Elmiron Vision Loss Problems May Appear Years After Patients Stop Taking Bladder Pain Drug, Case Report Warns
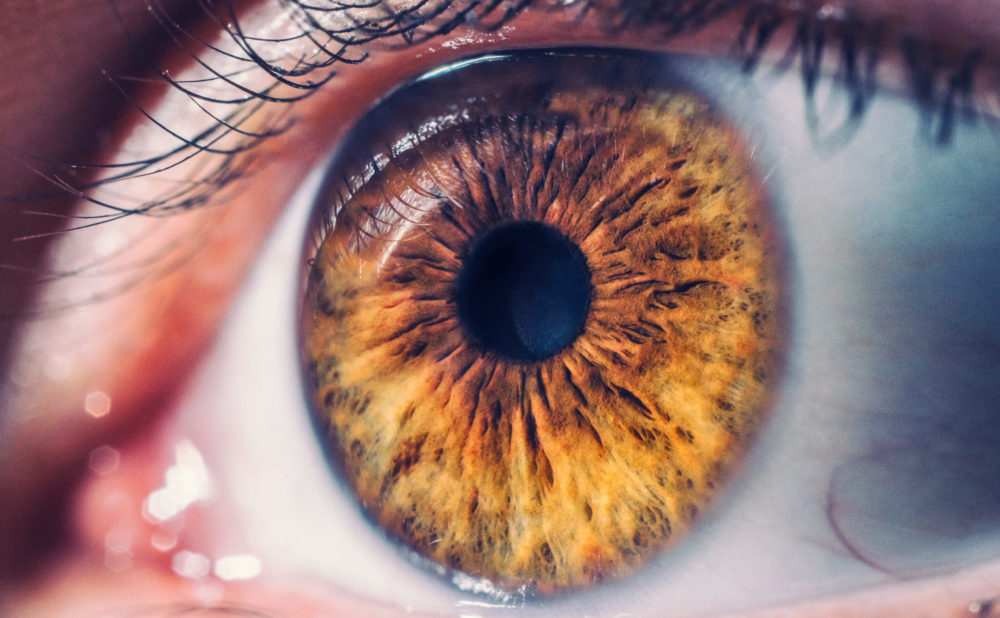
In recent months, a growing number of Elmiron lawsuits have been filed by former users of the interstitial cystitis drug diagnosed with form of retina damage known as pigmentary maculopathy. However, the drug maker may face years of litigation, as a new case report suggests that the vision loss problems may surface years after a patient has stopped the treatment.
In findings published in Retinal Cases & Brief Reports on November 17, doctors at Emory University School of Medicine describe a woman who first showed signs of Elmiron-induced retinal maculopathy three years after she stopped taking the treatment.
Elmiron (pentosan polysulfate sodium or PPS) has been on the market since 1996, and is often taken for years, since there is no known cure of the bladder and pelvis pain associated with interstitial cystitis.
Learn More About
Side effects of Elmiron have been associated with vision loss and retina damage known as pigmentary maculopathy.
Learn More About this Lawsuit SEE IF YOU QUALIFY FOR COMPENSATIONAlthough independent reports published in recent years have suggested a link between Elmiron and vision problems, the drug maker did not add information about the risk to the drug label until earlier this year.
As a result, dozens of product liability lawsuits have been filed by former users who indicate they have been left with permanent retina damage, typically involving difficulty adjusting to dark light, blurry vision, dark spots or floaters and other complications.
In this new case report, doctors describe a 44-year-old woman who sought treatment following six months of worsening vision problems, including night vision problems. Even though she had stopped using Elmiron 38 months earlier, doctors determined she was suffering from Elmiron-induced maculopathy.
The doctors looked at her eye records from 25 months earlier, including color retina imaging, and there were no signs of maculopathy at that time. Therefore, they concluded that the vision problems developed after long after she’d stopped taking the drug.
“This case suggests that new onset clinically detectable pentosan polysulfate maculopathy may develop years after drug cessation,” Barnett and Jain concluded. “If corroborated, this finding has important ramifications for PPS dosing and surveillance guidelines.”
The findings may also impact the extent of damages the drug makers may have to pay in Elmiron settlements, with new claims likely to continue emerging for years and a number of class action lawsuits already seeking to establish funds for future medical monitoring of prior users.
Given common questions of fact and law raised in complaints filed throughout the federal court system, the U.S. Judicial Panel on Multidistrict Litigation has scheduled oral arguments later this week to evaluate whether the cases should be centralized for coordinated pretrial proceedings.
While there are currently about 100 complaints filed in U.S. District Courts nationwide, it is widely expected that the litigation will eventually involve several thousand cases as Elmiron lawyers continue to review and file claims in the coming months and years.
Want a weekly update on top lawsuits, recalls & warnings?
"*" indicates required fields






0 Comments