Bard Recovery Filter Lawsuit Filed After Failure Leads to Open Heart Surgery

A product liability lawsuit has been filed by an Illinois man who experienced problems with a Bard Recovery inferior vena cava (IVC) filter, indicating that the device failed inside his body and caused him to require open heart surgery.
The complaint (PDF) was filed last month in the U.S. District Court for the Central District of Illinois, by Henry Kilver and his wife, Judy. Defendants named in the lawsuit include C.R. Bard, Inc. and their Bard Peripheral Vascular, Inc. subsidiary.
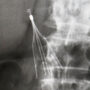
Kilver indicates that a Bard Recovery filter was implanted into his inferior vena cava in January 2005. The small, spider-like device is designed to be placed inside the vein to filter or “catch” blood clots that may travel from lower portions of the body to the heart or lungs, preventing a pulmonary embolism.
Learn More About
Design Problems with Certain IVC Filters Linked to Severe Injuries. Lawsuits Reviewed Nationwide.
Learn More About this Lawsuit See If You Qualify For CompensationAccording to allegations raised in the Bard Recovery filter lawsuit, Kilver’s device failed in early 2011 and the IVC filter migrated to his heart, causing serious and life-threatening injuries.
As a result of the problems, Kilver states that he has had to undergo extensive medical care, including emergency open heart surgery, and that he continues to suffer significant pain and suffering. His wife is included on the complaint for loss of consortium, comfort and society caused by the injuries Kilver suffered.
Lawsuits over Bard Recovery and G2 IVC Filter Failures
The Bard Recovery filter is one of two IVC filters marketed by C.R. Bard that have been the subject of a number of similar lawsuits in recent years, where plaintiffs allege that the device has failed and the legs or struts on the filter have broken off and travelled to other parts of the body. The Bard G2 is a second generation version the Recovery IVC filter, but it has also been plagued by similar problems.
Unlike traditional IVC filters, the Bard Recovery and G2 filters were designed so that they can be removed once the threat of a pulmonary embolism has passed. However, in many cases physicians have allowed the devices to remain in place, which may increase the risk of complications.
According to allegations raised in many of the lawsuits over the Bard Recovery and Bard G2 filter, the manufacturer knew or should have known about the unreasonably high rate of fracture, migration and perforation of the vena cava wall that was occurring after implant, yet failed to warn consumers or the medical community.
In August 2010, the FDA issued an alert about the risk of problems with removable IVC filters, indicating that the agency had received more than 900 adverse event reports associated with the products. Of those reports, 328 involved the IVC filter breaking free and migrating through the body, 146 involved components breaking loose, 70 involved the inferior vena cava being perforated and 56 involved the filter fracturing.
The FDA recommended that doctors remove the filters once the danger of the clot has passed, to reduce the risk of the filters breaking free and traveling through the body.
Two studies published earlier this year raised further concerns about IVC filters, indicating that there is a lack of evidence that the IVC filters are effective, as well as a lack of data on their proper use, resulting in doctors being unsure of how and when to implant them or remove them.
Last year, several Bard IVC filter class action lawsuits were filed on behalf of individuals who received the Recovery or G2 filters, seeking to force the manufacturer to pay for the medical monitoring that patients will need to ensure that the filters have not failed. An estimated 100,000 of the devices have been implanted in patients in the United States.
Get more articles like this sent directly to your inbox.
"*" indicates required fields




0 Comments