EU Regulators Recommend HES IV Solution Ban, Groups Call For Similar US Action
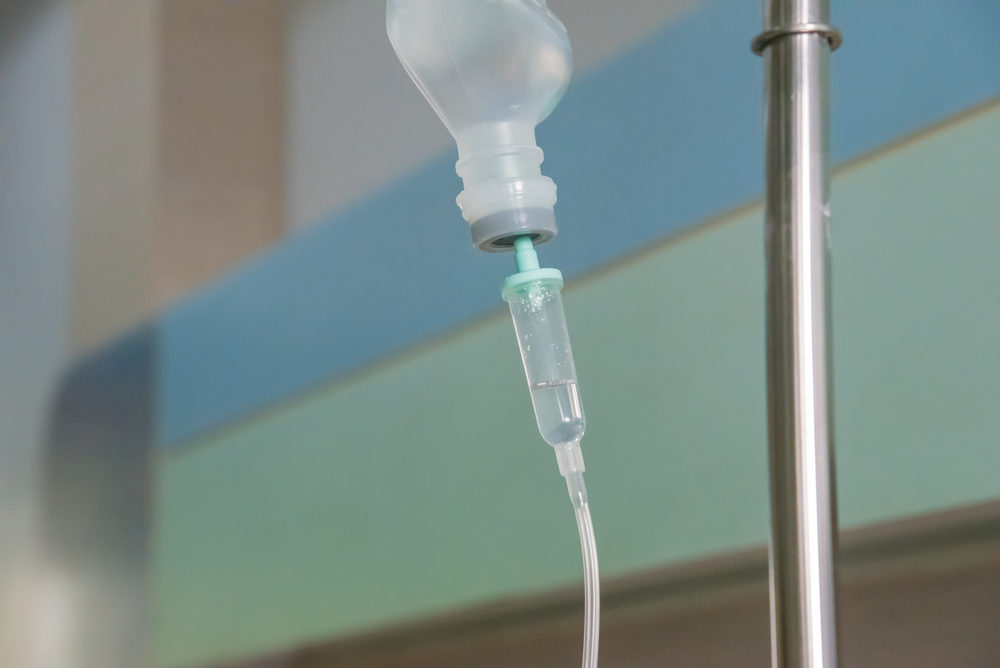
European regulators are calling for hydroxyethyl-starch (HES) solutions to be removed from the market in the EU, due to the risk of kidney problems and death, an action some grass roots consumer protection groups have asked for in the U.S. for years.
On January 12, the consumer watchdog group Public Citizen applauded actions by the European Medicines Agency (EMA) Pharmacovigilance Risk Assessment Committee (PRAC), which recommended HES solutions lose market authorizations last month.
Last year, Public Citizen petitioned the FDA to take the same regulatory action in the U.S.

Did You Know?
Millions of Philips CPAP Machines Recalled
Philips DreamStation, CPAP and BiPAP machines sold in recent years may pose a risk of cancer, lung damage and other injuries.
Learn MoreHES IV solutions are used to boost fluid volume among hospital patients, and it has been on the market since 1972. However, Public Citizen warns that HES remains in the body’s tissues for long periods of time, can reduce blood clotting ability, and has been linked to kidney failure and death.
In 2013, PRAC recommended that HES solutions administered through IVs be restricted, and said they should no longer be used on critically ill patients. However, the group says now that it has further reviewed drug studies on HES solutions, including clinical trials and observational studies, and has received feedback on stakeholders and experts.
“Based on this review, the PRAC has concluded that the restrictions introduced in 2013 have not been sufficiently effective. The Committee explored the possibility of introducing additional measures but concluded that such measures would be ineffective or insufficient,” PRAC said in a press release. “In view of the serious risks that certain patient populations are exposed to, the PRAC has recommended the suspension of the marketing authorisations for HES solutions. Alternative treatment options are available.”
Similar recommendations were made by Public Citizen’s petition, filed with the FDA last February. This summer, a group of 28 experts in intensive care and associated health care fields sent a letter to the FDA supporting the Public Citizen petition.
“The U.S. Food and Drug Administration (FDA) must now follow suit with an immediate ban on HES solutions in the U.S.,” wrote Dr. Sammy Almashat, a researcher with Public Citizen’s Health Research Group. “The EMA PRAC’s new recommendation unequivocally demonstrates that only a complete ban on these dangerous IV solutions will prevent further unnecessary patient deaths and injuries.”
Get more articles like this sent directly to your inbox.
"*" indicates required fields





0 Comments