Eligible for a Suboxone lawsuit?
Lack of Suboxone Film Tooth Decay Warnings Placed Users At Risk of Severe Dental Injuries, Erosion: Lawsuit
Pennsylvania woman used the opioid addiction treatment for nine years before Suboxone tooth decay warnings were finally placed on the drug label, leaving her with extensive dental injuries and tooth damage.
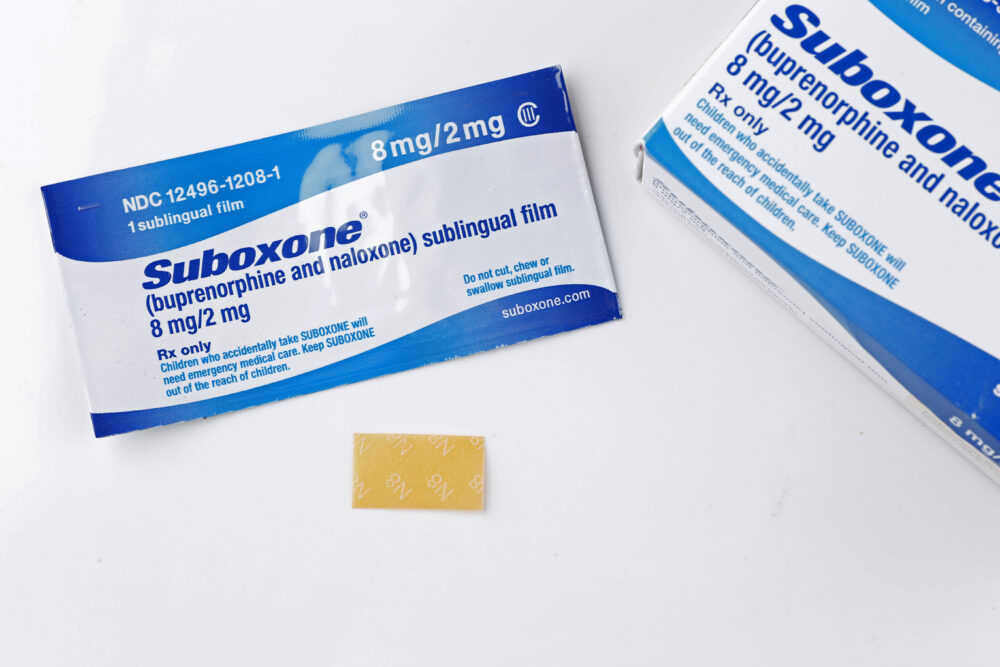
When Lindsay Haddad was first prescribed Suboxone film in 2013, to help overcome her opioid addiction, she was never warned that the treatment may leave her with disfiguring tooth decay and dental injuries, according to a lawsuit recently filed against the manufacturer.
The complaint (PDF) was brought in the U.S. District Court of Western Pennsylvania on January 12, pursuing damages against Indivioir, Inc. Aquestive Therapeutics, Inc., MonoSol, Rx, LLC, and Reckitt and Benckiser LLC as the defendants, claiming the drug companies failed to include adequate warnings and instructions for users and the medical community, which could have allowed her to avoid Suboxone film tooth decay problems.
Suboxone (buprenorphine and naloxone) was first introduced in 2002, as a dissolvable tablet used for treatment of recovering opioid addicts. However, the drug makers subsequently introduced a sublingual film version of Suboxone in 2013, which has been linked to a staggering number of reports involving permanent damage to teeth.
It was not until June 2022 that the drug makers provided updated warnings about Suboxone dental erosion problems, disclosing for the first time that use of the drug was associated with irreversible tooth damage, and instructing doctors to monitor dental health during treatments. However, Haddad indicates that was too late for her teeth, which had already experienced permanent damage from Suboxone film side effects.
Haddad now joins a growing number of individuals pursuing Suboxone film tooth decay lawsuits against the manufacturers, alleging that they could have avoided permanent tooth damage and substantial dental work if earlier warnings had been provided for consumers and the medical community.
Suboxone Lawsuit
Lawsuits are being pursued by users of Suboxone who experienced tooth loss, broken teeth or required dental extractions. Settlement benefits may be available.
Learn More About this Lawsuit SEE IF YOU QUALIFY FOR COMPENSATION“During the relevant time periods, Plaintiff and Plaintiff’s physicians were given no warning and had no knowledge of the serious risk of dental erosion and decay Suboxone film posed,” the lawsuit states. “Subsequently, and as a result of Plaintiff’s prescribed use of Suboxone film, Plaintiff now suffers from permanent tooth damage and/or had substantial dental work performed to repair the damage caused by Suboxone film.”
The lawsuit accuses the manufacturers of concealing or failing to completely disclose their knowledge of the risk of Suboxone tooth damage through the use of appropriate dental warnings. The lawsuit also indicates the manufacturers failed to test Suboxone film strips adequately and failed to instruct healthcare professionals how to safely monitor and identify signs of potentially serious dental complications from Suboxone film.
“Defendants marketed Suboxone film as a maintenance drug, knowing that Suboxone film patients would use the product for extended time periods, yet failed to warn of the risk of extended use of Suboxone film posed to dental health,” Haddad’s lawsuit indicates. “Due to the absence of any warning by Defendants as to the significant permanent health and safety risks posed by Suboxone film, Plaintiff was unaware that Suboxone film could cause serious and permanent dental injuries, as this danger was not known to Plaintiff, Plaintiff’s healthcare providers, or the general public.”
Haddad presents claims of strict product liability, negligent failure to provide adequate warnings and instructions, defective design, negligent design defect, and seeks both compensatory and punitive damages.
January 2024 Suboxone Film Lawsuit Update
Given common questions of fact and law raised in the complaint filed by Haddad and other consumer, the U.S. Judicial Panel on Multidistrict Litigation (JPML) is scheduled to hear oral arguments next week, to determine whether to centralize all Suboxone tooth damage lawsuits before one judge for coordinated discovery and pretrial proceedings.
In complex pharmaceutical litigation, where large numbers of claims are brought by users of the same medication or medical product, each experiencing the same or similar injuries, it is common for the U.S. JPML to centralize the litigation to reduce duplicative discovery into common issues that will arise in all claims, avoid conflicting pretrial rulings and to serve the convenience of certain witnesses and parties who will be required to testify in each of the lawsuits.
As Suboxone tooth decay lawyers continue to review and file claims for individuals who experienced dental erosion after receiving the opioid addiction treatment, it is ultimately expected that hundreds of claims will be brought throughout the federal court system.
After the hearing, the JPML will determine whether to transfer the claims to one court and confirm the judge appointed to preside over the proceedings. However, if Suboxone tooth damage settlements are not reached following coordinated pretrial proceedings in the MDL, each individual claim ma later be remanded back to the U.S. District Court where it was originally filed for separate trial dates.





0 Comments