now and I have been to the doctor several times since they checked it the couple weeks after getting it and they have not been able to remove it without causing me serious pain and I need to have it removed via surgery. I am afraid that it may be attached to my uterus somehow. I unfortunately have not been able to have it removed yet due to no insurance. I like so many people on here have multiple symptoms which include the following..... very bad abdomanal pain like a full blown contractions sometimes weight gain (no change in diet or excercise) headaches like every day some worse then others tired all the time hurts during sex no energy spot after sex all the time abdominal pains lower back pains reverse periods (3 weeks on and 1 week break) constant vaginal odor very low sex drive I need to get this out and don't know how without insurance. If anyone has any ideas please shoot them my way........I want my life back in every way!!!!
TiffanieJanuary 20, 2011 at 6:07 pm
I had Mirena inserted about 8 weeks after I had my 4th baby. The procedure (insertion) was AWFUL and the pain from just having it put in lasted close to a week. It took a few months and some research to connect the dots and decide that my severe back pain, ovarian cysts, headaches, tremendous weight gain, hair loss, crazy new hair growth(in very embarrassing places), acne, depression and the lis[Show More]I had Mirena inserted about 8 weeks after I had my 4th baby. The procedure (insertion) was AWFUL and the pain from just having it put in lasted close to a week. It took a few months and some research to connect the dots and decide that my severe back pain, ovarian cysts, headaches, tremendous weight gain, hair loss, crazy new hair growth(in very embarrassing places), acne, depression and the list goes on, were connected to this HORRID device. I have had it out now for about 18 months and I am sad to say that I am still suffering from most of the effects. The back pain is the worst, almost debilitating, and I've been to several specialists and had MANY tests( 2 MRI's, 2 CT's, numerous X RAY's, cytso, and even a colonoscopy) and no one can find the problem. I want my life back!! I can never get back the time I have lost with my babies because I hurt too bad to get off of my heating pad, and my kids will always look back on this time of their lives and remember mama being sad, crying, sick, hurt, and in their eyes lazy (b/c I CAN'T get up and play). The makers of this thing are responsible, however, we ALL trusted our doctors and they let us down as well. I believe that my OBGYN is as responsible for my problems as Bayer because not only did she push this on me, she dismissed every single side effect when I came to her for help. "That is NOT a side effect of Mirena." "I can't find any problem that would lead to your complaint." etc. I would LOVE to know just exactly what kind of throw-back these Dr.'s are getting from the manufacturer. I want this THING off the market, I want more research done, and I want these people to publicly recognize and admit that THEY WERE WRONG and that this device and the Dr's that push it are responsible for severly altering the quality of life for many, many women.
SabrinaJanuary 19, 2011 at 5:22 am
Up until today, when I read this site, I thought I was loosing my mind and was ready to voluntarily commit myself. I got Mirena 25 months ago and my life has been a living hell all but the first 3 months. Physically, I have experienced cramps, sharp abdominal pain, back pain, drowsiness, migraines, dizziness, blurred vision, spontaneous lactation, sore/swollen breast, drops in blood pressure, l[Show More]Up until today, when I read this site, I thought I was loosing my mind and was ready to voluntarily commit myself. I got Mirena 25 months ago and my life has been a living hell all but the first 3 months. Physically, I have experienced cramps, sharp abdominal pain, back pain, drowsiness, migraines, dizziness, blurred vision, spontaneous lactation, sore/swollen breast, drops in blood pressure, loss of appetite, nausea, pain and spotting after sex, metallic body oder, and hair loss. Mentally, I have suffered from anxiety attacks, violent mood swings, episodes of uncontrollable crying that last anywhere from a few hours to a few days and severe depression. I visited my GYN more that usual in the past 2 years with complaints as the symptoms arose. She treated the symptoms and contributed them to the amount of stress I was encountering at work and school. I was prescribed pain medications and anti-depressants but not once did she suggest removing the device. Since then, I have had to leave my job, as a result lost my house and am more miserable than I would have ever imagined. I can honestly say that Mirena has ruined my life. I was prescribed Mirena because of my very long, super heavy, painful periods and the promise of being able to live a full life without the worry of being bed confined 7-10 days of every month. Instead, I am an unemployed, walking time bomb with limited social interactions. I have no children and now fear that once this device is removed I will never be able to.
Proud MommaJanuary 17, 2011 at 8:07 pm
The Mirena almost destroyed my life, but after TONS of searching, I found a natural detox that took my symtoms away (mine were AWFUL anxiety attacks, no libido, back pain, abdominal pain, mood swings...you name it.) I'm better now, but I'm on a "mission" to help as many women as I can find relief. I have all of the information on a blog that I've written. The address is mylifeafternirena(dot)bl[Show More]The Mirena almost destroyed my life, but after TONS of searching, I found a natural detox that took my symtoms away (mine were AWFUL anxiety attacks, no libido, back pain, abdominal pain, mood swings...you name it.) I'm better now, but I'm on a "mission" to help as many women as I can find relief. I have all of the information on a blog that I've written. The address is mylifeafternirena(dot)blogspot(dot)com. I pray that it works for you as it did me. This device is a MONSTER, and SHAME ON YOU, BAYER for doing it to so many of us....knowing this....and STILL not doing anything about it.
ClaudiaJanuary 16, 2011 at 9:19 pm
hi, I had mirena insert on february of 2007, six months after the birth of my second child, i had some side effects like lower libido, mood swing,etc but the major thing was when I was diagnosted, with breast cancer on september of 2008, about 18 months after a had insert mirena , I was 35 at time, no family history of cancer.I just remember that after mirena i was not feeling so well, besides t[Show More]hi, I had mirena insert on february of 2007, six months after the birth of my second child, i had some side effects like lower libido, mood swing,etc but the major thing was when I was diagnosted, with breast cancer on september of 2008, about 18 months after a had insert mirena , I was 35 at time, no family history of cancer.I just remember that after mirena i was not feeling so well, besides the side effects that i alrealdy mention. I was at de doctors almost every month with diferent litlle things , and I was always very tired. My OB and my primary Doctor were telling me that I had nothing to worry about, That the way that I was felling was normal for a mother with 2 young children, and at age of 35 they don't send you for mamograms, I just thank God that a tumor pop out, with gave me a change to get treatment, and be to alive today .
SarahJanuary 13, 2011 at 3:50 pm
helllo out there! maybe someone out there can help me out i am 20 yrs old i have two beautiful children but i got mirena about a year and a half or so ago and since then i have had the worst stomach pain any one could endure lower back pain and pelvic pain and every time i go to my doctor they tell me nothing is wrong with you and send me back home and the pain continues im to the point that i don[Show More]helllo out there! maybe someone out there can help me out i am 20 yrs old i have two beautiful children but i got mirena about a year and a half or so ago and since then i have had the worst stomach pain any one could endure lower back pain and pelvic pain and every time i go to my doctor they tell me nothing is wrong with you and send me back home and the pain continues im to the point that i dont know what to do or say..
NicoleDecember 27, 2010 at 3:59 pm
hi i have had mine for about 2 1/2 years now and i have not been to the doctor since they checked it the couple weeks after getting it i have multiple symptoms in which i do not know is the cause but they are as followed very bad abdomanal pain like a full blown contraction, rapid weight gain like a pound a week headach like every day some worse then others tired all the time syrup smell dow[Show More]hi i have had mine for about 2 1/2 years now and i have not been to the doctor since they checked it the couple weeks after getting it i have multiple symptoms in which i do not know is the cause but they are as followed very bad abdomanal pain like a full blown contraction, rapid weight gain like a pound a week headach like every day some worse then others tired all the time syrup smell down below and when i am peeing hurts during sex no energy acasional harp pains in my butt that actually makes me jump up spot after sex sometimes blood in urine and stool at times frequent peeing stomach pains lower and upper back pains loss of breath tender breast under the boob sore muscles all the time i went to the ER two times the past year first time i had a lump on my rib and they took x rays and said oh its probbably on infection and gave me penasillion and sent me on my way it help for a little while then a couple months latter i went in and they said all my syptoms are of MONO they did the test and it was neggative but said they come back negative somethimes we still think it is MONO and gave me penacillion to help with any infection and pain killers to ease the pain it helped for a couple weeks then the sypmtioms slowely started coming back then about four weeks ago i had a three day epasode to where i went home i was in so much pain then i laid in bed and when i got up to go to the kitchen to get some water i calapsed to the ground i was in so much pain and couldnt breath. the pain not as bad but lasted for three days.
AprilDecember 21, 2010 at 11:35 pm
i had the iud mirena put in 3mths after i had my child. I have had mood swings dizzines head pain ive gained 50 lbs i with no change in diet i had the iud removed and it hasent changed anything now me and my husband want another baby we have been trying for 2yrs with dr help with no sucess i fear that my child will be the only one that will ever have i feel like somthing has been stole from me
MonicaDecember 20, 2010 at 8:55 pm
I have an appointment to have my Mirena removed in January after having it in place for three years. I was told when I had it inserted that 90% of women experienced no menstral bleeding whatsoever with MIrena - yet I bled for the first three months non stop. Then I had what I called "reverse periods" which were on for three weeks of bleeding, and a one week break. After a few months, I develope[Show More]I have an appointment to have my Mirena removed in January after having it in place for three years. I was told when I had it inserted that 90% of women experienced no menstral bleeding whatsoever with MIrena - yet I bled for the first three months non stop. Then I had what I called "reverse periods" which were on for three weeks of bleeding, and a one week break. After a few months, I developed SEVERE acne that could not be treated with any prescription or over the counter medicine. Around that same time, I started to get migraine headaches (which I did not associate with Mirena until later). I also experience extreme breast swelling and tenderness for about 1 1/2 weeks out of every month. It gets so bad that I have to wear a sports bra over my regular bra, it hurts to lift my arms over my head, and I have to put ice packs on my breasts at night to ease the pain. The reason I have waited three years to get this removed is because my doctor told me most side effects are only temporary. The excessive bleeding and severe acne did go away after several months, but it has been a long time and the headaches and breast swelling persist. It's time for me to get my life back. I just hope I don't suffer as some of these others have with adverse effects after removal!
JanieceDecember 11, 2010 at 4:19 am
I was on Mirena since January of 2010. By November of 2010, I was pregnant and Mirena is nowhere to be found. Dr. and I are now in the process of scheduling an Xray in order to make sure it has not traveled to my liver. I wouldn't recommend Mirena to anyone.
krystalDecember 9, 2010 at 2:23 pm
i had the mirena put in in 2008 and it was very very painful going in , the cramping after nad the bleeding was awful. i had it until this year when i had enough of the pain , i had two misscarriges with it because part of it fell out and i got pregnant , i went to my obgyn and he tried to take it out in office and that was painfull and couldnt get it out , then had to do surgry to get it out and [Show More]i had the mirena put in in 2008 and it was very very painful going in , the cramping after nad the bleeding was awful. i had it until this year when i had enough of the pain , i had two misscarriges with it because part of it fell out and i got pregnant , i went to my obgyn and he tried to take it out in office and that was painfull and couldnt get it out , then had to do surgry to get it out and it left me with three scars because it was embeded in to my utrus wall and was stuck ! this is the worse birthcontrol ever !! i am looking in to a lawsuit for it.
LoneseDecember 2, 2010 at 4:46 am
I was uneasy about getting the Mirena. I have always used the "pill" at the beginning, but I had to quite that because it made me sick. But after reading about Mirena I thought it would be the right thing for me to get. I had just had my 4th child and I did not want any more. I have had the Mirena since Feb. of 2007 and it is suppose to last 5 years. I have experienced severe migraines, lower back[Show More]I was uneasy about getting the Mirena. I have always used the "pill" at the beginning, but I had to quite that because it made me sick. But after reading about Mirena I thought it would be the right thing for me to get. I had just had my 4th child and I did not want any more. I have had the Mirena since Feb. of 2007 and it is suppose to last 5 years. I have experienced severe migraines, lower back and pelvic pain, breast soreness, moodiness, depression, crying for no reason, weight increase, ovaries feeling like they are about to pop, just so much discomfort. Sometimes during intercourse with my husband he would tell me it would poke him. Not what I exspected at all from Mirena. I am running to get this out as soon as possible.
NLasherNovember 30, 2010 at 5:17 pm
I had Mirena inserted in Mar 2007 and started having severe migraines in 2008. In Feb of 2010 I had it removed due to sever pelvic cramps and bleeding, not to mention still suffering with migraines. In May 2010 I was put on medical leave because my bladder was not working right and I had what they thought were kidney stones but were actually very large ovarian cysts. In July 2010 I ended up having[Show More]I had Mirena inserted in Mar 2007 and started having severe migraines in 2008. In Feb of 2010 I had it removed due to sever pelvic cramps and bleeding, not to mention still suffering with migraines. In May 2010 I was put on medical leave because my bladder was not working right and I had what they thought were kidney stones but were actually very large ovarian cysts. In July 2010 I ended up having to have a complete hysterectomy not by choice. I have never had female problems in the past and have 4 beautiful children. I truly feel that now at the age of 36 I am was put into surgical menopause all because I was told what a wonderful form of birth control Mirena is and how it would change my life. I didnt expect my life to change like this. Now I am on artificial hormones for who knows how long.
MelissaNovember 24, 2010 at 3:58 am
I have have the mirena in place for nearly 3 years now. I have seveer night sweats, loss of libido, depsression and a genearl feeling of weakness. I have done considerable research online and believe that only the mirena could be the culprit. Again like many of the other posts I've read, After numberous tests with no results and feeling like you're crazy., I have made an appointment to have m[Show More]I have have the mirena in place for nearly 3 years now. I have seveer night sweats, loss of libido, depsression and a genearl feeling of weakness. I have done considerable research online and believe that only the mirena could be the culprit. Again like many of the other posts I've read, After numberous tests with no results and feeling like you're crazy., I have made an appointment to have mine removed. I do not feel optimistic for what's to come. I have a sick feeling that at 33, Mirena has forced me into early menopuase. I don't want money from a lawsuit. I just want my life back.
J. PierceNovember 23, 2010 at 2:34 pm
I had Mirena inserted around April 2003 or sooner - not certain of the date. Like other women, I had severe abdominal pain after the insertion which made it difficult for me to walk without severe discomfort. My doctor told me that it was because the Mirena had to "settle" in my uterus. She took me out of work for 1 week so that I could have some recovery time out of work. I worked in correctio[Show More]I had Mirena inserted around April 2003 or sooner - not certain of the date. Like other women, I had severe abdominal pain after the insertion which made it difficult for me to walk without severe discomfort. My doctor told me that it was because the Mirena had to "settle" in my uterus. She took me out of work for 1 week so that I could have some recovery time out of work. I worked in corrections and standing for extended periods of time and excessive walking was unberable. Ultimately, the pain dissapated and I was able to resume normal activities. I had the Mirena IUD removed around October 2006 with the hopes of becoming pregnant. As of January 2010, I have been unable to become pregnant. I decided to get back on BC again and decided to use Orthotricycline LO. I took this BC from Jan 2010 to March 2010. I decided to stop the BC because my husband and I wanted to try again to get pregnant. So far as of Nov. 23, 2010, we have still been unsucessful. I am so concerned now that Mirena may have caused me to become infertile and that is why I am now unsuccessful with conceiving. I mean it has been 4 years with no success. The only way to verify this is to address my concerns with my OBGYN and see if everything is functioning properly in order to rule out Mirena as a contributing factor. It does concern me since I had my daughter with no problems prior to Mirena.
Tracie RNovember 23, 2010 at 11:59 am
I had the Mirena inserted 8 weeks after giving birth. It dislodged in less than three months, was stuck under - and perforated my uterus. Had to have laproscopic surgery to remove it, which was another week off of work in addition to the fourth months off for maternity due to complications. Nearly lost my job over all the time off. Luckily I didn't have it in long enough to suffer any of the OTHER[Show More]I had the Mirena inserted 8 weeks after giving birth. It dislodged in less than three months, was stuck under - and perforated my uterus. Had to have laproscopic surgery to remove it, which was another week off of work in addition to the fourth months off for maternity due to complications. Nearly lost my job over all the time off. Luckily I didn't have it in long enough to suffer any of the OTHER side effects I read about.
J QNovember 21, 2010 at 8:09 pm
I had the Mirena in for a year and 2mths. The Mirena was so pain I could not walk, play with my son, I was depressed all the time. I could not sleep unless I take pain medications. It got to the point where the Doctor that inserted the Mirena in for me to get a Hysterectomy to be done with the pain. I am now still dealing with pain and infections I got the Mirena took out back in October 2009. Som[Show More]I had the Mirena in for a year and 2mths. The Mirena was so pain I could not walk, play with my son, I was depressed all the time. I could not sleep unless I take pain medications. It got to the point where the Doctor that inserted the Mirena in for me to get a Hysterectomy to be done with the pain. I am now still dealing with pain and infections I got the Mirena took out back in October 2009. Someone Please Help me. GOD knows I need it.
M. RodriguezNovember 20, 2010 at 10:58 pm
I had Mirena inserted 1 1/2 year ago, I had many side affects, but worst off all i just found out Im pregnant with IUD, and it canot be removed because of the risk of abortion. I'm scared to death because I know all the risk that comes with being pregnant with IUD in place, 50% chance of miscarrying or even perforation that can cause infection and even worse death. This is happening to many wome[Show More]I had Mirena inserted 1 1/2 year ago, I had many side affects, but worst off all i just found out Im pregnant with IUD, and it canot be removed because of the risk of abortion. I'm scared to death because I know all the risk that comes with being pregnant with IUD in place, 50% chance of miscarrying or even perforation that can cause infection and even worse death. This is happening to many women not just 1/1000 there is absoultley a malfunction in this device!!
EboniNovember 13, 2010 at 6:46 pm
I had the Mirena inserted December 2009 and after months of discomfort and irregular bleeding, I had it removed in May 2010. A month later, I began bleeding large amounts of blood along with blood clots. The following month, i found myself having to get rushed to the hospital where I received 2 blood transfusions, as well as being told that I was experiencing Progestin withdrawal, I was diagnose[Show More]I had the Mirena inserted December 2009 and after months of discomfort and irregular bleeding, I had it removed in May 2010. A month later, I began bleeding large amounts of blood along with blood clots. The following month, i found myself having to get rushed to the hospital where I received 2 blood transfusions, as well as being told that I was experiencing Progestin withdrawal, I was diagnosed with Dysfunctional Uterine Bleeding, and Anemia, and they have found fibroids in my uterus. The ER doctors put me on another form of birth control to try to counteract with the side effects of the Mirena, but the bleeding has not stopped. Now several doctors have told me that if this next form of birth control doesn't work, the next step will have to be a hysterectomy (I am only 24). Since then, I have had to receive more transfusions, and have been told that I am bleeding to death every month. I am now bed ridden and unable to work to provide for my family.
AmandaNovember 11, 2010 at 4:11 am
I have had the Mirena IUD for about 3 years now. I went to my obgyn to get it inserted after being told that it was amazing birthcontrol with no side effects and that it was painless to insert. Getting the mirena put in was extremely painful for me! I could barely walk afterwards and had trouble driving home. I was bedridden for days bc the pain was so bad. The side effects i experienced initially[Show More]I have had the Mirena IUD for about 3 years now. I went to my obgyn to get it inserted after being told that it was amazing birthcontrol with no side effects and that it was painless to insert. Getting the mirena put in was extremely painful for me! I could barely walk afterwards and had trouble driving home. I was bedridden for days bc the pain was so bad. The side effects i experienced initially were lower pelvic pain, lower back pain, extreme breast pain and awful migranes. Within months i gained 30 lbs with no change to my diet. The other side effects never went away but depression, moodiness, bouts of crying and acne were added to the list. It has been 3 years of hell. Im still 30lbs heavier and have extreme breast and back pain and have also been diagnosed with high blood pressure, joint and muscle pain and have had several pelvic infections that may have left enough scar tissue to not allow me to have any more children. I am a 30 year old mother of one child and i feel like this birth control has ruined my life.