Injured by Ozempic, Wegovy or Mounjaro?
Lawsuits Over Ozempic, Wegovy, Mounjaro and Other GLP1-RA Drugs May Hinge on Label Warnings
Lawyers indicate that that questions about whether the GLP-1 RA medication label warnings adequately disclose the risk of gastroparesis and other severe injuries will play a large role in the growing litigation.

In position papers recently submitted, lawyers involved in lawsuits over Ozempic, Wegovy, Mounjaro and other similar medications detailed their stances on whether manufacturers provided adequate label warnings about the potential stomach paralysis side effects some users are experiencing, and how the growing litigation should be managed during pretrial proceedings.
Millions of Americans have used Ozempic, Wegovy, Mounjaro or other glucagon-like peptide-1 receptor agonists (GLP-1 RA) drugs in recent years, amid aggressive promotions by the drug makers that indicated the medications were safe and effective for weight loss. However, former users are now pursuing Ozempic lawsuits, Wegovy lawsuits and Mounjaro lawsuits against the manufacturers, after developing painful and debilitating injuries, including intestinal blockages and a form of stomach paralysis known as gastroparesis.
Given common questions of fact and law raised in the litigation, the U.S. Judicial Panel on Multidistrict Litigation decided earlier this year to centralize all GLP-1 lawsuits before U.S. District Judge Gene E.K. Pratter in the U.S. District Court for the Eastern District of Pennsylvania, and it is widely expected that tens of thousands of lawsuits may ultimately be included in the federal MDL (multidistrict litigation).
Find Out If You Have a Lawsuit
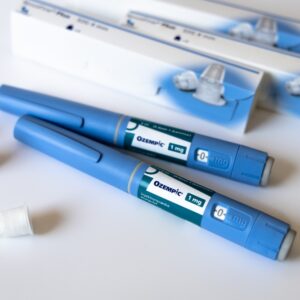
Lawyers are pursuing Ozempic lawsuits, Wegovy lawsuits and Mounjaro lawsuits over gastroparesis or stomach paralysis, which can leave users with long-term gastrointestinal side effects
Learn More About this Lawsuit See If You Qualify for CompensationGLP-1 RA Drug Concerns
Ozempic (semaglutide) is the most widely used of this new class of medications, and was originally introduced by Novo Nordisk as a treatment for Type 2 diabetes. However, the drug maker has been accused of engaging in “unprecedented marketing”, which promoted the weight loss benefits, leading to the widespread use of Ozempic as a diet drug.
As a result of the popularity of Ozempic for weight loss, Novo Nordisk has introduced a higher dose version under the brand name Wegovy, which is specifically approved as a diet drug. However, it contains the same active ingredient.
Eli Lilly has also released competing GLP-1 drugs in the form of Mounjaro (tirzepatide), approved for diabetes treatment in 2022, and a weight loss version of the drug was approved by the FDA last year, which is marketed under the brand name Zepbound.
Although advertisements and promotional materials suggested that the drugs carry few serious side effects, former users claim that the manufacturers withheld critical safety information and warnings to increase profits from the drugs.
Parties Debate Adequacy of GLP-1 Label Warnings
On April 9, lawyers representing the drug makers and plaintiffs involved in the litigation each submitted position papers to Judge Pratter, explaining their differing opinions on the litigation, which will largely focus on whether the label warnings for Ozempic, Mounjaro, Wegovy and Zepbound adequately disclosed the risks of gastroparesis and other intestinal injuries.
Plaintiffs’ position paper (PDF) indicates that the labels failed to provide enough information, particularly given the rising popularity and widespread use of the drugs.
“The labels for GLP-1 RAs have never adequately warned of the drugs’ propensity to impair the ability to digest food or that relentless vomiting requiring hospitalization was a risk,” plaintiffs noted. “No GLP-1 RA labels warn of the risk of gastroparesis, a paralysis of the stomach resulting in cessation of digestion. Rather, the labels downplay the severity of gastroparesis symptoms, and omit that they can be a feature of potentially life-threatening digestive impairment.”
Plaintiffs also noted that defendants failed to warn users that they need to stay on the drugs for the rest of their lives, or else they are likely to gain the weight back.
In a separate position paper (PDF) submitted by the drug makers, the Defendants maintain the label warnings were adequate, even though they never mention gastroparesis specifically, and argue that each plaintiff bringing a lawsuit over Ozempic, Wegovy or Mounjaro should be required to undergo a specific gastroparesis diagnostic test before they are allowed to assert claims that they suffered stomach paralysis injuries.
The manufacturers’ attorneys indicate that they intend to fight to have some of these cases, and some of the common claims, such as manufacturing defect claims dismissed. The position paper notes that they expect to seek summary judgment on issues such as causation, preemption, statute of limitations, and failure to warn.
In a response (PDF) issued on April 15, plaintiffs argued that discovery will reveal how much the manufacturers, Eli Lilly and Novo Nordisk, knew about the risks of GLP-1 RA gastroparesis risks, demonstrating that they intentionally downplayed those risks to patients and the medical community.
“Both Novo and Lilly claim in their Position Statements that their GLP-1RA labels warn of the injuries alleged in this case, such as gastroparesis and ileus,” plaintiffs noted in their response. “But that is inaccurate. In the warning section of a label (governed by specific FDA regulations), the mere reference to potential general gastrointestinal symptoms is insufficient to warn of the risks of the potential severity and longevity of gastroparesis, ileus, and intestinal blockage to which GLP-1RA users are exposed.”
They propose that the cases should continue to move forward “but not at the expense of fairness.” Plaintiffs oppose any proposal seeking to dismiss claims, or the Court adopting a single diagnostic test as a means of proving causation.
Thousands of GLP-1 Injury Lawsuits Likely
The papers indicate there are currently 88 GLP-1 injury lawsuits brought by 117 plaintiffs involved in the multidistrict litigation, with 100 filed against Novo Nordisk, eight filed against Eli Lilly, and nine claims filed against both. However, these are believed to be a small fraction of the cases likely to be filed in the coming months and years.
In complex pharmaceutical litigation, where large numbers of claims are brought by users of the same medication or medical product, each experiencing the same or similar injuries, it is common for the U.S. JPML to centralize the litigation to reduce duplicative discovery into common issues that will arise in all claims, avoid conflicting pretrial rulings and to serve the convenience of certain witnesses and parties who will be required to testify in each of the lawsuits.
Following coordinated discovery and any bellwether trials scheduled in the MDL, if the parties are unable to agree to GLP-1 RA stomach paralysis settlements or another resolution for the Ozempic injury claims, each individual claim would later be remanded back to the U.S. District Court where it was originally filed for trial.




0 Comments