Philips Faces Inspections and Potential Fines, As FDA Monitors CPAP Recall Repair/Replacement Program
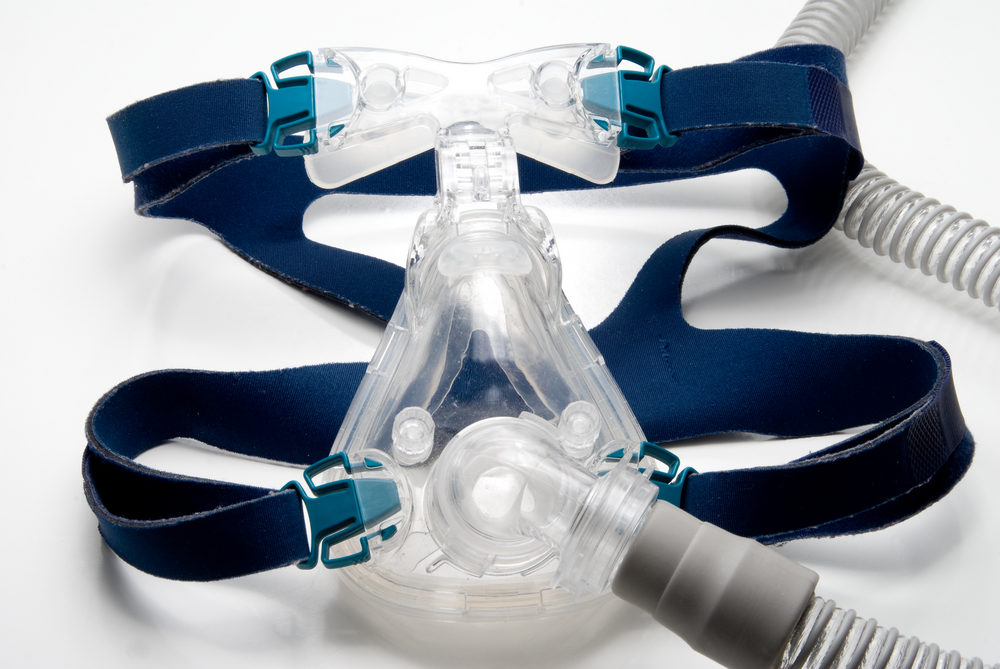
Following a massive recall of Philips CPAP machines and other breathing devices, consumers continue to remain in the dark about when and if their devices will be replaced or repaired. However, late last week, the FDA issued an update to address some of the concerns being expressed by consumers.
In response to Frequently Asked Questions, the FDA provided information on Friday about the manufacturer’s plans for repairing and replacing CPAP machines. The agency also indicates Philips will face inspections, monitoring, and could face potential fines as it works to address problems caused by a toxic polyester-based polyurethane (PE-PUR) sound abatement foam used in millions of devices sold in recent years.
Customers have already brought a growing number of class action lawsuits alleging the recent Philips CPAP recall left them without much needed sleep apnea therapy, and with no indication whether the company would repair or replace the recalled machines.

Philips CPAP Recall Lawsuit
Millions of recalled Philips DreamStation, CPAP, BiPAP and ventilator machines may release toxic foam particles and chemicals into the air pathway.
Learn More About this Lawsuit See If You Qualify For CompensationAn estimated 3.5 million Philips Respironics DreamStation, CPAP, BiPAP and mechanical ventilators were removed from the market in June, after it was discovered that defective sound abatement foam used in the machines may degrade and release black particles or toxic chemicals in the lungs of users who rely on the machines for treatment of sleep apnea or other conditions. As a result of the serious health risks, consumers were immediately told to stop using the CPAP machines, even though alternative devices were not available.
Philips and the FDA warn that the PE-PUR foam can release toxic particles and gases, which have been linked to reports of cancer, severe respiratory problems and other health complications, according to allegations raised in a number of Philips CPAP machine lawsuits and class action claims filed in courts nationwide in recent weeks.
Last week the company received authorization from the FDA to “rework” recalled first-generation DreamStation CPAP devices, and the FDA’s updated FAQ provides some answers to customers on how the program will work and what they should expect.
According to the new information provided by the federal regulators, Philips plans to begin reworking the recalled devices this month, and has already started replacing certain first-generation DreamStation CPAP machines with second generation devices that do not contain PE-PUR foam. It intends to complete all repairs and replacements within the next 12 months.
The FDA indicates implementation of the plan is a “high priority” for the agency.
“The FDA is committed to using every tool at our disposal to increase the availability of these medical products,” the FDA FAQ states. “The FDA is working with Philips Respironics to monitor the repair or replacement of impacted devices as expeditiously as possible and is continuing to gather information to inform our actions. We are collaborating with other manufacturers and government partners to support availability of CPAP and BiPAP machines.”
However, the agency also noted Philips Respironics has not yet given the agency all of the information it needs to evaluate its plans to repair all breathing machine devices affected by the recall, such as Trilogy ventilators, A-series BiPAP machines and others.
In addition, the agency announced in the FAQ that it has initiated on-site inspections of Philips Respironics’ manufacturing facilities in order to determine whether they are in compliance with federal regulations and said the company could potentially face fines and other penalties.
“If a firm’s voluntary action is not rapid or complete, the FDA has the option to take enforcement action,” the FAQ states. “The FDA has identified this as a Class I recall, the most serious type of recall, and the FDA will continue to work with the company to ensure that they provide sufficient evidence demonstrating the safety and effectiveness of its proposed actions to correct the product defect.”
While waiting for repair and replacement of CPAP devices, the FDA indicated most patients should talk with their doctor about the potential risk, and should continue using them as long as their healthcare provider determines the benefits outweigh the risks in their individual situation.
Philips CPAP Lawsuits
Later this month, the U.S. Judicial Panel on Multidistrict Litigation (JPML) will hear oral arguments on the CPAP recall lawsuits, to decide whether to consolidate and centralize claims filed in federal courts nationwide before one judge for pretrial proceedings.
Consolidation is common in complex product liability litigation, where a large number of claims have been presented by former users of the same product, alleging similar injuries. Centralizing the Philips CPAP/BiPAP claims would help avoid duplicative discovery, prevent contradictory rulings from different judges, and serve the convenience of the parties, witnesses, and the courts, according to the motion.
Those affected by the recall can register their devices at www.philips.com/src-update.
Get more articles like this sent directly to your inbox.
"*" indicates required fields
1 Comments






JohnNovember 29, 2021 at 8:08 pm
I for one do not want someone else’s DreamStation that has been repaired. I either want my DreamStation repaired and returned to me or a new one.