Cochlear Recall Issued for Nucleus CI500 Ear Implant
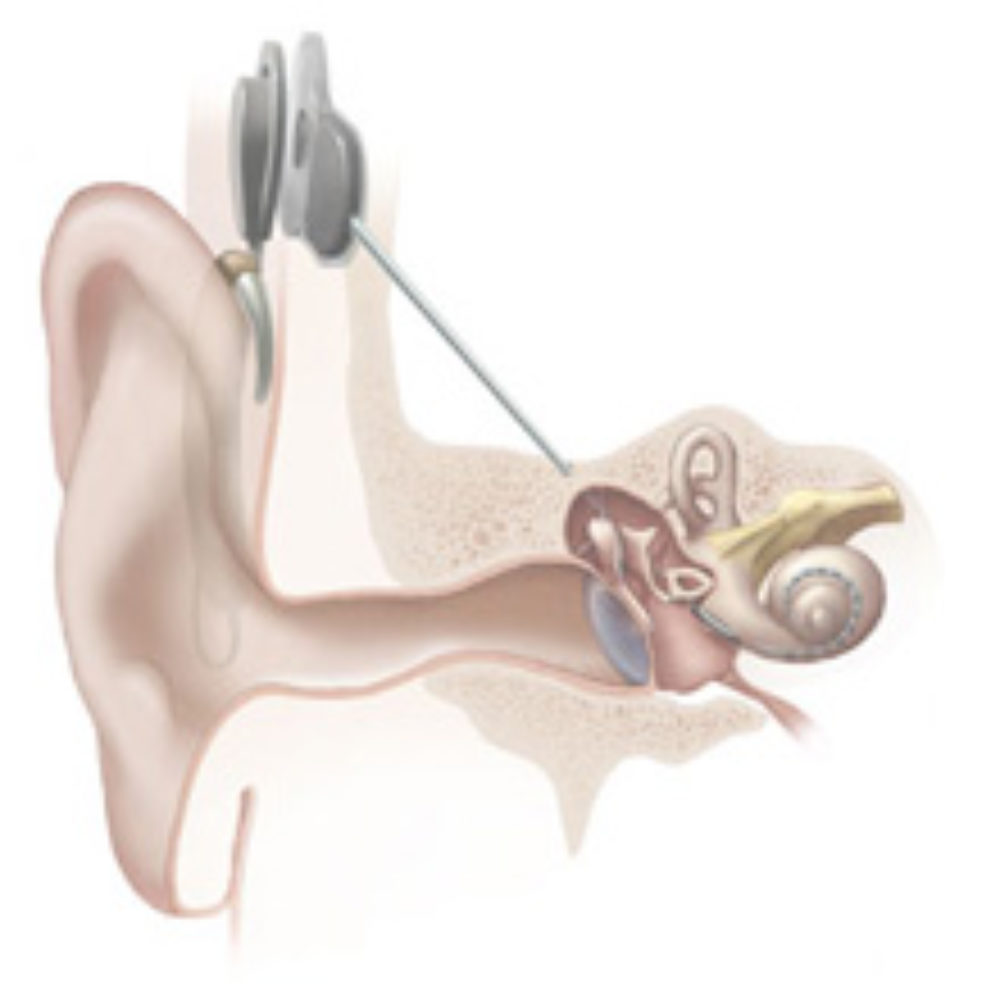
Cochlear Limited has announced a worldwide recall for its Nucleus CI500-series ear implants, after receiving an increasing number of reported failures.
The manufacturer of the popular cochlear ear implants issued a press release announcing the Nucleus CI500 cochlear ear implant recall, indicating that less than 1% of the CI512 implants had failed since the devices were put on the market in 2009. However, a recent increase in failure reports has led the company to recall the devices and launch an investigation. The press release does not say how many devices have failed or how many have been implanted in patients around the world.
Company officials have said the devices are shutting down unexpectedly, but they do not know the cause of the problem. They also indicated that the recalled Cochlear implants are unlikely to cause problems for recipients beyond the bionic ear failing.

Did You Know?
Millions of Philips CPAP Machines Recalled
Philips DreamStation, CPAP and BiPAP machines sold in recent years may pose a risk of cancer, lung damage and other injuries.
Learn MoreThe Cochlear recall affects all Nucleus CI500-series cochlear implants. However, individuals who already have the implant can continue to use their system as normal. If the implant fails, the company says it will safely shut down and recipients can be re-implanted with the Nucleus Freedom cochlear implant, which is not affected by the recall.
A cochlear implant, or bionic ear, is an electrionic device that is surgically implanted to provide a senses of sound for individuals who are profoundly deaf or hard of hearing.
Last November, Advanced Bionics recalled some of the HiRes 90k cochlear implants due to defects that could cause pain and loud noises for recipients. There were at least two confirmed cases where patients experienced malfunctions that resulted in severe pain, overly loud sounds and shocking sensations less than two weeks after receiving the implants.
Photo via Wikimedia Commons, public domain work of the National Institutes of Health
Want a weekly update on top lawsuits, recalls & warnings?
"*" indicates required fields
3 Comments




RodneyOctober 25, 2016 at 8:22 pm
I have bilateral implants. Cochlear. I have been experiencing headaches, super loud roaring noises, sensitivity shocks me and even will cause me to stumble. My stability/balance is like full time virtigo. Surgeon said there is nothing he can do. Been to 4 ENTS they brush me off. Also pressure in ears extreme...
MarvinApril 8, 2015 at 3:31 am
implant failed, now constant ringing in ear and head hurts
JoaquínNovember 25, 2012 at 6:34 am
My wife had such a problem with this Cochlear CI500 implant recall. It started to fail, and she had to be re-implanted, with the complex lenghty and risky surgery. I wonder if there is somebody else in this situation, and whether somebody ahd lawsuits about this. Maybe is there a group, blog or similar, of people affected by this? I live in Spain, the surgery was done here. Thanks.