Link Between Side Effects of Plavix and Hepatitis Described in Case Study
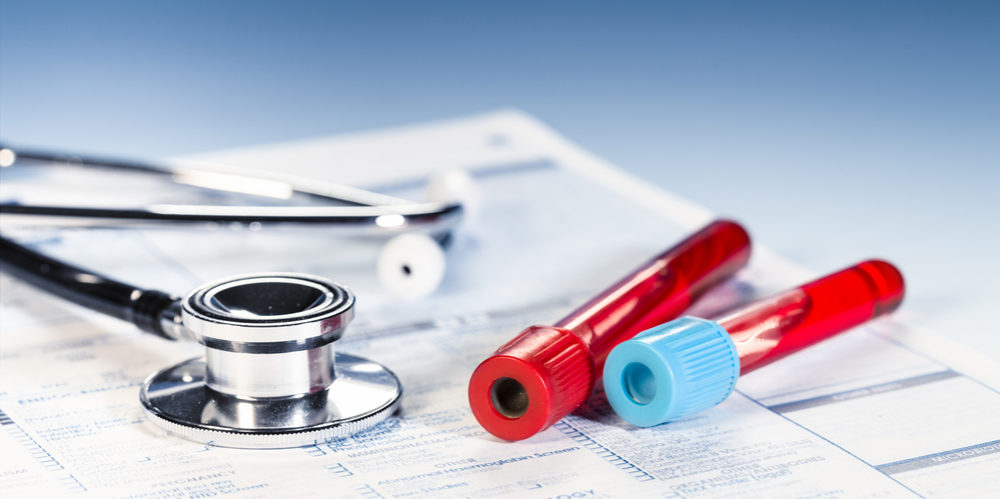
A new case study suggests that the side effects of Plavix may be linked to reports of hepatitis infections, raising concerns about the widely used blood thinner.
In an article published in Case Reports in Hepatology, researchers from Advocate Christ Hospital, in Illinois, highlight of a case of severe Plavix-induced hepatitis. The researchers reviewed previous reports and studies, identifying several cases of hepatoxicity linked to the drug.
Plavix (clopidrogrel) is a blockbuster medication that has been used by millions of people throughout the United States, and is commonly prescribed to prevent blood platelets from sticking together and forming clots. There are between 2.5 million and 3 million Plavix prescriptions handed out each month in the U.S.

Did You Know?
Millions of Philips CPAP Machines Recalled
Philips DreamStation, CPAP and BiPAP machines sold in recent years may pose a risk of cancer, lung damage and other injuries.
Learn MoreThe case report indicates that a 34 year old man with a history of heart disease and a coronary artery stent was prescribed aspirin plus Plavix. He took the drug for two months, 12 years ago, but then restarted it. Four and a half months after restarting Plavix treatment, the patient showed signs of jaundice and fatigue. He was diagnosed with Plavix-induced severe hepatitis.
Researchers said this was one of the few times a liver biopsy was performed on a patient with Plavix hepatitis.
“Our patients liver biopsy revealed severe hepatocellular injury. This adds to the histological findings in clopidogrel-induced hepatitis, as liver biopsy was only performed in 3 of the previously reported cases,” the researchers noted. “Clopidogrel-induced hepatitis is a rare but potentially serious adverse effect. A high degree of clinical suspicion is required in patients presenting with abnormal liver biochemistries within a few months after starting clopidogrel. Prompt recognition and discontinuation of the offending agent are necessary, as progressive liver injury and even death can occur.”
Plavix Litigation
The study comes as the makers of Plavix, Bristol-Myers Squib and Sanofi-Aventis, face hundreds of Plavix lawsuits.
The cases all involve similar claims that Bristol-Myers Squibb and Sanofi-Aventis failed to adequately warn consumers or the medical community about risks associated with Plavix, including gastrointestinal bleeding, severe ulcers, a rare blood disorder known as thrombotic thrombocytopenic pupura (TTP) and other injuries.
Since February 2013, all product liability cases filed throughout the federal court system involving injuries allegedly caused by Plavix side effects have been centralized for coordinated pretrial proceedings before U.S. District Judge Freda L. Wolfson in the District of New Jersey, as part of an MDL or Multidistrict Litigation (MDL).
Following expert witness discovery and any motions for summary judgment, Judge Wolfson indicates that any remaining cases not dismissed by November 1, 2016 will comprise a Trial Pool, from which the first case will be selected for trial beginning in July 2017.
Get more articles like this sent directly to your inbox.
"*" indicates required fields





0 Comments