Doctors Should Warn Patients About Elmiron Vision Complications and Consider Alternative IC Therapies: Study
Researchers warn in a new study that concerns over Elmiron vision complications and side effects are the main reason patients stop using the interstitial cystitis (IC) drug, indicating doctors should warn about the potential risks and consider alternative therapies for the bladder condition before prescribing the drug.
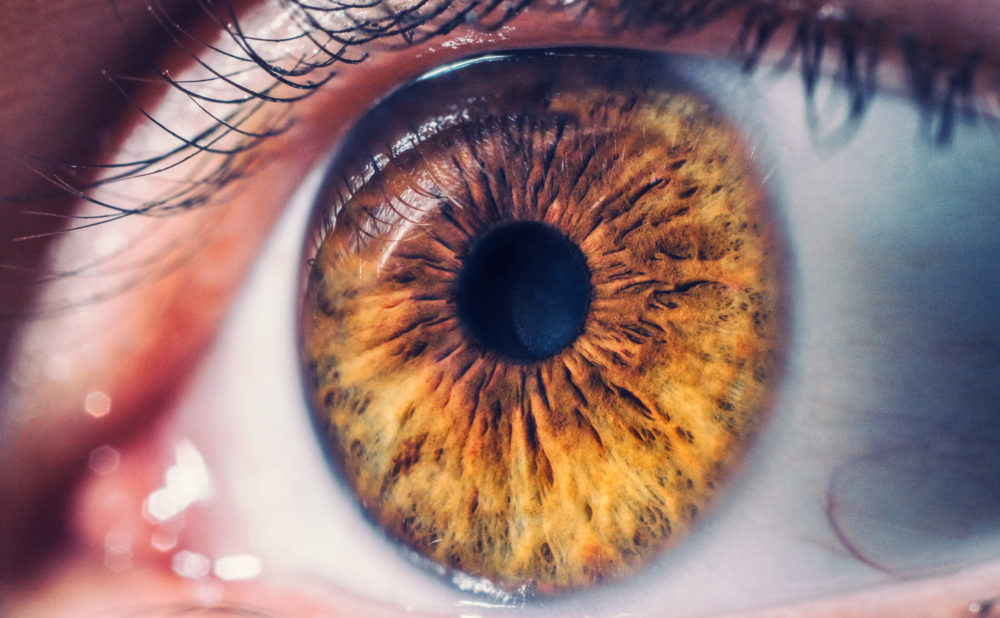
Elmiron (pentosan polysulfate sodium or PPS) is a prescription medication which has been on the market since 1996, for treatment of interstitial cystitis or “painful bladder syndrome”. Since there is no cure for this condition, many users remain on the drug for years, if not decades, unaware they may be causing irreversible damage to their eye sight and vision.
In June 2020, the Elmiron warning label was updated to provide information about the potential vision complications for the first time, indicating a number of users have been diagnosed with a retinal condition as pigmentary maculopathy, which can result in blurry vision, dark spots, difficulty adjusting in dim light, as well as blindness. However, increasing evidence suggests the drug makers knew or should have known about this risk, and withheld warnings and information from consumers, urologists and ophthalmologists.
Learn More About
Side effects of Elmiron have been associated with vision loss and retina damage known as pigmentary maculopathy.
Learn More About this Lawsuit SEE IF YOU QUALIFY FOR COMPENSATIONIn this latest study, researchers from the University of California, Los Angeles, found that two out of five patients who were prescribed Elmiron and discontinued taking it said they did so due to concerns over damage to their eyes. The findings, published in the September issue of the Journal of Urology, also found that those who had confirmed eye injuries took higher doses than those who did not.
The researchers looked at data on 64 patients who underwent screening examination at the UCLA Stein Eye Institute. They contacted those patients and surveyed them on their use of Elmiron.
According to the study, of the 64 patients, 37 were no longer taking the drug. The findings indicate patients who were diagnosed with maculopathy were more likely to have discontinued the drug, and reported having taken daily doses in excess of the recommended 300 mg daily dose. They also had a significantly higher mean lifetime cumulative dose.
The researchers found that the primary reason for discontinuing Elmiron use was concern for ocular injury, which was the main reason 41% of those who quit using Elmiron did so. The study indicates 32% quit using Elmiron due to lack of efficacy or decreased benefit, 16% ceased Elmiron use due to cost, and 11% quit due to other side effects.
“While the mechanism of maculopathy in PPS use is not fully understood, this risk must be weighed against its potential benefit as a treatment for interstitial cystitis,” the researchers concluded. “Physicians should counsel patients on risk of ocular complication in long term and high dose PPS use, and consider alternative therapies and early ophthalmologic referral in patients with lifetime cumulative doses greater than 500 g.”
Elmiron Vision Lawsuits
Johnson & Johnson now faces more than 360 Elmiron lawsuits brought in federal and state courts nationwide, each involving similar allegations that the bladder drug left users with permanent vision damage, and the size and scope of the litigation is expected to increase dramatically over the next year.
Given common questions of fact and law raised in individual injury claims and Elmiron class action lawsuits pending throughout the federal court system, the litigation has been centralized in the U.S. District Court for the District of New Jersey, where one judge is presiding over coordinated discovery and pretrial proceedings.
As Elmiron lawyers continue to review and file claims in the coming months and years, it is ultimately expected that several thousands claims will eventually be included in the litigation.
In the coming years, it is expected that a small group of representative cases will be selected for early “bellwether” trials to help the parties gauge how juries will respond to the evidence and testimony that will be repeated throughout the claims.
While the outcome of these early bellwether trials will not be binding on other claims they will likely have a big influence on potential in Elmiron settlements the drug makers may pay to avoid the need for hundreds of individual cases to be remanded to U.S. District Courts nationwide for individual trials in future years.
Get more articles like this sent directly to your inbox.
"*" indicates required fields






0 Comments