Relpax Recall Issued Over Potential Microbial Contamination
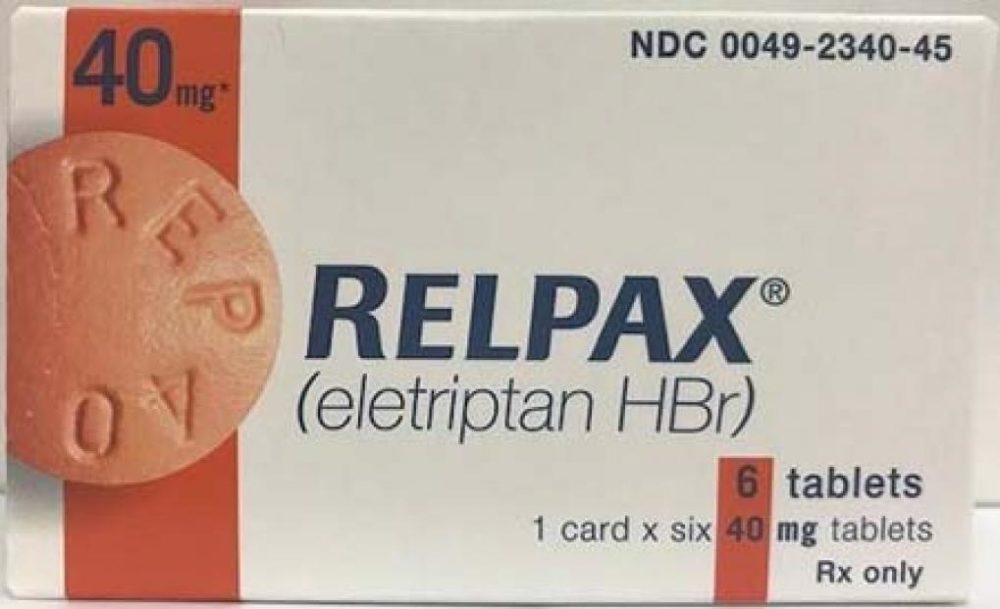
Pfizer is recalling two lots of the migraine medication Relpax, due to a risk that the pills may be contaminated with bacteria that could cause life-threatening infections for some patients.
The FDA announced the Relpax recall on August 15, indicating that the drug could be contaminated with Genus Pseudomonas and Burkholderia.
Although no reports of illness or infection have been linked to the recalled Relpax, this could cause severe health complications if a bacterial infection disseminates from the gut to the bloodstream, where it could result in death.

Did You Know?
Millions of Philips CPAP Machines Recalled
Philips DreamStation, CPAP and BiPAP machines sold in recent years may pose a risk of cancer, lung damage and other injuries.
Learn MoreRelpax (eletriptan hydrobromide) was approved by the FDA in 2002, for treatment of migraines in adults.
According to the recall notice, two lots of 40 mg tablets of Relpax may not have met the company’s in-house microbiological specifications and could be contaminated with the bacteria.
For the general population, the risks of serious infection are low. However, patients with compromised immune systems, like cystic fibrosis patients or those with chronic granulomatous disease, may suffer serious life-threatening infections and face serious risks.
The recall affects two lots of Relpax, including lot number AR5407 with cartons containing one blister card with six tablets, with carton NDC 0049-2340-45; and lot number CD4565 with cartons containing two blister cards with six tablets each, with carton NDC 0049-2340-05. The medication is packaged in cartons and distributed nationally from June 2019 to July 2019.
Pfizer notified direct sales customers via a recall letter. The recall warns that anyone with the recalled pills in their existing inventory should stop distributing and quarantine them immediately. Pfizer is also asking retailers, hospitals, and healthcare providers who have distributed the recalled pills to patients to notify them about the recall. Customers with questions can call Stericycle at 877-255-9750.
Consumers using Relpax should consult with their doctor or pharmacy to determine if their medication is among the lots affected by the recall The FDA asks that side effects or adverse reactions linked to taking Relpax be reported to the FDA’s MedWatch adverse event program.
Get more articles like this sent directly to your inbox.
"*" indicates required fields
1 Comments




PennySeptember 4, 2019 at 5:26 pm
I've already taken 20+ of these tabs w/this recall lot # since June 2019 AND NOW A RECALL IS POSTED! I am having GI problems! Scheduled for testing before this recall even came out!!! They had a "recall" in May for a, so-called, misprint on packaging (uh huh). I called Pfizer 6/2019 directly to find out what's going on.