Problems with Tanzeum Diabetes Drug Pen Liked to Overly Complex Instructions, Medication Errors: Report
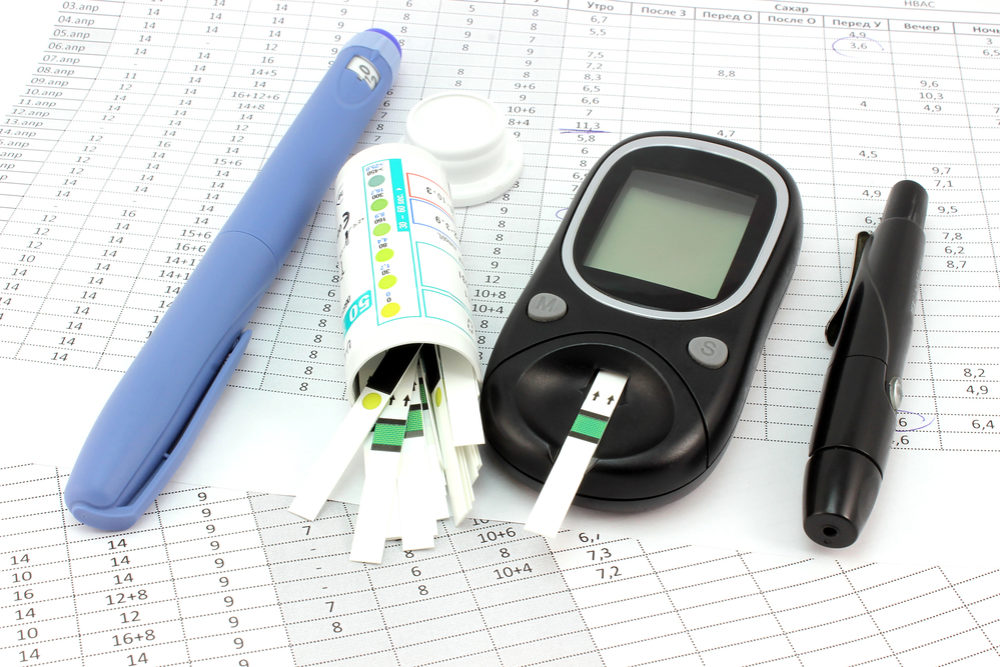
A new report warns that overly complex instructions may pose serious problems for Tanzeum self-injection diabetes pen users, potentially leading to errors dispensing diabetic medication that could result in adverse health consequences.
The Institute for Safe Medication Practices (ISMP) released its QuarterWatch report on October 5, indicating that the FDA has received more than 1,500 adverse event reports involving the Tanzeum pen problems over the course of a year.
Most of the reported issues, 1,404 of them, involved device use errors that could be linked to how complex it is to use the dabetes pen, which injects albiglutide, a glucagon-like-peptide-1 (GPL-1) agonist. The class of drugs also includes Victoza, Byetta and Trulicity.

Did You Know?
Millions of Philips CPAP Machines Recalled
Philips DreamStation, CPAP and BiPAP machines sold in recent years may pose a risk of cancer, lung damage and other injuries.
Learn More“Examination of the 10-page albiglutide patient instruction leaflet revealed it was indeed challenging to assemble the self-injection pen for weekly use,” the report notes. “The process took more than 30 minutes, required more than a dozen separate steps, including gently shaking and then twisting the pen assembly to dissolve and prepare the drug injection.”
The instructions warn patients not to use the pen if they see undissolved particles, and calls for them to tap the pen to bring large air bubbles to the top, but says small air bubbles are not a problem. The ISMP reports that users may have problems discerning between large and small air bubbles, and identifying undissolved particles.
In addition to the 1,404 reports of device use problems, there were also 490 reported cases of accidental exposure, 293 reports of device leakage, and some reports of wrong dose injections and device failure.
The ISMP report calls on the manufacturer, GlaxoSmithKline, to increase efforts to educate patients about the use of the pen, and advised doctors to consider ease of use when prescribing the pen to their patients.
Get more articles like this sent directly to your inbox.
"*" indicates required fields
5 Comments





WandaAugust 26, 2017 at 5:42 pm
I have had trouble with several of the syringes and not being able to twist them to the 2nd level so that the end will pop out and it is ready for dispensing. I am a registered nurse so I do not think technique is the issue, but this is for my own self administration.
EdmundAugust 17, 2017 at 10:38 pm
I have been on the meds for over a year.my doctor uppef my dose from 30 mil to 50. It is the sickest I have ever been in my life with sever diarrhea. I had it so bad that I went to hospital all my tests came back negative. Borland and Groover could not find nothing wrong with my body thank God. stoppef taking my shot and got well. i would never ever take this med again it almost killed me. Edmund [Show More]I have been on the meds for over a year.my doctor uppef my dose from 30 mil to 50. It is the sickest I have ever been in my life with sever diarrhea. I had it so bad that I went to hospital all my tests came back negative. Borland and Groover could not find nothing wrong with my body thank God. stoppef taking my shot and got well. i would never ever take this med again it almost killed me. Edmund wilder. p.s. stopped taking in July of2017.
JanetteJune 8, 2017 at 4:13 am
I have been on Tanzeum 50mg for about two years. Occasionally, the pen has a tiny bit of fluid leak. But for the most part it's been fine. My A1c went from 8.2 to 6.2 in 3 months. I'm actually very happy with the medicine.
DoloresJanuary 30, 2017 at 11:08 pm
I had similar problems with mine 30 mg I am able to twist #2 then it freezes, had to go through 3 pens in one day. that gets awfully expensive! Please advise
DeniseJanuary 2, 2017 at 10:54 pm
I have attempted many times to administer the 50 mg dose and many times the syringe is so hard to turn to the appropriate dose that it freezes at a dose that has discharged fluid and will not allow the administration of the remainder. I have called and reported the problem, and have had to refill before the correct time to be able to have dose available. please advise course of action, as I will b[Show More]I have attempted many times to administer the 50 mg dose and many times the syringe is so hard to turn to the appropriate dose that it freezes at a dose that has discharged fluid and will not allow the administration of the remainder. I have called and reported the problem, and have had to refill before the correct time to be able to have dose available. please advise course of action, as I will be asking for another drug that will actually work.