Zostavax Shingles Vaccine Problems Surfaced Two Days After Injection, Lawsuit Alleges

An Illinois man has filed a product liability lawsuit claiming that the problems with the Zostavax shingles vaccine actually caused him to suffer an outbreak of shingles just days after receiving the injection.
Thomas Vlcek filed the complaint (PDF) in the U.S. District Court for the Eastern District of Pennsylvania on April 4, alleging that Merck & Co. sold a defective shingles vaccine, which contained a version of the live virus that was not sufficiently weakened to avoid causing users to suffer the actual disease it was designed to prevent.
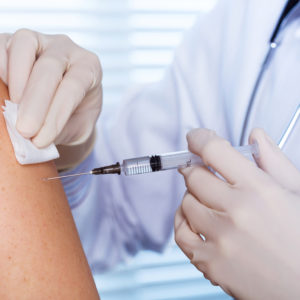
According to the lawsuit, Vlcek received a Zostavax injection in December 2015, as part of his routine health maintenance for the prevention of shingles. However, just two days later, he began suffering an outbreak of blisters the right side of his back, and he was later diagnosed with shingles.
Learn More About
Side effects of the shingles vaccine Zostavax may result in the development of a painful and persistent strain of shingles
Learn More About this Lawsuit SEE IF YOU QUALIFY FOR COMPENSATIONZostavax was introduced in May 2006, as a single dose vaccine for prevention of shingles among individuals over the age of 50, and amid aggressive marketing it quickly grew to become a part of standard recommended care for nearly all older Americans. However, an alarming number of Zostavax shingles vaccine problems have been reported, where users suffered longer and more painful shingles outbreaks and other infections caused by the use of an “under-attenuated” version of the virus.
“Since Zostavax’s introduction in 2006, vaccine adverse event reports (VAERs)
appeared in significant numbers addressing various adverse effects, including, but not limited to, viral infection resulting in disease of the central nervous system, including acute disseminated encephalomyelitis and acute transverse myelitis,” the lawsuit states. “Despite this information and the potential correlation between being administered the Zostavax vaccine and within a relatively short period of time developing an infection, leading to the development of shingles or varicella-zoster virus pneumonia, Merck failed to properly address and provide this information both to the patient and the medical providers prescribing the vaccine.”
Hundreds of Zostavax shingles vaccine lawsuits are now being pursued nationwide. Given similar questions of fact and law raised in the complaints, the federal litigation has been centralized before U.S. District Judge Harvey Bartle III in the Eastern District of Pennsylvania, for coordinated discovery and pretrial proceedings as part of a federal MDL, or multi-district litigation.
As lawyers continue to investigate and file additional claims on behalf of people who have experienced problems from Zostavax, the size of the litigation is expected to continue to grow over the coming months and years. If Merck fails to reach Zostavax settlements or another resolution for the claims following bellwether trials, each individual lawsuit may eventually be remanded back to different U.S. District courts nationwide for separate trial dates in the future.
Get more articles like this sent directly to your inbox.
"*" indicates required fields





0 Comments