Muscle Paralysis Drug Recall Follows ISMP Warning of “Extremely Hazardous Packaging”
Meitheal Pharmaceuticals is issuing a nationwide recall for cisatracurium besylate injections, due to a risk the injections may have been distributed with a dangerous packaging error, which may increase the risk of serious injury or death.
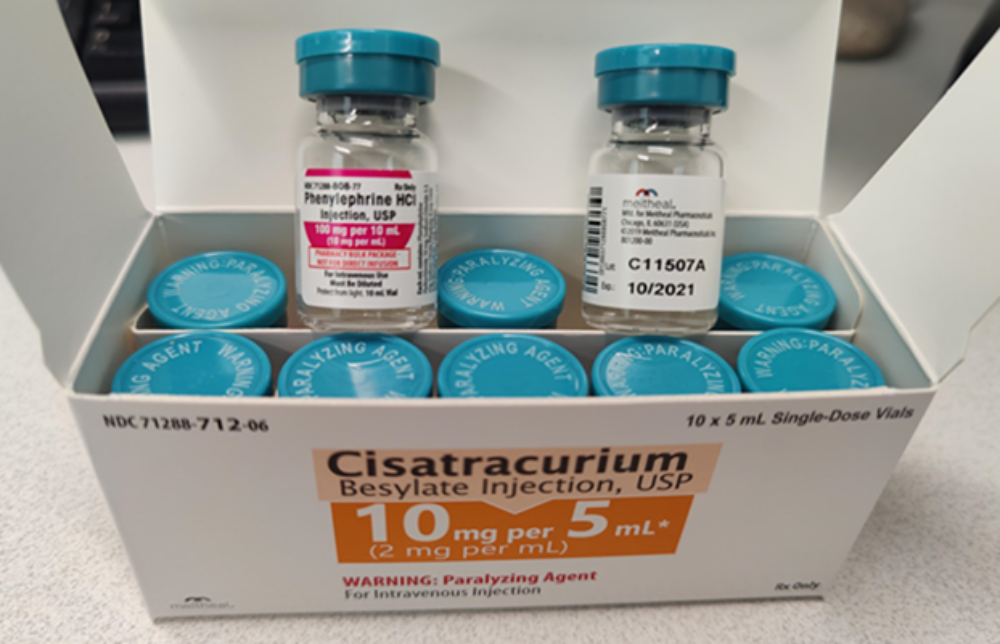
The cisatracurium recall was announced on Wednesday, indicating one lot of 10 mg per 5 mL vials distributed by Meitheal Pharmaceuticals, Inc. may be mislabeled as phenylephrine hydrochloride 100 mg per 10mL.
Cisatracurium besylate is used as a non-depolarizing neuromuscular blocker, which is used to induce muscle paralysis as part of general anesthesia. Patients given this drug must be intubated or ventilated due to the paralysis. However, the labeling error increases the risk of a medication error, where a patient who was meant to receive the blood pressure medication phenylephrine may instead be given the mislabeled cisatracurium, they could suffer from muscle paralysis. This could lead to serious illness or death if left untreated.

Did You Know?
Millions of Philips CPAP Machines Recalled
Philips DreamStation, CPAP and BiPAP machines sold in recent years may pose a risk of cancer, lung damage and other injuries.
Learn MoreThe lack of skeletal muscle relaxation could also cause a hyperadrenergenic state which would lead to elevated blood pressure, heart arrhythmia and cardiac/brain ischemia. Again, this state could lead to serious illness or death.
While the recall was announced January 27 by the FDA and Meitheal, the Institute for Safe Medication Practices issued an alert on January 26, the day before the recall was announced, calling the problem an “extremely hazardous packaging error” requiring urgent attention.
“Due to the nature of this situation and the potential for death if the these vials are used as phenylephrine injection in patients who are not intubated and ventilated, we urge facilities to immediately examine any and all cartons of cisatracurium from Meitheal Pharmaceuticals for this serious packaging error,” the ISMP warning states. “The possibility that any of these vials were actually distributed should also be considered.”
Customers who have purchased the product should not open the carton or use the contents and should immediately separate the products and return the recalled lot.
The recalled lot of cisatracurium besylate vials have a lot number of C110507A and an EXP date of October 2021 stamped on the bottle. The product can be identified as a 5 mL vial topped with a rubber stopper and sealed with an aluminum seal having an Aqua color flip-off seal.
Meitheal stopped shipping the vials to customers August 19, 2020, after the product was distributed to wholesalers nationwide in the U.S. The company has also notified its distributors and customers in writing to arrange for return of all recalled product.
To date, there have been no reports of adverse events linked to the recall
Consumers can contact Meitheal’s Customer Service at 844-824-8426 regarding the recall. If you experience any side effects, the company recommends you contact your healthcare provider immediately.
Adverse reactions should also be reported to the FDA’s MedWatch Adverse Event Reporting program.
Get more articles like this sent directly to your inbox.
"*" indicates required fields




0 Comments