Bard Eclipse IVC Filter Embedded In Internal Organs, Lawsuit Claims

According to allegations raised in a recent product liability lawsuit filed against C.R. Bard, one of the company’s Eclipse blood clot filters embedded inside the internal organs of a South Dakota woman, causing her to suffer severe health complications.
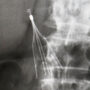
The complaint (PDF) was filed by Amber Nordin in the U.S. District Court for the District of South Dakota on January 12, indicating that she received a Bard Eclipse IVC filter in October 2012. It was subsequently discovered that the small device designed to prevent blood clots from traveling to the lungs had migrated out of position and became lodged in her organs.
The Bard Eclipse is designed to be implanted within the inferior vena cava (IVC) for individuals at rusk of suffering a pulmonary embolism. However, the device and several other retrievable filter designs sold by Bard have been linked to reports of catastrophic and devastating problems for thousands of individuals nationwide.
Learn More About
Design Problems with Certain IVC Filters Linked to Severe Injuries. Lawsuits Reviewed Nationwide.
Learn More About this Lawsuit See If You Qualify For CompensationNordin indicates that after the Bard Eclipse IVC filter embedded in her internal organs, she underwent unsuccessful surgery to remove the blood clot filter in January 2014, but it remains stuck inside her body.
The lawsuit notes that Nordin will face a continuing risk of problems from the IVC filter, including the potential for life-threatening complications and ongoing medical care to ensure the Bard Eclipse filter does not cause further injury.
The case joins a number of similar Bard IVC filter lawsuits filed in recent years, alleging that the devices are unreasonably dangerous and defective. In addition to the Eclipse, similar problems have been reported involving the Bard G2, Bard Recovery and Bard Meridian filter designs.
Given the similar allegations raised in cases filed by individuals nationwide, the U.S. Judicial Panel on Multidistrict Litigation has established coordinated pretrial proceedings in the federal court system, centralizing all IVC filter lawsuits against Bard before U.S. District Judge David G. Campbell in the District of Arizona to reduce the risk of duplicative discovery into common issues in the cases, avoid conflicting pretrial rulings from different judges and to serve the convenience of the parties, witnesses and the courts.
Similar coordinated pretrial proceedings, known as an MDL or multidistrict litigation, have been established for all Cook Celect and Gunther Tulip IVC filter lawsuits, which involve another retrievable blood clot filter design.
As part of MDL cases, it is expected that a series of early trial dates will be scheduled against each of the manufacturers to help the parties gauge how juries may respond to certain evidence and testimony over the long-term risks associated with the IVC filters.
While the outcomes of these early trial dates will not be binding on other claims, they will be closely watched as they may influence eventual IVC filter settlement negotiations
Get more articles like this sent directly to your inbox.
"*" indicates required fields




0 Comments