Opana ER Risks Outweigh Benefits, Despite “Abuse-Deterrant” Claims: FDA Panel
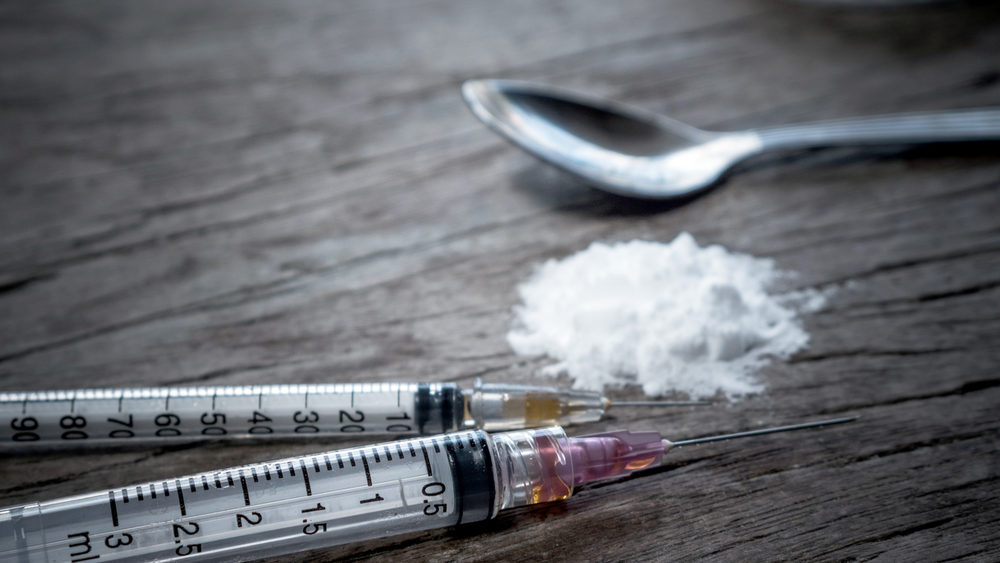
Despite marketing claims that the narcotic painkiller Opana is “abuse-deterrent”, a panel of outside advisers to the FDA have concluded that the risks associated with the opioid medication outweigh the potential benefits.
Members of two FDA advisory panels voted 18 to 8 on Tuesday to recommend that the agency issue an Opana ER recall, indicating that it poses too great of a risk and should be pulled from the market.
The joint FDA panel involved members of the Anesthetic and Analgesic Drug Products Advisory Committee (AADPAC) and the Drug Safety and Risk Management Advisory Committee (DSARM).

Did You Know?
Millions of Philips CPAP Machines Recalled
Philips DreamStation, CPAP and BiPAP machines sold in recent years may pose a risk of cancer, lung damage and other injuries.
Learn MoreOpana ER is Endo Pharmaceutical’s extended-release version of the opioid oxymorphone. The drug is widely abused, especially after the reformulation was made to deter addicts from snorting it, the panel found. This pushed many addicts to begin injecting the drug instead.
The FDA is not required to follow the panel’s recommendation, but often does. It is now up to the agency to determine if it will act on the panels vote. The FDA could enforce an Opana recall and national ban, change the warning labels, or restrict prescriptions of the drug.
Opioid abuse has come to the forefront of media attention in recent years. A study published in 2015 indicated more people are dying from overdose deaths after using powerful morphine-like narcotic painkillers, like Opana. Other studies also warn that high-potency opioids had significantly increased.
Critics say the new reformulated extended-release version of Opana is more dangerous than the original version it replaced. The abuse increased significantly after OxyContin was reformulated in 2010, to make it harder to crush and snort.
Abusers Began Injecting Abuse-Deterrent Pills
Since the Opana reformulation, addicts have begun injecting the drug. The change has caused multiple overdose deaths, lead to several outbreaks of HIV, hepatitis C, vascular issues and an outbreak of Thrombotic Thrombocytopenia Purport (TTP).
TTP is a life-threatening blood disease. Endo Pharmaceuticals said TTP was also reported with intravenous use of abuse-deterrent OxyContin tablets. The company speculated TTP stems from the inactive ingredient PEO, which is used to create the gel coating and has not been well studied.
Opana injections were the main cause of an HIV outbreak in Indiana in 2015. Addicts began injecting Opana to bypass the abuse-deterrent aspects. It also becomes 10 times more potent than morphine when injected. More than one user will share one quarter of a pill, leading to more needle sharing.
A study by the U.S. Centers for Disease Control and Prevention indicated users must inject Opana several times a day because of its short half-life, which causes withdrawal symptoms to begin much sooner. Users melt the gel coating, which creates the extended release effect. They then crush and mix the drug with a significant amount of water. The diluted solution forces the user to inject several times a day.
Critics warn calling a drug “abuse deterrent” is misleading, and may lead to doctors overprescribing the medication. Others say calling a drug “abuse-deterrent” is simply a marketing trick by pharmaceutical companies.
The FDA recently launched a competition to spur technology developers to create an app for abusers experiencing an opioid overdose to get access to nearby life saving medication. The FDA also held a hearing in early 2016, focusing on the role government can play in reducing opioid painkiller abuse.
Get more articles like this sent directly to your inbox.
"*" indicates required fields
1 Comments





TammyJune 10, 2017 at 6:49 am
I am.currrntly undergoing treatment for an infection from using opanas intravenously for last 6 years. It has left me with a debilitating back condition which requires surgery. It has eaten the bones away in my spine. I may be paralyzed. I was prescribed opanas for years. I even complained.to Endo directly when the formula was changed. All I got was a thank you. They should be held accountable. Th[Show More]I am.currrntly undergoing treatment for an infection from using opanas intravenously for last 6 years. It has left me with a debilitating back condition which requires surgery. It has eaten the bones away in my spine. I may be paralyzed. I was prescribed opanas for years. I even complained.to Endo directly when the formula was changed. All I got was a thank you. They should be held accountable. They have helped ruin my health and changed my life. I am in a skilled nursing facility.