Plaintiffs’ attorneys in federal product liability lawsuits involving the use of pain pumps following arthroscopic shoulder surgery, have filed a petition seeking to consolidated all cases for pre-trial litigation as part of a coordinated process known as an MDL, or multi-district litigation.
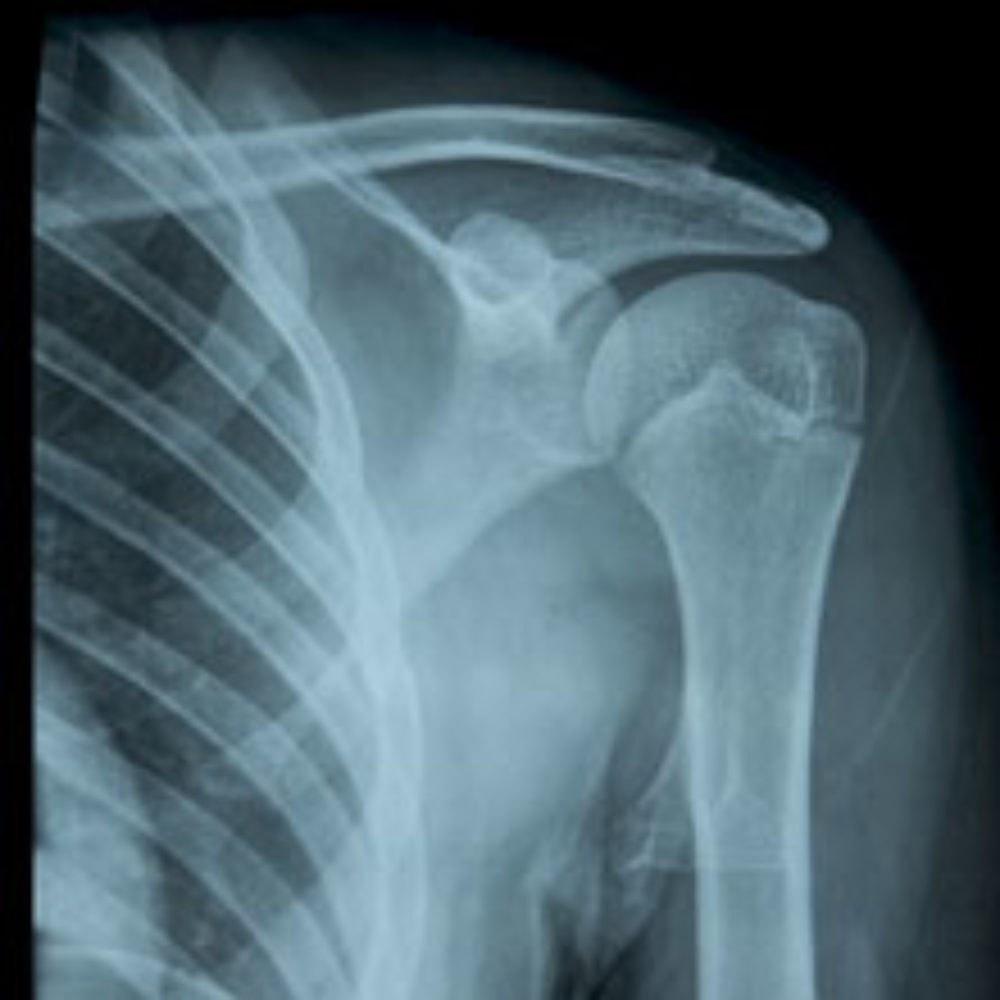
Various lawsuits have been filed in different federal courts throughout the United States alleging that the use of the intra-articular pumps to deliver pain medication after arthroscopic surgery caused the development of a rare, disabling shoulder condition known as Post-Arthroscopic Glenohumeral Chondrolysis (PAGCL). The condition is associated with the progressive destruction of cartilage, resulting in pain and often permanent disabilities.
The petition was filed with the Judicial Panel on Multidistrict Litigation to transfer, coordinate and consolidated all of the lawsuits for purposes of pretrial proceedings. An MDL, or multi-district litigation, is common in complex product liability cases where many people allege injuries as a result of the same medical device or medication. The procedure is designed to speed up the process of handling such cases by transferring them all to one court with a judge coordinating all of the pre-trial proceedings. If the cases do not settle or otherwise resolve, they will be remanded back to the originating court for trial.

Did You Know?
Millions of Philips CPAP Machines Recalled
Philips DreamStation, CPAP and BiPAP machines sold in recent years may pose a risk of cancer, lung damage and other injuries.
Learn MoreAccording to the petition, at least 13 shoulder pump lawsuits are pending in federal courts throughout the United States. However, hundreds of additional cases are currently being reviewed and investigated by attorneys who are preparing to file suits.
An article published in the July 2007 issue of the American Journal of Sports Medicine linked the use of intra-articular pain pumps after arthroscopic shoulder surgery to the development of PAGCL. Since that time lawyers have been reviewing potential shoulder pain pump lawsuits on behalf of individuals who developed symptoms of PAGCL, such as pain, stiffness, popping, grinding, decreased range of motion and loss of strength, after arthroscopic surgery.
In the lawsuits that have been filed, plaintiffs allege that the manufacturers encouraged the use of pain pumps following arthroscopic surgery as an alternative to other forms of pain management, even though they are not approved for that use by the FDA, and have not been adequately studied to determine they are safe for that purpose. In addition, the plaintiffs claim that the manufacturers and drug makers misled doctors about the dangers of developing Post-Arthroscopic Glenohumeral Chondrolysis (PAGCL).
Get more articles like this sent directly to your inbox.
"*" indicates required fields




0 Comments