Meningitis Cases Diagnosed Following Epidural Injections Rises
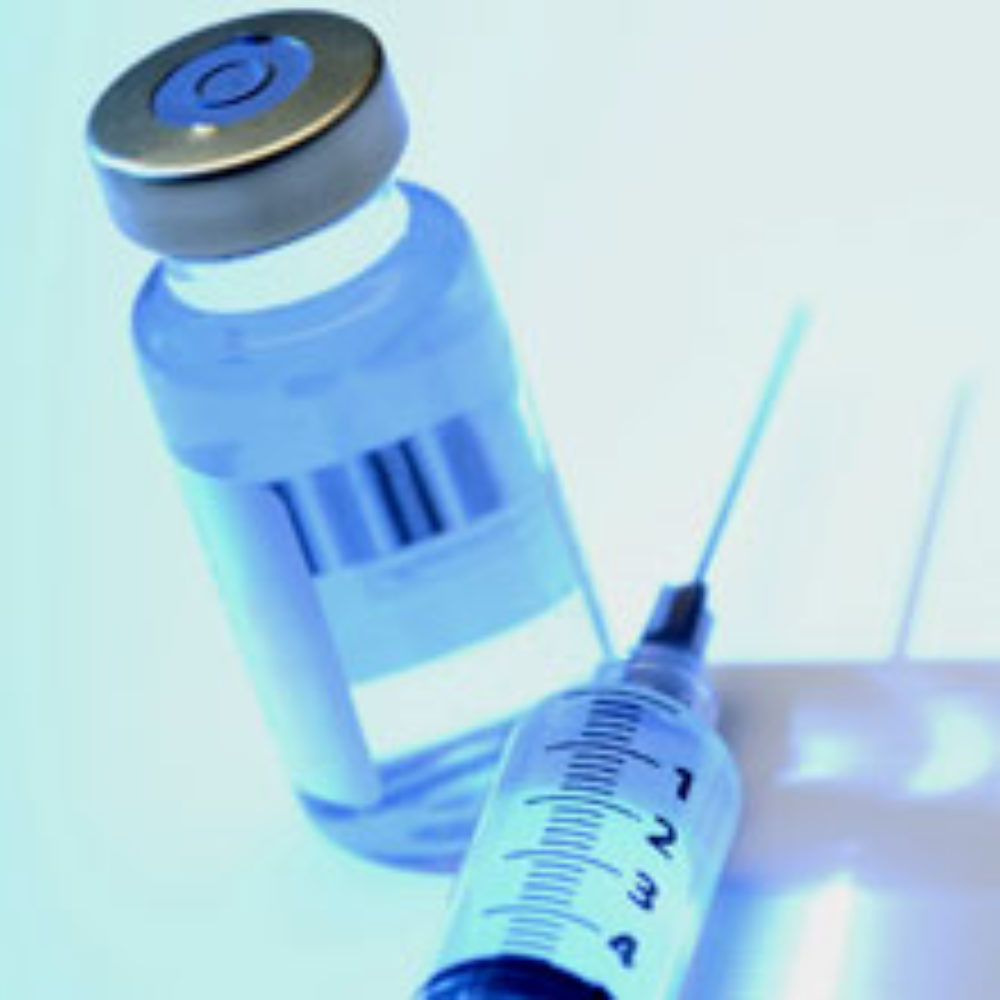
At least seven are confirmed dead and 91 have fallen ill with a form of fungal meningitis, which has been linked to epidural steroid injections distributed by a Massachusetts compounding pharmacy.
According to information released this weekend by the U.S. Centers for Disease Control and Prevention (CDC), the number of cases linked to the epidural injection meningitis outbreak continues to rise, and may continue to do so for some time because it may take as long as a month for some people to contract meningitis after being given tainted epidural steroid shots directly in the spine.
The contaminated shots came from the New England Compounding Center (NECC), a compounding pharmacy that issued a steroid injection recall last week amid the meningitis outbreak, and has suspended operations.

Did You Know?
Millions of Philips CPAP Machines Recalled
Philips DreamStation, CPAP and BiPAP machines sold in recent years may pose a risk of cancer, lung damage and other injuries.
Learn MoreMore than 17,500 potentially tainted epidural injections were shipped nationwide to 75 clinics in 23 states. The CDC and state health agencies have been scrambling to locate every patient who received one of the epidural injections, to make sure they are aware of the risk of meningitis and to see if they have gotten sick.
The CDC indicates that it is unable to know just how widespread the outbreak will be, but fungal meningitis is not contagious, so only those who were injected directly are at risk.
Tennessee, Michigan Report Majority of Meningitis Cases, Deaths
Tennessee has been the hardest hit by the outbreak, with 32 reported meningitis cases and three deaths. Michigan has had 20 cases and 2 deaths, and Virginia has had 18 cases and 1 death. Illnesses have also been reported in Florida, Indiana, Minnesota, North Carolina, Ohio, and Maryland, with the latter reporting one death as well.
The original recall was for methylprednisolone acetate compounded at the pharmacy. It is a steroidal pain reliever. However, the company has now recalled all its injectable drugs and suspended operations.
The CDC has identified at least two strains of fungal meningitis that have been traced back to the company’s injections, raising fears that there may have been a systemic problem with conditions at the New England Compounding Center.
Fungal meningitis is a type of meningitis that in this case was caused by aspergillius, a common mold that somehow tainted vials of the epidural injections. It causes inflammation of the spinal cord and protective membranes covering the brain. The inflammation generally causes an infection of the fluid surrounding the brain and spinal cord and can also be caused by parasites, viruses and bacterial infection.
Individuals with a weakened immune system may be at a particularly high risk of contracting fungal meningitis.
Symptoms of meningitis following an epidural steroid injection may include fever, headache, stiff neck, nausea, vomiting, increased sensitivity to light and mental confusion. Signs of meningitis usually develop within three to seven days after exposure. As the disease progresses, symptoms may become severe, resulting in seizures, coma and death.
CDC Issues Treatment Guide For Doctors
On October 6 the CDC issued interim treatment guidance for health care professionals treating patients who have been diagnosed with meningitis after receiving one of the tainted shots. It called for clinicians to continue routine empiric treatment protocols for bacterial pathogens and also called on them to add empiric broad-spectrum antifungal therapy to their treatment regimens.
“These recommendations are based upon current evidence that at least two fungal pathogens are involved, and the possibility that additional pathogens may be identified as the investigation continues,” the CDC explained. The CDC recommended doctors consult with an infectious disease physician for help with treatment and diagnosis, since treatment may be “complex and prolonged.”
Following the outbreak, a number of product liability lawyers are reviewing potential lawsuits for individuals diagnosed with fungal meningitis after receiving one of the contaminated steroid injections.
Get more articles like this sent directly to your inbox.
"*" indicates required fields
1 Comments




MagnolaOctober 10, 2012 at 7:38 pm
With the current outbreak of Meningitis I am more than curious about an episode of Meningitis that I survived in 2010. I received a steroid injection on October 29, 2010. About 2 weeks later I started to feel really lethargic and running low grade fevers at night with headaches. Around November 23rd, Thanksgiving Weekend, the "IT" hit the fan. The temps became increasingly higher, the headach[Show More]With the current outbreak of Meningitis I am more than curious about an episode of Meningitis that I survived in 2010. I received a steroid injection on October 29, 2010. About 2 weeks later I started to feel really lethargic and running low grade fevers at night with headaches. Around November 23rd, Thanksgiving Weekend, the "IT" hit the fan. The temps became increasingly higher, the headaches worsened; thinking it was sinus My doctor sent me a round of ABT, but got no better, 1 week later I was sent a "Z" pack, took only one does on Monday, by then I couldn't get up to dress myself. I went the emergency room on Tuesday, unable to shower, and was diagnosed as having bacterial meningitis. For 9 days my body was infused with the Cadillac's of ABTS, eventually being sent home on an additional week of IV ABTS , with a subsequent week of oral ABTS. I am now thinking I was truly blessed.