Elmiron Exposure Linked To “Strong Dose-Dependent Association” To Retinal Maculopathy: Study
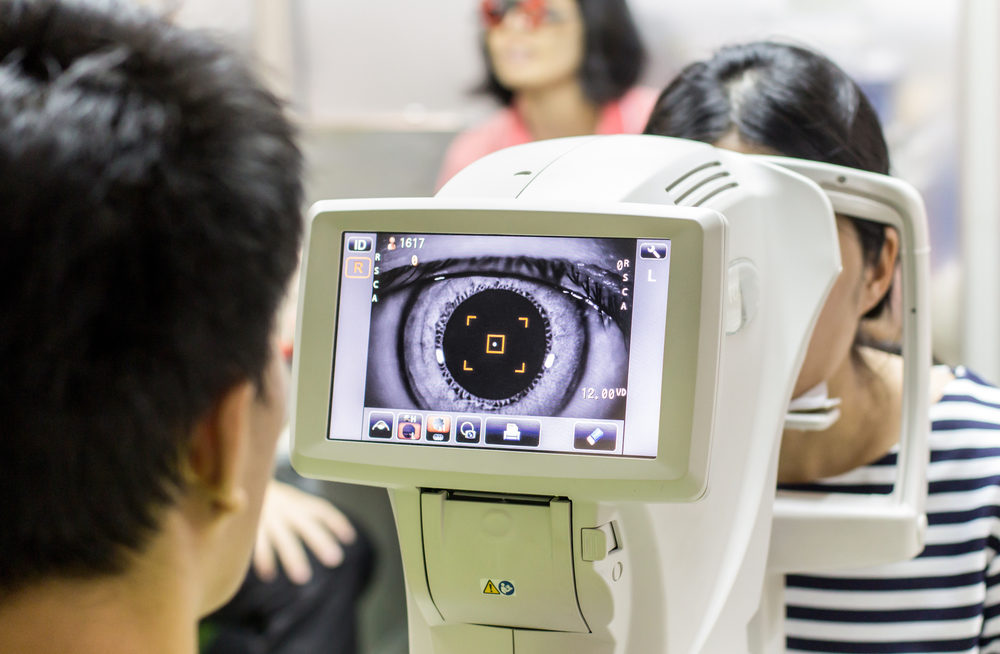
The findings of a new study point to a strong causal link between Elmiron and eye damage known as retinal maculopathy, which can result in various forms of vision loss.
Researchers from Emory University School of Medicine published a report last month in the medical journal Survey of Ophthalmology, which looked at data from numerous prior studies, coming to the conclusion that there is a strong dose-dependent risk of retinal maculopathy associated with use of the bladder drug Elmiron (pentosan polysulfate sodium, or PPS).
The link is so strong, in fact, that many scientists and doctors describe the eye damage as pentosan polysulfate maculopathy, referring to the active pharmaceutical ingredient in Elmiron.
Learn More About
Side effects of Elmiron have been associated with vision loss and retina damage known as pigmentary maculopathy.
Learn More About this Lawsuit SEE IF YOU QUALIFY FOR COMPENSATIONThe FDA first approved Elmiron in 1996, for the treatment of interstitial cystitis (IC), which affects more than 1 million people in the U.S. alone. Also known as “painful bladder syndrome”, interstitial cystitis involves severe and chronic pain in the bladder and groin.
Although the drug has been on the market for decades, increasing evidence published in recent years has found Elmiron may have a toxic impact on the retina, leading to irreversible vision loss.
In this latest study, researchers conducted a review of all of the currently available data to see how strong the connection was between Elmiron and retinal maculopathy. They sought to consolidate known clinical features, as well as evaluate the strength of the science behind the association between the drug and the vision problems.
“Current literature supports a strong dose-dependent association between PPS exposure and a progressive maculopathy impacting the retinal pigment epithelium (RPE) and RPE-photoreceptor interface that may worsen even after drug cessation,” the researchers wrote. “Initial symptoms may include prolonged dark adaptation and difficulty reading with relative visual acuity preservation.”
The researchers noted that the Elmiron maculopathy can be diagnosed through multimodal examinations, which involves a series of images that can reveal macular pigment clumps linked to lesions, spots in the posterior pole, and, in some cases, a “characteristic peripapillary hypoautofluorescent halo.”
They also noted recent evidence has linked Elmiron use with a loss of central vision acuity, which they identified as a cause for concern.
“This newly described association implies significant public health risk,” the researchers warned. “Ophthalmologists should screen PPS users with multimodal retinal imaging, and prescribers should minimize dose and duration of PPS use.”
Elmiron Litigation
The findings will likely provide compelling evidence in hundreds of Elmiron vision loss lawsuits now being pursued against the drug makers, alleging that users and the medical community were not adequately warned about the risk of retinal maculopathy from Elmiron.
The lawsuits allege users could have avoided permanent vision loss if earlier information had been provided about the importance of monitoring and use of multimodal imaging.
Given common questions of fact and law raised in individual injury claims and Elmiron class action lawsuits pending throughout the federal court system, the litigation has been centralized in the U.S. District Court for the District of New Jersey, where one judge is presiding over coordinated discovery and pretrial proceedings.
As Elmiron lawyers continue to review and file claims in the coming months and years, it is ultimately expected that several thousands claims will eventually be included in the litigation.
Get more articles like this sent directly to your inbox.
"*" indicates required fields






0 Comments