Magnevist MRI Contrast Agent May Cause Hypersensitivity Reactions: FDA
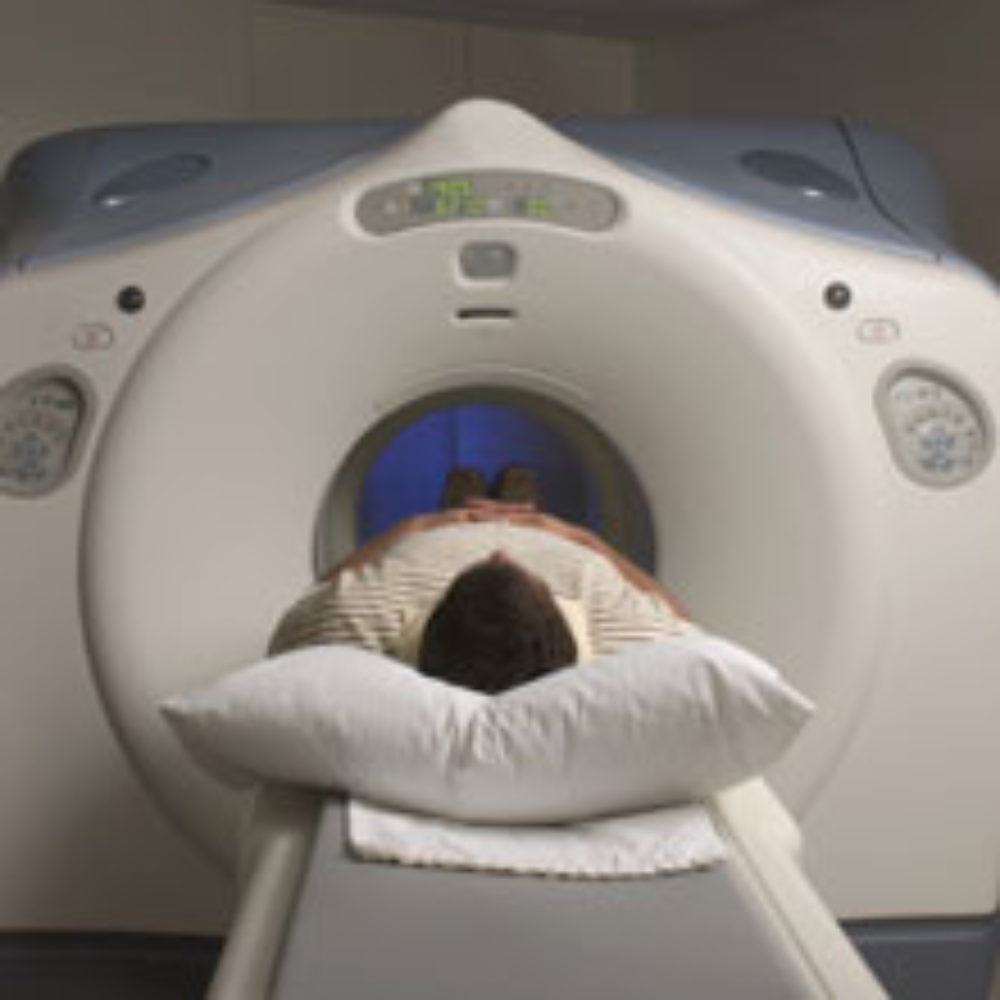
Federal drug regulators are warning that some patients may have a hypersensitivity to Magnevist, an MRI contrast solution used to improve the outcome of MRI scans.
The FDA has updated the Magnevist label warnings to indicate that some patients have shown signs of severe hypersensitivity to the drug. Healthcare providers have been advised to keep resuscitative equipment immediately available in case such a reaction occurs.
The hypersensitivity reactions from the Magnevist MRI contrast agents are not always immediate, the FDA noted, with some delayed hypersensitivity occurring several hours after the solution was administered.

Did You Know?
Millions of Philips CPAP Machines Recalled
Philips DreamStation, CPAP and BiPAP machines sold in recent years may pose a risk of cancer, lung damage and other injuries.
Learn MoreMagnevist (gadopentetate dimeglumine) is a gadolinium-based contrast agent (GBCA) manufactured by Bayer Healthcare. Gadolinium-based contrast agents are solutions given to patients before they take a magnetic resonance image (MRI) in order to get better results.
The FDA warned that the risk of Magnevist hypersensitivity appeared to be higher in patients with a history of reactions to contrast solutions, bronchial asthma or allergic disorders. The reactions can occur with or without prior exposure to GBCAs.
In recent years, use of Magnevist and other gadolinium MRI contrast agents have also been linked to a risk of a severe and potentially life-threatening reaction, known as nephrogenic systemic fibrosis (NSF), which is known to occur among individuals with impaired kidney function. This reaction causes a hardening and thickening of the skin and other tissue throughout the body.
In September 2010 the FDA banned the use of Magnevist on patients with kidney problems due to the risk of NSF. The agency also put similar restrictions on GE Healthcare’s Omniscan and Covidien’s Optimark.
Several hundred lawsuits over nephrogenic systemic fibrosis have been filed in the United States by individuals who developed the condition after receiving a gadolinium-based contrast agent.
Get more articles like this sent directly to your inbox.
"*" indicates required fields



0 Comments