Eligible for a AGGA lawsuit?
Anterior Growth Guidance Appliance (AGGA) Lawsuits Claim Dental Device Destroyed Teeth, Caused Other Permanent Damage
Plaintiffs who have filed Anterior Growth Guidance Appliance lawsuits say the device is not fit for adults, and causes damage to the upper jaw and teeth.
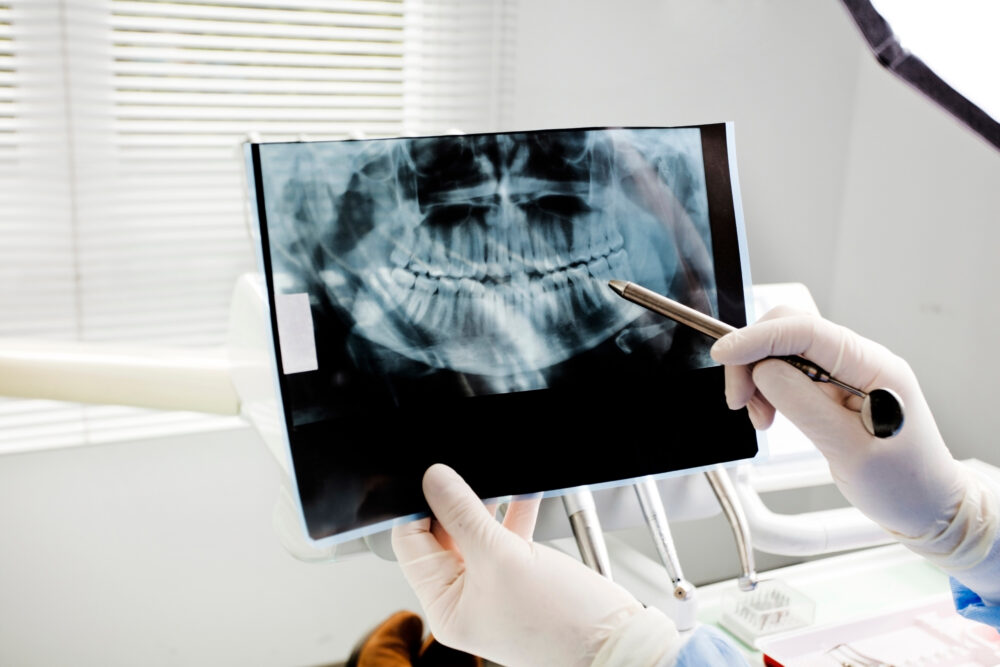
A growing number of AGGA lawsuits have been filed over the past few years, each raising similar allegations that a dental device known as an Anterior Growth Guidance Appliance, or AGGA, destroyed teeth, disfigured their face and caused other serious health problems.
The Anterior Growth Guidance Appliance (AGGA) is a metal, wire insert placed between a patient’s teeth, which is supposed to help remodel or expand an adult’s jaw without the need of surgery. However, over the past three years, at least 20 patients have filed APPA lawsuits against the manufacturers and designers, including Steve Galella, D.D.S, OtrhoMatrix Corp, and John’s Dental Laboratory, Inc.
The dental device is supposed to cause three-dimensional changes in adult jaws, which can move the maxilla forward horizontally over time as much as 10 mm or more. However, plaintiffs indicate that, instead, the device pushes the upper teeth out of their housings, resulting in serious and painful injuries.
The lawsuits date back to at least 2000, indicating that the AGGA device was originally designed for children, who are still growing naturally, but alleging that it is unreasonably dangerous for adults, whose bone growth has stopped.

Learn More About
Problems with the AGGA dental device have caused severe teeth damage and disfiguring injuries. Financial compensation may be available through an AGGA lawsuit.
Learn More About this Lawsuit SEE IF YOU QUALIFY FOR COMPENSATIONOne of the most recent lawsuits was filed on March 3, when Michael Haile and Melanie Pappalardi filed a complaint (PDF) in the U.S. District Court for the Southern District of Indiana.
Haile, of New York, indicates that he was prescribed an AGGA in May 2019, due to problems with his airway and open bite. The device was removed and replaced in February 2021. However, even a year before that, in February 2020, Haile began to suspect the device was causing damage, but was reassured by his provider that his discomfort was a normal part of the procedure.
The lawsuit indicates that use of the AGGA caused significant dental damage, including loosening of anterior maxillary teeth; pain; flaring of the front teeth; gum recession; probably root resorption and alveolar bone loss.
The other plaintiff, Pappalardi, received her AGGA device in August 2020, but began to suffer tooth sensitivity and loosening, and had the device removed in August 2021. She indicates the device caused her front upper teeth to be pushed out of the alveolar bone, and also caused her to suffer nerve damage, extreme tooth sensitivity, and will probably lose teeth because of the device’s side effects.
AGGA Lawsuit Indicates Device Insufficiently Tested on Adults
According to allegations raised in the AGGA lawsuit, the manufacturers for not sufficiently testing the design on adults, and did not considering the difference between adult and child bone growth.
“Dr. Galella and OrthoMatrix, knew or should have known that, while the representations may have been true in regard to the use of AGGA by children (who are still growing naturally), the representations as to adults were unproven, not supported by medical knowledge or science, and were false and materially misleading,” the lawsuit states. “AGGA is unreasonably dangerous to adult patients in whom it is installed, and is not reasonably safe for use by such patients; and, AGGA is not a substitute for jaw surgery for adults.”
The lawsuit presents claims of negligence, breach of warranty, violation of the Indiana Product Liability Act and the Indiana Deceptive Consumer Sales Act.
AGGA Dental Treatment Lawsuits
AGGA dental device injury lawyers are now pursuing lawsuits for individuals claiming the AGGA devices were negligently designed, and that plaintiffs were given false representations about the device’s efficacy and safety. Settlement compensation from an AGGA lawsuit payout may be available for individuals who received an AGGA treatment and suffered any of the following injuries;
- Loosing of upper teeth (anterior maxillary teeth)
- Tooth loss and/or damage
- Tooth or Jaw pain
- Flared teeth
- Gum Recession
- Tissue breakdown around tooth (Root Resorption)
- Tooth socket bone loss (Alveolar bone loss)
- Facial disfigurement
- Nerve damage
- Required corrective surgery
Get more articles like this sent directly to your inbox.
"*" indicates required fields
1 Comments





ResearchSeptember 24, 2023 at 3:33 pm
A female's intra-oral photo of the damage caused by AGGA also shows that an oral surgeon had extracted premolars in accordance with an unproven orthodontic claim that children can not grow forward enough to accommodate all 32 adult teeth. Both AGGA and EXTRACTION ORTHODONTICS DO NOT HAVE ANY PEER -REVIEW, INTER-DISCIPLINARY( ENT, Neurology, gastroenterology, pulmonology) studies to prove that they[Show More]A female's intra-oral photo of the damage caused by AGGA also shows that an oral surgeon had extracted premolars in accordance with an unproven orthodontic claim that children can not grow forward enough to accommodate all 32 adult teeth. Both AGGA and EXTRACTION ORTHODONTICS DO NOT HAVE ANY PEER -REVIEW, INTER-DISCIPLINARY( ENT, Neurology, gastroenterology, pulmonology) studies to prove that they do not disfigure/injure bone, teeth, tongue space, airway, cervical spine, lungs, digestion, and posture. In fact, the female was a professional wind-instrument player, who sought AGGA as a consequence of extractions causing retrognathic arches with her tongue shoved back into her airway. No person should have to endure the severe risks of jaw surgery to correct damage from wilfully negligent, unscientific, falsely promoted extraction ortho, the false hope of AGGA, or other unethical practice. THERE SHOULD BE A CLASS ACTION AGAINST EXTRACTION ORTHO AND AGGA with a requirement that the dental industry do the interdisciplinary peer review that is needed to stop dangerous extractions and resulting snake oil salesmen.