ADHD Drug Use Increasing Among Young Women, Trend Carries Pregnancy Risks: CDC

A new study by federal health experts indicates that more young women are using attention deficit and hyperactivity disorder drugs like Ritalin and Vyvanse, and warn that the use of these drugs may have adverse health effects on them and their unborn children if they become pregnant.
The U.S. Centers for Disease Control and Prevention (CDC) released the findings in a study published in the Morbidity and Mortality Weekly Report (MMWR) on January 19, calling for additional research into the potential side effects of ADHD drugs on women of reproductive age, amid significantly increased use among that population.
The study used the Truven Health MarketScan Commercial Database to look at data on outpatient pharmacy prescription drug claims for ADHD drugs from 2003 to 2015 among women between the ages of 15 and 44 who were enrolled in private health insurance.
Learn More About
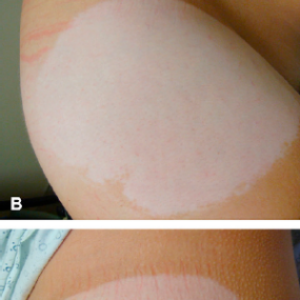
Side Effects of Daytrana Patch May Cause Permanent Skin Color Changes and Scarring.
According to the findings, prescriptions of ADHD drugs among reproductive-aged women increased by 344% from 2003 through 2015. The findings indicate that the increase was specifically among stimulant-based ADHD drugs, and did not affect non-stimulant ADHD drugs like Strattera. The study did not look at why the drugs were prescribed.
The study found that the increase was among all age groups overall, but the highest increase during the period of the study was among women from ages 25 through 29, who saw a 700% increase in the use of ADHD drugs.
The highest increases were among prescriptions of methylphenidate-based drugs like Ritalin and Daytrana, and lisdexamfetamine-based drugs like Vyvanse.
“ADHD medication use among pregnant women is increasing, but consensus about the safety of ADHD medication use during pregnancy is lacking,” the CDC states in the study. “Given that nearly half of U.S. pregnancies are unintended, and early pregnancy is a critical period for fetal development, examining trends in ADHD medication prescriptions among reproductive-aged women is important to quantify the population at risk for potential exposure.”
The findings come about a month after the release of a study in Pediatrics, where researchers from Sweden found a link between the pregnancy use of ADHD drugs and birth complications such as central nervous system disorders and seizures.
That study found that the use of ADHD drugs during pregnancy resulted in a 50% increased risk of admission to a neonatal intensive care unit (NICU).
ADHD Drug Overuse Concerns
The findings come amid increasing concerns over the perceived overprescribing of ADHD drugs.
About 15% of all high school-age children in the United States have been diagnosed with ADHD, but some experts say that number should be closer to 5%.
One early advocate of stimulate treatment for children with ADHD, Dr. Keith Conners of Duke University, said that the rate of children now being diagnosed with ADHD and placed on drug treatments is “preposterous” and called ADHD an epidemic manufactured by drug companies.
Conners and others say that the inflated diagnoses and prescriptions are the result of a 20 year effort by the pharmaceutical industry to cash in on concerned parents hoping that poor grades and typical childhood behavior can be cured with drugs.
While some manufacturers paid off doctors to speak on their drugs behalf, others have gone as far as releasing comic books encouraging children to take medication to address ADHD. At some point since 2000, Conners noted that the FDA has cited every major ADHD drug manufacturer for false and misleading advertising about their ADHD drugs.
Those efforts led to $9 billion in sales for the ADHD drug industry in 2012, and 3.5 million children using ADHD medications.





0 Comments