Ovation iX Abdominal Stent Graft Recall Issued Over Polymer Leaks
More than 5,000 stent grafts used to treat patients with aorta complications have been recalled, over a production error that may allow liquid polymer to leak, increasing the risk of patient injury or death.
The FDA announced the Endologix Ovation iX Abdominal Stent Graft System recall on June 16, due to potential for liquid polymer leaks during implantation, which could significantly worsen a patient’s condition and require additional emergency surgery.
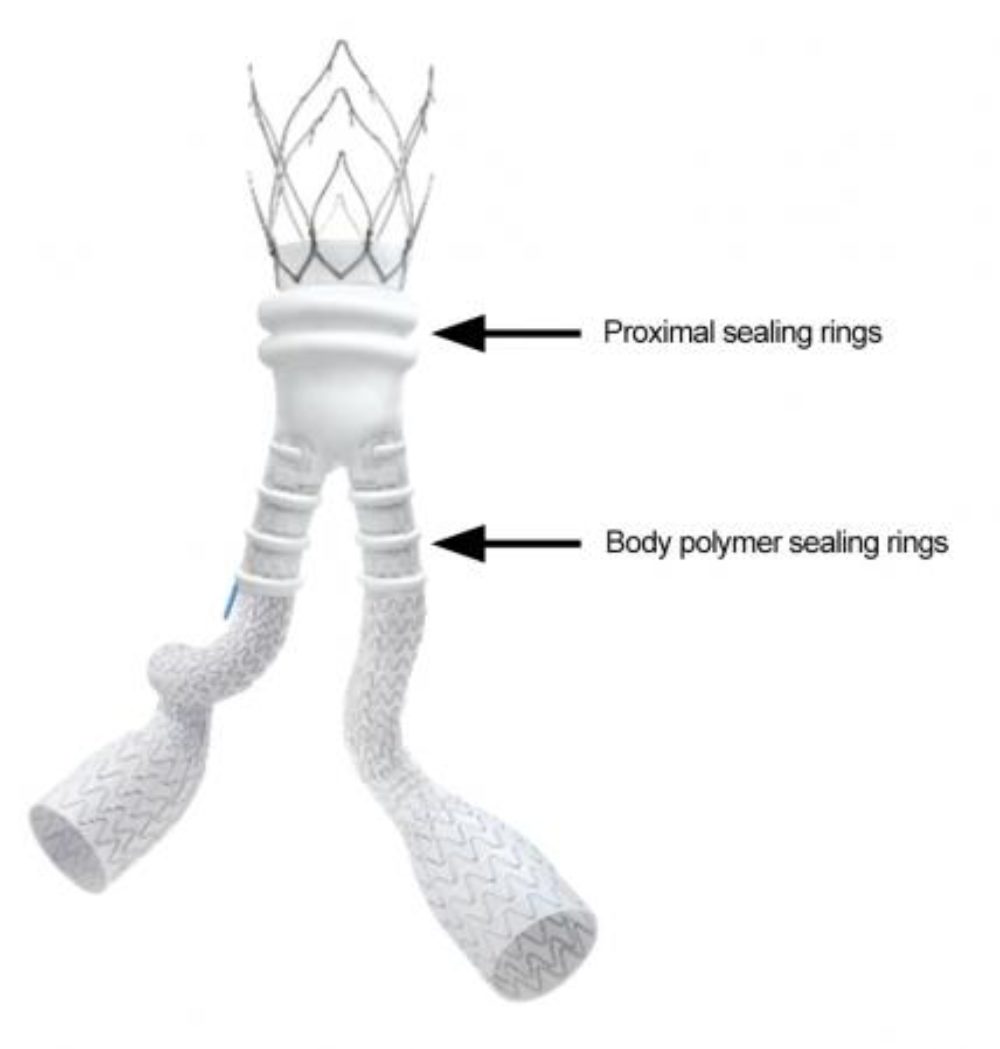
The Ovation iX Abdominal Stent Graft Systems are used to treat patients with an abdominal aortic or aortoiliac aneurysm, which is a condition that occurs when the aorta becomes stretched and thin, causing the vessel to bulge or expand. The stents are implanted and designed to use a polymer-based technology to seal off blood from flowing into the aneurysm and help secure the device in place.

Did You Know?
Ticketmaster Data Breach Impacts Millions of Customers
A massive Ticketmaster data breach exposed the names, addresses, phone numbers, credit card numbers and other personal information of more than 560 million customers, which have now been released on the dark web. Lawsuits are being pursued to obtain financial compensation.
Learn MoreAccording to the recall, the stent grafts have the potential to leak polymer liquid during implantation due to a material weakness that occurred during the manufacturing process. FDA officials are warning the weakened material may open up or gap during use, which may allow the liquid polymer to leak and allow blood to continue flowing into the aneurysm, requiring additional procedures to seal it off.
Officials are also warning if the liquid polymer leaks into other parts of the body, patients could suffer from severe allergic reactions, necrosis, unstable blood pressure, cardiac arrest, organ failure, central nervous system problems and even death.
The recall includes an estimated 5,403 Ovation iX Abdominal Stent Graft Systems with model numbers TV-AB2080-J, TV-AB2380-J, TV-AB2680-J, TV-AB2980-J and TV-AB3480-J. The recalled stent systems were manufactured from 2015 to present by Endologix Inc., and were distributed for sale from August 21, 2015 to present to healthcare facilities throughout the U.S.
Endologix issued an Urgent Medical Device Correction Notice to customers last month informing them of the cause of the polymer leak and instructing doctors to carefully consider the risk of polymer leaks and the use of alternative devices.
However, this month the FDA categorized the recall as a Class I, indicating it is the most serious type of recall, and that the use of the product is associated with risk of severe adverse health consequences including death.
Endologix announced on June 15, 2020 that the company plans to replace Ovation iX Abdominal Stent Graft System with the recently FDA-approved ALTO Abdominal Stent Graft System by October 31, 2020, and will be issuing a new warning on the label of the Ovation models.
Customers with additional questions or concerns are encouraged to contact Endologix customer service at 1-800-983-2284 and to report any and all adverse reports to the FDA Medwatch Adverse Event Reporting Program.





0 Comments