Stryker Accolade, LFit Hip Failure Led To Need For Extensive Rehab, Lawsuit Claims
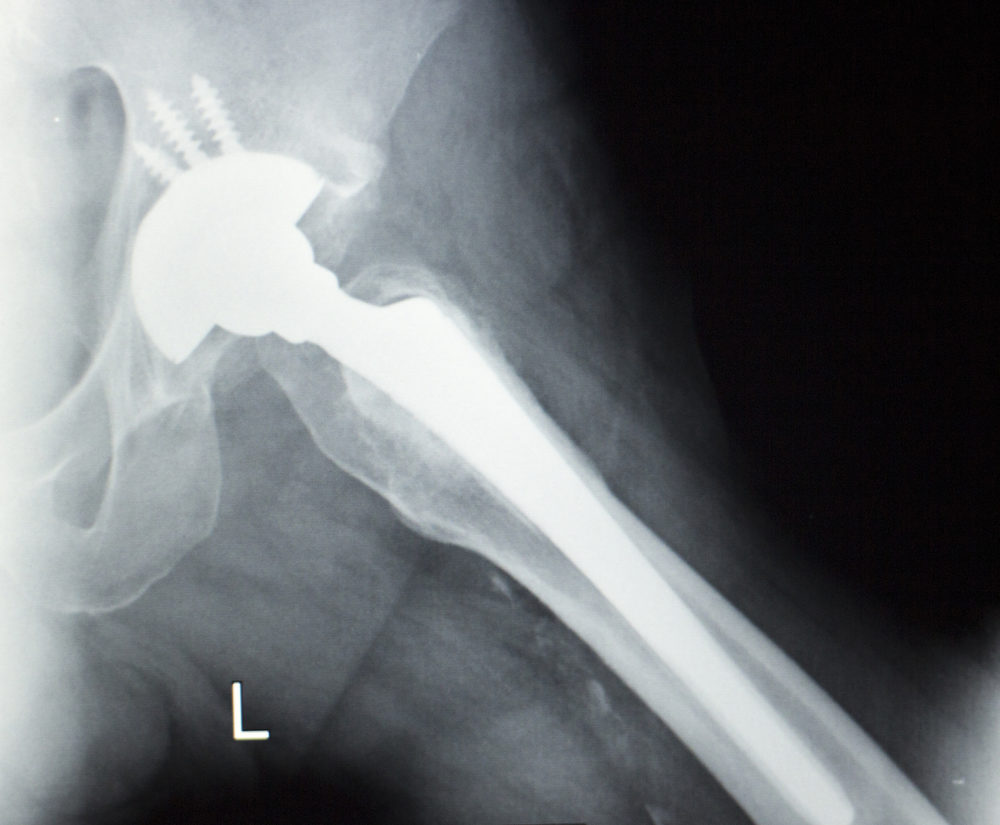
As a result of the failure of a Stryker Accolade hip stem and LFit V40 femoral head, a California woman indicates that she suffered bone loss, extensive rehab treatment and, ultimately, required revision surgery to remove recalled hip replacement components from her body.
In a complaint (PDF) filed in the U.S. District Court for the Central District of California on June 5, Joyce Ulrich claims that all of her injuries were caused by the defective and unreasonably dangerous character of a Stryker hip implant received in February 2009.
Following her right hip replacement procedure, where doctors combined a Stryker Accolade TMZF Hip Stem with a Stryker LFit Anatomic V40 Femoral Head, Ulrich indicates that she began to experience pain and discomfort. Although artificial hip implants are typically expected to last much longer, Ulrich indicates that she required revision surgery on May 17, 2016, to remove the components.

Learn More About
Lawsuits are being reviewed for several different dangerous and defective hip replacement systems.
Learn More About this Lawsuit See If You Qualify For Compensation“During surgery, it was discovered that, in fact, there was significant evidence if trunnionosis around the proximal femur at the junction between the head and neck,” the lawsuit states. “The operative report also noted areas of osteolysis, bone loss, and loosening of the femoral component.”
As a result of the Stryker hip failure, Ulrich indicates that she has suffered impairment, debilitating loss of mobility, and pain and has had to undergo extensive physical rehabilitation since the revision surgery.
After receiving similar reports of problems among individuals nationwide, a Stryker LFit v40 hip recall was issued last summer by the manufacturer, impacting certain large-diameter femoral heads sold before 2011. At that time, Stryker acknowledged that a higher-then-expected number of individuals were experiencing problems with trunnion failure, metal wear, adverse tissue reactions and other complications.
Ulrich presents claims of negligence, defective manufacturing, defective design, inadequate warning, failure to conform to representations, failure to adequately test, breach of warranty, negligent misrepresentation and fraud. She is seeking punitive and compensatory damages.
The case filed by Ulrich joins a growing number of other product liability lawsuits filed in recent months by those who have suffered complications due to Stryker Accolade and LFit V40 hip components.
As hip replacement lawyers continue to review cases for individuals who are just now learning that complications experienced in recent years may have been caused by design problems, it is expected that several thousand lawsuits may be filed in the coming months and years.
Get more articles like this sent directly to your inbox.
"*" indicates required fields






0 Comments