Lawsuits over Cordis OptEase, TrapEase IVC Filters Removed to the Federal Court

As a growing number of Cordis IVC filter lawsuits continue to be filed in California state court over problems with the OptEase and TrapEase devices, the manufacturer is attempting to remove the cases to the federal court system, arguing that the claims constitute a mass tort action that fall under federal jurisdiction.
In a notice of removal (PDF) filed on June 6, Cordis identified at least 8 different complaints filed in the Superior Court of California over the past two months, which each involve complications experienced by large groups of individuals who received a Cordis OptEase or TrapEase IVC filter to prevent blood clots from traveling to their lungs.
The manufacturer indicates that a recent motion filed in California state court to consolidate the cases for pretrial purposes and bellwether trials gives rise to federal jurisdiction under the Class Action Fairness Act of 2005, since it is a mass action in which monetary relief claims involving more than 100 persons are proposed to be tried jointly.
Learn More About
Design Problems with Certain IVC Filters Linked to Severe Injuries. Lawsuits Reviewed Nationwide.
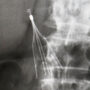
Learn More About this Lawsuit See If You Qualify For CompensationThe Cordis TrapEase and OptEase are inferior vena cava (IVC) filters, which are spider-like devices placed into the vein to “catch” blood clots that may break free in the body for individuals at risk of suffering a pulmonary embolism.
“More than one hundred and fifteen plaintiffs allege that following implantation of their TrapEase or OptEase IVC filters, they may suffer or have suffered harm, such as ‘life-threatening injuries and damages[,] and require[d] extensive medical care and treatment,’ or that they were subject to ‘significant medical expenses, extreme pain and suffering, loss of enjoyment of life, [and] disability,’ among other injuries,” according to the removal notice, which suggests that the claims seek an amount in controversy that exceeds $5 million in the aggregate.
The Cordis OptEase lawsuits and Cordis TrapEase lawsuits raise similar claims to those presented against manufacturers of other IVC filters, including C.R. Bard and Cook Medical, alleging that the devices are prone to tilt out of position, perforate the vena cava or fracture, causing small pieces to travel to the heart and lungs.
There are currently more than 527 Bard IVC filters lawsuits and 383 Cook IVC filter lawsuits pending in the federal court system, which have each been centralized as part of separate multidistrict litigations (MDLs) for coordinated pretrial proceedings, including discovery and a series of early bellwether trials.
The litigation over IVC filters began to emerge in 2010, after the FDA indicated that hundreds of adverse event reports had been received involving the devices, including instances where devices sold by various different manufacturers broke free, migrated through the body, perforated the vein or fractured.
In May 2014, the FDA urged doctors to remove IVC filters within about one to two months after an individual was no longer at risk of suffering a pulmonary embolism, since the risk of problems appeared to be greater the longer the filter was left in place. However, many manufacturers sold the devices for years without adequately informing physicians about the risks associated with leaving them in place.
As IVC filter injury lawyers continue to review and file complaints on behalf of individuals who have suffered migration or perforation problems, it is ultimately expected that several thousand cases will be filed against Cordis, Bard and Cook Medical.
Get more articles like this sent directly to your inbox.
"*" indicates required fields




0 Comments