Prolia Discontinuation Increases The Risk Of Vertebral Fractures, Study Found
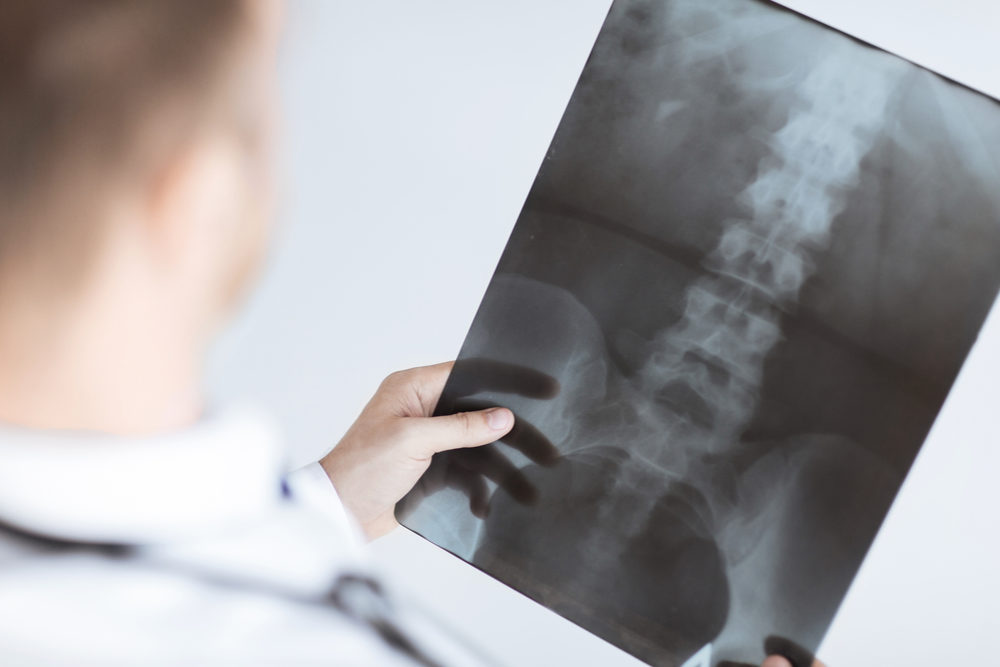
The side effects of Prolia may increase the risk of vertebral fractures when consumers stop using the osteoporosis drug, according to the findings of a study by Israeli researchers.
In research published in the journal Calcified Tissue International last year, serious concerns were raised about the impact of Prolia discontinuation, adding to a growing body of evidence about the fracture risk associated with the medication.
Prolia (denosumab, or DMAB) was introduced by Amgen in 2010, and was the first drug approved by the FDA for the treatment of postmenopausal osteoporosis.

Did You Know?
Millions of Philips CPAP Machines Recalled
Philips DreamStation, CPAP and BiPAP machines sold in recent years may pose a risk of cancer, lung damage and other injuries.
Learn MoreAccording to the researchers, the drug was approved after showing a reduction in vertebral and hip fractures over the course of three years, and keeping the risk reduced up to 10 years. However, they noted that a number of reports raised concerns about the risk of fractures once users discontinued the drug, due to rapid bone density declines and reports of multiple vertebral fractures.
The researchers conducted a phone survey of doctors engaged in bone metabolism in nine Israeli hospitals. They looked at clinical data on patients with vertebral fractures after Prolia discontinuation and found nine elderly female patients, most of who had prolonged exposure previously to Fosamax or another bisphosphonate drug.
According to the findings, the women collectively suffered 36 vertebral fractures, eight of whom had multiple fractures; most of which were spontaneous.
“In line with previous reports, the timing and severity of the fractures raise concern of DMAB discontinuation effect,” the researchers concluded. “Care providers, patients, and regulatory authorities should be aware of the possible risk of DMAB treatment interruption.”
Prolia Bone Fracture Concerns
In 2013, the prominent consumer watchdog group Public Citizen raised concerns about the Prolia fracture risks, noting that the drug has been linked to reports of bone brakes following little or no trauma. The group indicated that Prolia appears to interfere with bone metabolism and the body’s immune system, resulting in breaks that may occur in situations that would not typically result in such an injury.
Although Amgen issued warnings about the link between Prolia and bone fractures in Canada as early as 2012, concerns have been raised that insufficient warnings are still being provided in the United States.
Earlier this month, Public Citizen filed a petition with the FDA calling for the agency to require a “black box” Prolia fracture warning, which would be the strongest warning the drug maker could be required to add to the label.
As more consumer learns that problems suffered in recent years may have been avoided with stronger warnings, it appears likely that Amgen may face Prolia bone fracture lawsuits, raising allegations similar to those presented in several thousands Fosamax lawsuits in prior years.
Get more articles like this sent directly to your inbox.
"*" indicates required fields
3 Comments




FredOctober 2, 2022 at 12:43 pm
My wife has just sustained compression fractures of at least four vertebrae after Prolia was stopped for dental surgery. This week there is going to be a kyphoplasty procedure to fix two of those compression fractures.
DIANEMay 26, 2020 at 1:39 pm
I continue to be deeply alarmed by the marketing of Prolia as a safe treatment for OP, and the absence of adequate warnings to women of all ages. I am a retired lawyer and a victim of Prolia, having suffered SVF. I am aware of at least a dozen other American victims of SVF caused by Prolia. The studies indicating that it is rare are, in my humble opinion, underestimating the frequency of this crip[Show More]I continue to be deeply alarmed by the marketing of Prolia as a safe treatment for OP, and the absence of adequate warnings to women of all ages. I am a retired lawyer and a victim of Prolia, having suffered SVF. I am aware of at least a dozen other American victims of SVF caused by Prolia. The studies indicating that it is rare are, in my humble opinion, underestimating the frequency of this crippling aftereffect. Not that anyone taking the drug is EVER warned about the requirement that you stay on Prolia PERMANENTLY, absent an available exit protocol, which involves expensive monitoring and a further drug therapy (which drug therapy is an option for many, many women - leaving them exposed to the risk!) None of this is revealed to the patient when their "informed" consent to treatment is purportedly obtained. This inexcusable practice of prescribing this drug without proper warnings and fully informed consent has to stop. Class actions are a powerful consumer protection device. I do hope that the viability of a class action will be fully investigated.
JamesDecember 18, 2019 at 7:33 pm
Since the beginning of my treatment of what I was told was a very serious case of osteoporosis I have experienced more problems than I ever had prior to the medical treatments. I have suffered from avascular necrosis, cervical spine issues, jaw bone issues, skin rashes, uncontrolled cholesterol issues, joint pain issues, severe cramping.