Lawsuits Over IVC Filter Migration, Perforation Injuries Begin to Mount

C.R. Bard and Cook Medical face a growing number of product liability lawsuits over inferior vena cava (IVC) filters, with claims filed throughout the federal court system alleging that the small blood clot filters may migrate out of position, perforate the vena cava or fracture, potentially causing small metal pieces to travel to the heart or lungs.
IVC filters are often implanted into the vein for individual at risk of blood clots, to “catch” clots that may break free within the body and prevent them from traveling to the lungs, where they may cause a pulmonary embolism. However, several retrievable IVC filter designs introduced by Bard and Cook have been linked to reports of severe and sometimes fatal complications.
Given the similar allegations raised in cases filed by individuals nationwide, the U.S. Judicial Panel on Multidistrict Litigation has established two separate proceedings for complaints brought against each of the medical device makers.
Learn More About
Design Problems with Certain IVC Filters Linked to Severe Injuries. Lawsuits Reviewed Nationwide.
Learn More About this Lawsuit See If You Qualify For CompensationAll Cook Celect and Cook Gunther Tulip IVC filter lawsuits have been centralized before U.S. District Judge Richard L. Young in the Southern District of Indiana since October 2014, and all Bard Recovery, Bard G2 and other Bard IVC filter lawsuits have been centralized before U.S. District Judge David G. Campbell in the District of Arizona since August 2015.
According to the latest case list (PDF) released by the U.S. JPML last week, there are already 172 cases pending in the Cook MDL and another 72 claims pending in the Bard MDL.
As IVC filter injury lawyers continue to review and file complaints on behalf of individuals who have suffered migration or perforation problems, it is ultimately expected that several thousand lawsuits will be involved in the two litigations.
IVC Filter Litigation
All of the lawsuits raise similar allegations that the small devices are unreasonably dangerous, and contain inadequate warnings about the potential injury risks associated with IVC filters
In one of the more recent lawsuits, filed on December 14, a New York woman alleges that she was implanted with a Bard G2 filter in January 2010. However, in May 2015, she began suffering stabbing and burning pains in her abdomen and lower back.
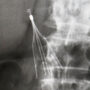
According to the complaint (PDF), a CT scan revealed that her IVC filter had migrated and fractured, causing her pain. However, her doctor told her that removing the broken filter would require a “massive” surgical operation to remove because it had punctured her aorta and possibly her duodenum.
The plaintiff underwent IVC filter removal surgery in September, but faces the risk of future procedures due to complications and has to undergo careful rehabilitation to avoid reinjuring her body while the tissue slowly mends.
As part of the coordinated proceedings in each of the MDLs, it is expected that a series of early trial dates will be scheduled to help the parties gauge how juries may respond to certain evidence and testimony over the long-term risks associated with the IVC filters.
In the litigation over Cook IVC Filters, Judge Young has indicated that a series of four cases will be selected for trials that will go before juries after September 15, 2016. In the more recently established Bard IVC filter litigation, the organizational structure of the MDL is still being established, and a bellwether trial program has not yet been established.
While the outcomes of these early trial dates will not be binding on other claims, they will be closely watched as they may influence eventual IVC filter settlement negotiations
Get more articles like this sent directly to your inbox.
"*" indicates required fields




0 Comments