The FDA announced last week that they are working with the National Eye Institute and the Department of Defense to study the impact on quality of life from Laser-Assisted In Situ Keratomileusis (LASIK), to determine how frequent corrective eye surgery problems are and to identify potential predictors for the complications.
Their review of the problems with LASIK will be divided into three phases. The first phase, which began in July, was the design and implementation of a web-based questionnaire for LASIK patients, which focuses on quality of life problems after LASIK surgery. The second phase will focus on determining quality of life for active military personnel who have undergone LASIK at the Navy Refractive Surgery Center. The final phase will be a similar clinical trial on the general population, which is expected to end in 2012.
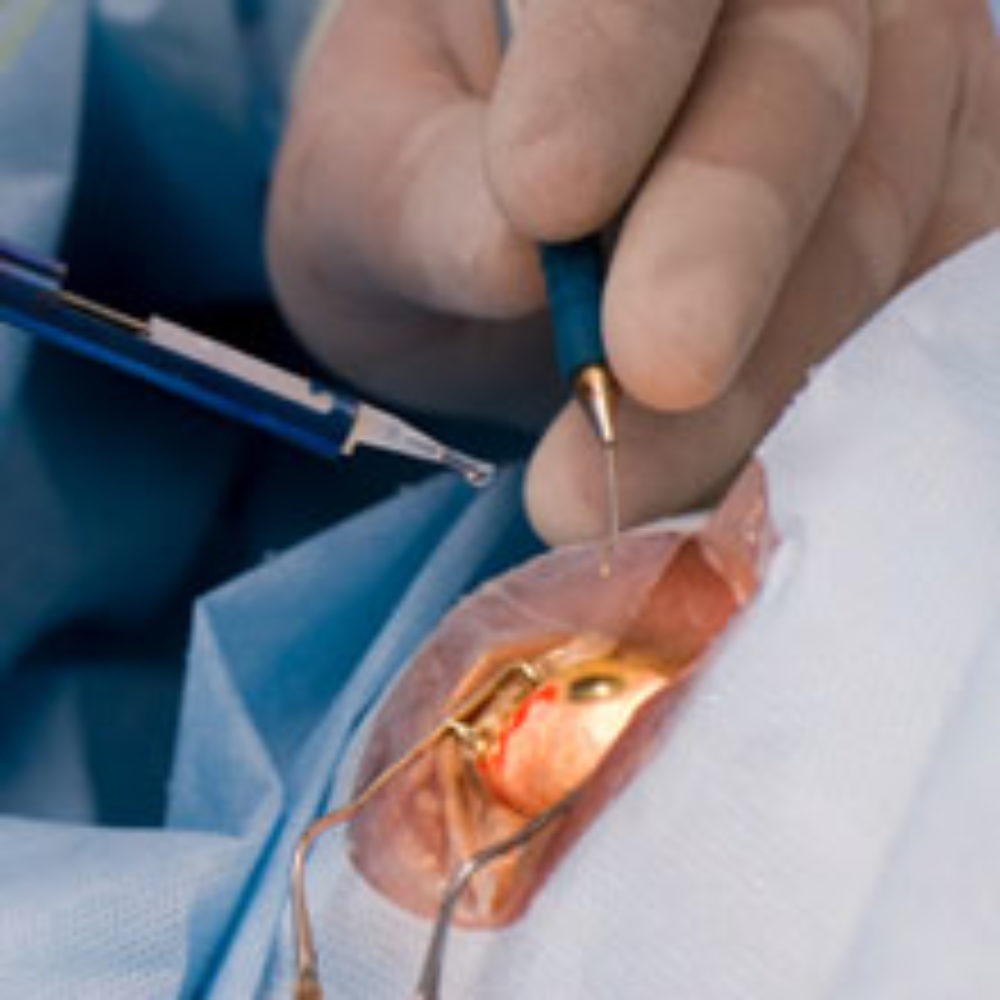
LASIK is a form of eye surgery that uses a laser to improve a patient’s vision and reduce their need to wear glasses or contact lenses. During the eye surgery, a surgical instrument known as a microkeratome is used to cut through the top layers of the cornea to create a flap. A laser beam is then used to reshape the cornea for vision correction and the corneal flap is replaced.

Did You Know?
Millions of Philips CPAP Machines Recalled
Philips DreamStation, CPAP and BiPAP machines sold in recent years may pose a risk of cancer, lung damage and other injuries.
Learn MoreA 2008 American Journal of Ophthalmology report found that additional surgeries were needed for 28% of the eyes treated with LASIK less than 10 years after the initial surgery, due to undercorrection, overcorrection, or regression.
Reports of LASIK problems commonly involve double vision, halos around objects at night and dry eyes. In more severe cases, complications can have a severe impact on an individual’s quality of life.
The FDA hopes that the results of the study will help the agency understand the adverse effects of LASIK, and identify means to reduce the risks for future patients. However, the study also may result in new regulations on the safe use of LASIK if the FDA deems necessary.
In addition to the study, the FDA also announced that it has sent warning letters to 17 LASIK eye surgery centers. The warnings came after FDA inspectors discovered inadequate systems for reporting adverse events suffered by LASIK patients at the centers. Legislation passed in 1990 makes it a requirement that medical facilities of all kinds report all device-related serious injuries or deaths related to the FDA and the device manufacturer.
Although rare, a number of patients have filed Lasik surgery malpractice lawsuits against eye surgeons for complications that were allegedly caused by a failure to follow the appropriate standard of medical care, most commonly involving problems that occur after a doctor fails to properly screen patients out as a potential candidate for the eye surgery.
Get more articles like this sent directly to your inbox.
"*" indicates required fields
2 Comments




NormaOctober 22, 2009 at 9:41 pm
I had Lasix eye surgery 3-25-09--very unhappy- my vision is worse now than before i had surgery a lot of things are wrong ,blurred vision depth vision is very bad.feels grainy.when I set at the Pc my left eye wants to far away & my right eye wants to be up close- if i had known the risks of this type of surgery I would never had it done saw another Dr.& he said it will likely always be this way-so[Show More]I had Lasix eye surgery 3-25-09--very unhappy- my vision is worse now than before i had surgery a lot of things are wrong ,blurred vision depth vision is very bad.feels grainy.when I set at the Pc my left eye wants to far away & my right eye wants to be up close- if i had known the risks of this type of surgery I would never had it done saw another Dr.& he said it will likely always be this way-so never have Lasix done
DeanOctober 19, 2009 at 10:11 pm
My website says it all: www.LifeAfterLasik.com , Dean