Stryker Accolade and LFit Hip Lawsuit Filed Over Elevated Metal Ions in Blood, Resulting in Revision Surgery
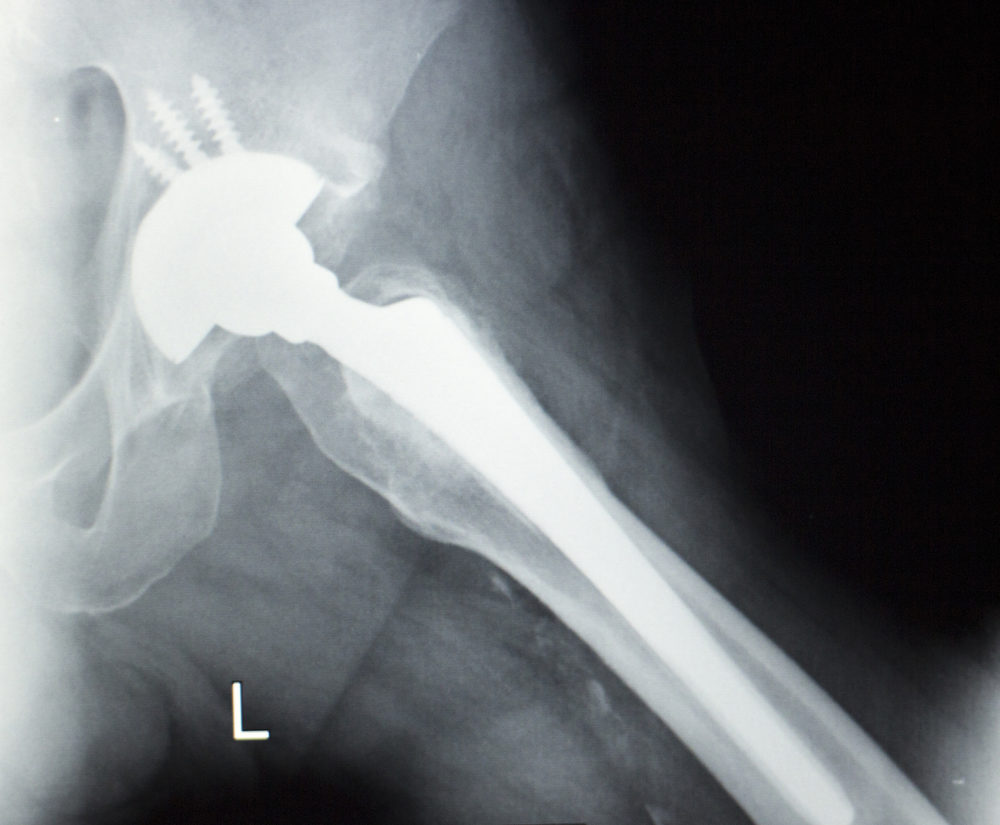
Metal debris shed from Stryker Accolade and LFit hip implant components caused a Maine man to develop elevated metal ions in his blood, which resulted in failure of the hip replacement and the need for revision surgery, according to a recently-filed lawsuit.
Mark Irving and his wife, Lorraine, filed a complaint (PDF) against Howmedica Osteonics Corporation, doing business as Stryker Orthopedics, on November 9 in the U.S. District Court for the District of Maine.
According to the lawsuit, Irving underwent left total hip replacement in December 2011, during which he received a Stryker LFit Anatomic V40 CoCr Femoral Head, with a Stryker Accolade TMZF Plus Hip Stem, among other components. However, by October 2016, he had to undergo revision surgery to have the devices removed due to failure of the hip implant, which was allegedly caused crevice corrosion where the two components met.

Learn More About
Lawsuits are being reviewed for several different dangerous and defective hip replacement systems.
Learn More About this Lawsuit See If You Qualify For Compensation“After implantation, Plaintiff Mark Irving developed pain in his replaced left hip,” the lawsuit states. “Metal ion testing demonstrated elevated cobalt and chromium levels.”
After receiving similar reports involving hip failures, a Stryker LFit v40 hip recall was issued by the manufacturer in August 2016, impacting certain large-diameter femoral heads. At that time, Stryker acknowledged that a higher-then-expected number of individuals were experiencing problems with trunnion failure, metal wear, adverse tissue reactions and other complications.
The case filed by Irving joins a growing number of other hip replacement lawsuits filed in recent months by individuals have suffered complications that resulted in revision surgery only a few years after receiving the implant. In particular, complications with the Stryker Accolade stem in combination with the LFit V40 femoral head have resulted in reports of excessive metallic debris, limited range of motion, disassociation, fractures and failed hip replacements.
Given similar questions of fact and law raised in complaints filed throughout the federal court system, the U.S. Judicial Panel on Multidistrict Litigation (JPML) established coordinated pretrial proceedings earlier this year for all lawsuits involving use of the Stryker LFit v40 head, centralizing the cases before U.S. District Judge Indira Talwani in the District of Massachusetts.
Similar centralized proceedings were previously established in the federal court system following a 2012 Stryker Rejuvenate and ABG II recall, which resulted in thousands of lawsuits brought by individuals nationwide. Following coordinated discovery in that litigation, the manufacturer agreed to pay more than $1 billion in hip implant settlements to resolve cases for individuals who required revision surgery after these recalled implant failed.
Want a weekly update on top lawsuits, recalls & warnings?
"*" indicates required fields






0 Comments