Eligible for a Suboxone lawsuit?
Suboxone Tooth Damage Lawsuit Indicates Drug Maker Knew About Dental Erosion Risks, Yet Failed to Warn Users or Doctors
Despite medical case studies and consumer complaints involving tooth damage from Suboxone, lawsuit indicates that the drug maker waited years before being forced to add new warnings

According to allegations raised in a recently filed product liability lawsuit against Suboxone manufacturers, users of the opioid addiction drug were not adequately warned about the risk of dental erosion for years, which has left hundreds, if not thousands, of individuals with severe and permanent damage to their teeth.
The complaint (PDF) was filed by Jeremy Schie in the U.S. District Court for the Northern District of Ohio on October 16, indicating that Indivior, Inc., Aquestive Therapeutics, Inc. MonoSol Rx, Inc., and Reckitt Benckiser LLC. knew or should have known about the Suboxone tooth damage risks, yet withheld information from users and the medical community prescribing the drug.
Suboxone (buprenorphine and naloxone) was approved by the U.S. Food and Drug Administration (FDA) in 2002, for treatment of recovering opioid addicts; helping them avoid withdrawal symptoms while undergoing therapy to help them break their addiction. While originally sold as a tablet, the drug makers subsequently introduced Suboxone film, which users place beneath the tongue until it is dissolved.
Schie indicates that the drug makers continued to market and promote the treatment as safe and effective years, despite an influx of adverse event reports and published case studies describing devastating tooth damage among users. It was not until early 2022 that Suboxone dental side effects were added to the label, after the FDA identified more than 300 cases of tooth decay and dental problems reported to the agency.
Suboxone Lawsuit
Lawsuits are being pursued by users of Suboxone who experienced tooth loss, broken teeth or required dental extractions. Settlement benefits may be available.
Learn More About this Lawsuit SEE IF YOU QUALIFY FOR COMPENSATIONSchie, of Ohio, indicates he was prescribed Suboxone by a doctor to help break an opioid addiction he acquired when being treated for pain management. Neither he, nor his doctor, were warned about the risk of serious dental erosion linked to Suboxone use by the drug’s label or the manufacturers, the lawsuit states.
The lawsuit points out that medical literature began to be published as early as 2012, which included case reports of dental erosion linked to Suboxone use. However, that should have confirmed what the manufacturers already knew from hundreds of complaints of Suboxone tooth damage it had received through the years. Despite that knowledge, the makers of Suboxone failed to provide adequate warning labels for patients or the medical community, Schie claims.
“Suboxone is a combination of buprenorphine and naloxone designed to be ingested through oral absorption either as a tablet or film. Buprenorphine is acidic. And the inclusion of buprenorphine in a dissolvable form leads to dental erosion and decay,” the lawsuit notes. “Defendants knew or should have known that Suboxone, when used as prescribed and intended, causes harmful damage to the teeth due to the acidity of buprenorphine.”
According to the complaint, Schie has had substantial dental work performed to repair Suboxone tooth damage, some of which is permanent.
Previous Suboxone Lawsuit Settlements
Originally designed as a dissolving pill, Indivior allegedly waited until its patent exclusivity was about to expire before introducing Suboxone film versions in 2009, which has been linked to the greatest risk of dental erosion side effects.
Schie and other plaintiffs pursuing a Suboxone tooth damage lawsuit now claim the introduction of the film version of the drug was not done to benefit patients, but to help the drug maker avoid generic competition and extend patent protections.
The federal government and various states previously pursued a lawsuit against Suboxone manufacturers, claiming that it illegally sought to control supply and inflate prices for the critical opioid addiction drug, including what state Medicaid programs paid for it.
In 2019, Reckitt Benckiser Pharmaceuticals reached a $700 million settlement with the federal government and states over Suboxone Medicaid fraud allegations. Indivior spun off from the company shortly afterwards and reached a $600 million Suboxone settlement agreement with the Justice Department in 2020, to resolve claims of aggressive and deceptive marketing. Then, in 2021, the company agreed to pay another $300 million to settle similar claims filed by all 50 states and the District of Columbia.
Find Out If You Qualify for Suboxone Tooth Decay Compensation
6 Comments
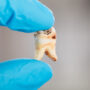





FranNovember 21, 2023 at 12:36 pm
I've been on suboxone for many years. I'm addicted to it now. I had 4 teeth pulled in the front where I would put the strip under my tongue. I had to get a bridge whi h was very expensive.
ErinOctober 27, 2023 at 5:32 pm
6 years ago I had never had a cavity in my life. I had a beautiful white smile. Now I'm missing 9 teeth. I have a cavity on every tooth that I have. My teeth are yellow and black and there's no white to them at all. I can barely eat Anything I eat causes them to crumble and break. I sometimes wake up with bits of teeth enamel in my mouth
JamesOctober 22, 2023 at 6:01 am
I had great teeth before starting it!
SladdonOctober 21, 2023 at 8:22 pm
I had a tooth break/chip, I had to have it removed, paid by myself I do not have insurance. Now I have several very deep cavities and have constant severe tooth pain
ElizabethOctober 20, 2023 at 7:57 pm
I started suboxen 2005 due to pregnancy and being on oxycotins. I was never told there could be a issue with the naxlone while pregnant nor was ever offered subtext. My teeth started to wear break get infected. Fall out. I had to get them all pulled out.
JohnOctober 19, 2023 at 8:40 pm
I have severely damaged teeth.from the use of Suboxone strips.