Opioids To Carry Naloxone Overdose Drug Information On Labels: FDA
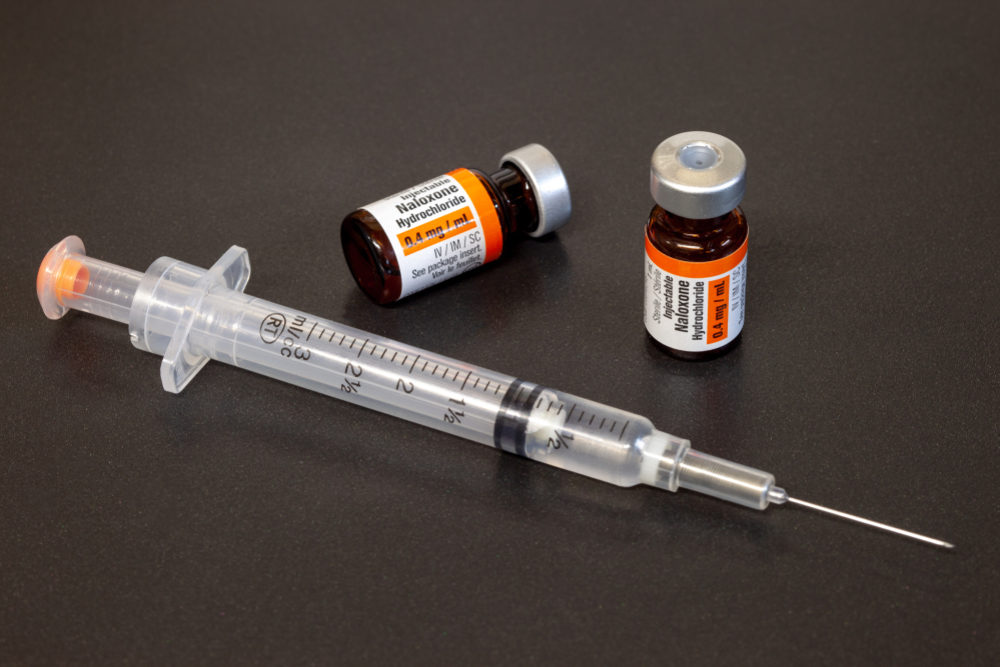
To help battle the continuing opioid abuse epidemic in the United States, federal regulators issued new requirements for drug manufacturers to place information about the overdose drug Naloxone on the warning labels for opioid medications, and the agency is calling on doctors to educate patients on the benefits of the overdose reversal drug.
The new FDA requirements were announced in a MedWatch Safety Alert on July 23, which also indicates that patient medication guides for opioid-based painkillers will be updated to provide information about Naloxone.
The requirements are part of an effort to help ensure doctors discuss the availability of the potential life-saving drug for individuals taking opioids. It can also help doctors assess each patient’s need for a naloxone prescription when opioids are prescribed, or if medicines to treat opioid use disorder are being prescribed or renewed.

Did You Know?
Millions of Philips CPAP Machines Recalled
Philips DreamStation, CPAP and BiPAP machines sold in recent years may pose a risk of cancer, lung damage and other injuries.
Learn MoreOpioids are strong narcotic painkillers used to help manage severe pain when other treatments and medicines are not able to provide enough pain relief. However, opioids do have risks, including misuse and abuse, addiction, overdose and death.
Naloxone is a generic drug that has been available for more than 40 years, and is designed to counter overdoses of heroin and prescription opioids such as hydrocodone, oxycodone, codeine, and tramadol. The drug is considered safe among government health officials and has a low risk of serious side effects.
Opioids account for more than 70% of drug overdose deaths and doctor overprescribing has been largely to blame for stemming the opioid abuse crisis in the U.S. Opioids are prescribed 30% of the time without a documented pain diagnosis.
In recent years, the opioid abuse epidemic has been fueled by lax oversight, as studies have found that many patients were given excessive painkillers following various surgeries, even though there may not be a need for the drugs to control pain.
Earlier this year, a study emphasized the need for doctors to prescribe the least amount of opioid painkillers as are necessary.
Because of the worsening abuse epidemic the FDA is encouraging doctors and health care providers to raise awareness of the availability of naloxone to prevent opioid overdose when prescribing painkillers.
In 2014, the World Health Organization indicated an estimated 20,000 lives could be saved annually in the United States if the drug were more prevalent.
In 2018, the Surgeon General issued a statement urging Americans to become familiar and carry naloxone as a response to the growing opioid abuse crisis. The warning indicated many American lives can be saved by naloxone becoming more accessible to the public.
Get more articles like this sent directly to your inbox.
"*" indicates required fields




0 Comments